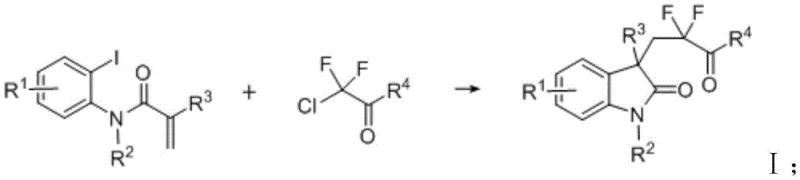
Scalable Nickel-Catalyzed Synthesis of Difluorocarbonylated Indolones for Commercial API Production
The pharmaceutical industry continuously seeks efficient pathways to access fluorinated heterocycles, particularly 3,3-disubstituted indolones, due to their enhanced bioavailability and metabolic stability in drug candidates. Patent CN114874127B introduces a transformative preparation method for difluorocarbonylated indolone compounds that addresses critical economic and operational bottlenecks in current synthetic routes. This technology leverages a robust nickel-catalyzed coupling system between enamide aryl iodides and difluoro compounds, utilizing a specific catalytic composition that includes nickel iodide, dual ligands, and a magnesium chloride co-catalyst. By shifting away from precious metal dependency, this innovation offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The method operates under remarkably mild conditions, typically around 60°C, which contrasts sharply with the harsh thermal requirements or complex equipment needed for alternative methodologies.
Furthermore, the broad substrate scope demonstrated in the patent data suggests that this protocol is not merely a niche solution but a versatile platform capable of generating diverse libraries of fluorinated scaffolds. For R&D teams focused on lead optimization, the ability to introduce difluoroalkyl groups with high regioselectivity at the C3 position of the indolone ring is a significant strategic advantage. The process utilizes commercially available reagents such as manganese powder as a reducing agent, which drastically simplifies the procurement landscape compared to specialized organometallic reagents. As we delve deeper into the technical specifics, it becomes evident that this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing practices for complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated indolone derivatives has relied heavily on methodologies that impose significant financial and logistical burdens on large-scale production. Prior art, such as the work by Wang et al. in 2016, utilized silver iodide (AgI) mediated direct difluoromethylation, which necessitates the use of stoichiometric amounts of expensive silver salts. Similarly, visible-light-induced protocols reported by Liu's team in 2019 depend on sophisticated photocatalysts and specialized irradiation equipment, creating barriers to entry for standard chemical manufacturing facilities. These conventional approaches often suffer from high operational expenditures due to the cost of catalysts and the energy intensity of maintaining specific photochemical environments. Moreover, the removal of heavy metal residues like silver from the final active pharmaceutical ingredient (API) intermediate requires additional purification steps, further eroding profit margins and extending production timelines.
Beyond the economic drawbacks, traditional methods frequently exhibit limited substrate tolerance, failing to accommodate bulky or electronically diverse substituents without significant drops in yield. The reliance on radical pathways in some photocatalytic methods can also lead to unpredictable side reactions, complicating the impurity profile and challenging quality control teams. For supply chain managers, the volatility in the price of precious metals like silver and palladium introduces unacceptable risk factors into long-term procurement contracts. Consequently, there is an urgent industry demand for a transition metal-catalyzed approach that utilizes earth-abundant metals, offers predictable scalability, and eliminates the need for exotic reaction conditions while maintaining high selectivity for the desired 3,3-disubstituted architecture.
The Novel Approach
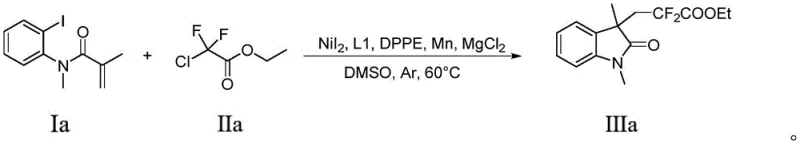
The methodology disclosed in CN114874127B fundamentally reengineers the synthetic route by employing a nickel-catalyzed reductive coupling strategy that is both economically viable and chemically robust. Instead of expensive silver or complex photoredox systems, this novel approach utilizes nickel iodide (NiI2) as the primary catalyst, supported by a synergistic ligand system comprising 2,2'-bipyridine-4,4'-dicarboxylic acid methyl ester (L1) and 1,2-bis(diphenylphosphine)ethane (DPPE). This specific combination facilitates the activation of the carbon-iodine bond in the enamide aryl iodide and the subsequent insertion of the difluoro moiety with high efficiency. The inclusion of magnesium chloride as a co-catalyst is a critical innovation, as comparative data within the patent indicates that its absence leads to a precipitous drop in yield from 63% to merely 5%, highlighting its essential role in stabilizing the catalytic cycle.
Operationally, this new method simplifies the workflow by conducting the reaction in common polar aprotic solvents like dimethyl sulfoxide (DMSO) at a moderate temperature of 60°C under an inert argon atmosphere. The use of manganese powder as a terminal reductant is a masterstroke in cost reduction, as it is significantly cheaper than zinc or other metallic reductants often used in similar cross-couplings. This approach not only lowers the direct material costs but also streamlines the downstream processing, as the reaction mixture can be quenched with standard aqueous ammonium chloride and extracted with ethyl acetate. For procurement managers, this translates to a reliable supply of high-purity intermediates without the volatility associated with precious metal markets, ensuring consistent availability for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
The catalytic cycle underpinning this transformation involves a sophisticated interplay between the nickel center, the dual ligand system, and the metal reductant. Initially, the nickel(II) precursor is reduced in situ by manganese powder to generate the active low-valent nickel(0) species, which is stabilized by the coordination of the bipyridine and phosphine ligands. This active catalyst then undergoes oxidative addition into the carbon-iodine bond of the enamide aryl iodide substrate, forming an aryl-nickel(II) intermediate. Subsequently, the difluoro compound, likely activated through single-electron transfer or halogen atom abstraction mechanisms facilitated by the nickel complex, interacts with this intermediate. The specific role of the magnesium chloride co-catalyst is believed to involve Lewis acid activation of the carbonyl oxygen or stabilization of the halide leaving group, thereby lowering the activation energy for the crucial C-C bond formation step.
Following the coupling event, the resulting nickel species undergoes reductive elimination to release the difluorocarbonylated indolone product and regenerate the nickel catalyst for the next turnover. Importantly, the patent data suggests that this process does not proceed via a free radical chain mechanism, as evidenced by the fact that adding the radical inhibitor BHT did not suppress the reaction yield significantly. This mechanistic distinction is vital for R&D directors concerned with safety and reproducibility, as it implies a controlled, catalyst-dependent pathway rather than a potentially hazardous uncontrolled radical propagation. The precise tuning of the ligand environment ensures that the nickel center remains active throughout the cycle, preventing catalyst deactivation through aggregation or formation of inactive nickel black, which is a common failure mode in base metal catalysis.
Understanding the impurity profile is equally critical for ensuring the final product meets stringent regulatory standards. The high selectivity of this nickel system minimizes the formation of homocoupling byproducts or defluorinated species that often plague fluorination reactions. The mild reaction temperature of 60°C further suppresses thermal decomposition pathways that could lead to complex, difficult-to-remove impurities. By controlling the molar ratios of the nickel catalyst, ligands, and co-catalyst within the optimized range of 1:1-3:30-40, manufacturers can achieve a clean reaction profile that simplifies the final purification via column chromatography or crystallization. This level of control over the chemical outcome directly supports the goal of producing high-purity pharmaceutical intermediates with minimal batch-to-batch variation.
How to Synthesize Difluorocarbonylated Indolones Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding reagent quality and atmospheric control to maximize yield and reproducibility. The process begins with the careful weighing of the nickel catalyst, ligands, and manganese powder, followed by the addition of the solvent and substrates under an inert gas line to exclude oxygen and moisture. Detailed standard operating procedures for the reaction setup, monitoring, and workup are essential for transferring this laboratory-scale success to pilot and commercial plants. The following guide outlines the critical operational parameters derived from the patent examples to ensure successful execution.
- Prepare the reaction mixture by combining nickel catalyst (NiI2), ligands (L1 and DPPE), manganese powder, magnesium chloride, enamide aryl iodide, and difluoro compound in DMSO under inert atmosphere.
- Stir the reaction mixture at 60°C for approximately 24 hours until TLC analysis indicates complete consumption of the starting material.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, dry over sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol offers tangible strategic benefits that extend far beyond simple reaction chemistry. The primary advantage lies in the drastic reduction of raw material costs achieved by substituting expensive silver salts and photocatalysts with commodity-grade nickel salts and manganese powder. This shift decouples the production cost from the volatile precious metal market, allowing for more stable long-term pricing agreements with customers. Additionally, the use of common solvents like DMSO and standard workup procedures reduces the complexity of waste management and solvent recovery systems, leading to substantial operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The elimination of high-cost catalysts such as silver iodide and specialized photocatalysts results in a significantly lower bill of materials for every kilogram of product produced. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the simplified purification process lowers the demand for expensive chromatography media or multiple recrystallization steps. These cumulative efficiencies translate into a much more competitive cost structure for the final API intermediate, enhancing margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: By relying on earth-abundant metals like nickel and manganese, the supply chain becomes more resilient to geopolitical disruptions that often affect the availability of rare earth elements or precious metals. The reagents required for this process are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This reliability ensures consistent production schedules and shorter lead times for high-purity pharmaceutical intermediates, which is critical for meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The protocol's compatibility with standard stainless steel reactors and its operation at moderate temperatures make it highly amenable to scale-up from gram to ton quantities without requiring specialized equipment investments. Moreover, the avoidance of toxic heavy metals like silver simplifies environmental compliance and waste disposal, aligning with increasingly stringent green chemistry regulations. This sustainability profile enhances the corporate social responsibility standing of the manufacturer and facilitates smoother regulatory approvals for new drug applications containing these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the key advantages of this nickel-catalyzed method over previous silver or photocatalytic methods?
A: This method replaces expensive silver salts and complex photocatalysts with inexpensive nickel catalysts and manganese powder, significantly reducing raw material costs while maintaining mild reaction conditions (60°C) and broad substrate tolerance.
Q: Is the reaction sensitive to air or moisture?
A: Yes, the protocol specifies carrying out the coupling reaction under an inert atmosphere, such as argon or nitrogen, to prevent oxidation of the low-valent nickel species and ensure high conversion rates.
Q: What is the typical yield range for this synthesis?
A: According to the patent examples, isolated yields typically range from 47% to 64%, depending on the specific substituents on the enamide aryl iodide and the difluoro compound used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluorocarbonylated Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative method are fully realized at an industrial level. Our state-of-the-art facilities are equipped to handle the specific requirements of air-sensitive nickel catalysis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for drug substance manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and scalable synthesis route for their fluorinated indolone projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to accelerate your development programs with a reliable supply of high-quality difluorocarbonylated indolone intermediates.
