Advanced Photocatalytic Synthesis of Alpha-Substituted Glycine Derivatives for Scalable Agrochemical Production
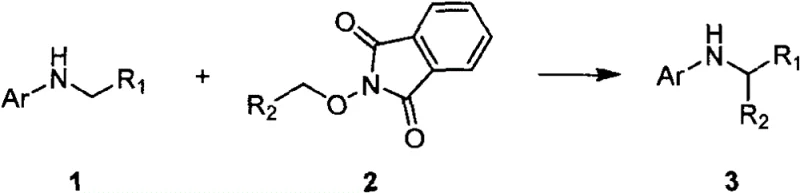
The landscape of agrochemical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic methodologies. Patent CN115611756A, published in early 2023, introduces a groundbreaking preparation method for alpha-substituted glycine derivatives that leverages visible-light-induced Single Electron Transfer (SET) processes. This technology represents a paradigm shift away from traditional, harsh chemical transformations, offering a pathway to construct complex non-natural amino acid scaffolds with exceptional precision. For R&D directors and procurement specialists in the agrochemical sector, this patent outlines a robust strategy for accessing high-value intermediates that serve as critical building blocks for next-generation fungicides. The core innovation lies in the utilization of stable N-alkoxyphthalimides as radical precursors, which couple efficiently with glycine derivatives under mild irradiation conditions. This approach not only simplifies the experimental operation but also drastically reduces the environmental footprint associated with synthetic chemistry, aligning perfectly with global sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-substituted glycine derivatives has relied heavily on classical organometallic reagents or thermal radical initiation methods that pose significant challenges for large-scale manufacturing. Traditional routes often involve the use of Grignard reagents or organolithium species, which require strictly anhydrous conditions, cryogenic temperatures, and generate substantial amounts of hazardous waste. Furthermore, these methods frequently suffer from poor chemoselectivity, leading to complex impurity profiles that are difficult and costly to remove during downstream processing. The reliance on stoichiometric amounts of reactive metals also introduces the risk of heavy metal contamination, a critical failure point for agrochemical products destined for field application where environmental toxicity is a major regulatory concern. Additionally, thermal radical methods often lack the spatial and temporal control offered by photochemistry, resulting in uncontrolled side reactions and lower overall yields when dealing with sensitive functional groups present in complex molecular architectures.
The Novel Approach
In stark contrast, the methodology disclosed in CN115611756A utilizes a photoredox catalytic cycle that operates under remarkably mild conditions, typically at 60°C in dimethyl sulfoxide (DMSO) under blue LED irradiation. This novel approach replaces dangerous stoichiometric reagents with a catalytic amount of an organic photocatalyst, such as 4CzIPN, which facilitates the generation of carbon-centered radicals via a controlled Single Electron Transfer mechanism. The use of N-alkoxyphthalimides as radical precursors is particularly advantageous because these compounds are stable, easy to prepare from readily available alcohols, and undergo predictable beta-scission to release the desired alkyl radicals. This strategy ensures excellent functional group compatibility, allowing for the direct modification of complex substrates including those derived from natural products like sclareolactone and camphor. By shifting to this photocatalytic regime, manufacturers can achieve higher purity profiles with significantly reduced safety risks, paving the way for cost reduction in agrochemical manufacturing through streamlined purification protocols.
Mechanistic Insights into Visible Light Induced SET Coupling
The mechanistic elegance of this transformation lies in the interplay between the excited state of the photocatalyst and the redox potentials of the substrates. Upon absorption of blue light photons, the 4CzIPN photocatalyst enters an excited state capable of acting as a potent single-electron oxidant or reductant. In this specific protocol, the catalyst facilitates the reduction of the N-alkoxyphthalimide substrate, triggering the fragmentation of the N-O bond. This cleavage event generates a phthalimide anion and a highly reactive alkoxy radical, which subsequently undergoes rapid beta-scission to produce a stable carbon-centered radical species. This carbon radical then attacks the alpha-position of the glycine derivative, forming the new carbon-carbon bond that defines the alpha-substituted structure. The catalytic cycle is closed by the regeneration of the ground-state photocatalyst, ensuring that only trace amounts of the catalyst are required to drive the reaction to completion. This precise control over radical generation minimizes homocoupling side reactions and ensures that the reactive intermediates are consumed efficiently by the intended coupling partner.
From an impurity control perspective, this mechanism offers distinct advantages over thermal methods. Because the radical flux is governed by the intensity of the light source rather than thermal energy, the concentration of free radicals in the solution remains low and steady. This "steady-state" radical concentration suppresses bimolecular termination events that typically lead to oligomerization or polymerization byproducts. Furthermore, the mild reaction temperature of 60°C prevents the thermal degradation of sensitive ester or amide functionalities often present in glycine derivatives. The result is a cleaner reaction profile with fewer side products, which translates directly to simplified workup procedures and higher isolated yields. For quality control teams, this means a more consistent product batch-to-batch, reducing the burden on analytical resources and accelerating the release of materials for biological testing or further synthesis.
How to Synthesize Alpha-Substituted Glycine Derivatives Efficiently
The practical implementation of this photocatalytic method is straightforward and amenable to standard laboratory equipment, requiring no specialized high-pressure reactors. The process begins with the dissolution of the glycine derivative and the N-alkoxyphthalimide precursor in DMSO, followed by the addition of the 4CzIPN catalyst. The reaction mixture is degassed to remove oxygen, which can quench the excited state of the catalyst, and then subjected to blue LED irradiation. Monitoring is typically performed via TLC until the starting material is fully consumed. The detailed standardized synthesis steps, including specific molar ratios and purification parameters for scaling this reaction, are outlined in the guide below.
- Prepare the reaction mixture by dissolving the glycine derivative substrate, N-alkoxyphthalimide radical precursor, and 4CzIPN photocatalyst in anhydrous DMSO under an inert nitrogen atmosphere.
- Seal the reaction vessel and irradiate with blue LEDs while maintaining a temperature of 60°C to initiate the Single Electron Transfer (SET) process and radical coupling.
- Upon completion, quench the reaction with water, extract with dichloromethane, dry the organic layer, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift from stoichiometric metal reagents to catalytic organic systems fundamentally alters the cost structure of production by eliminating expensive metal salts and the associated waste disposal costs. Moreover, the mild reaction conditions reduce the energy load on manufacturing facilities, as there is no need for cryogenic cooling or high-temperature heating, contributing to significant operational expenditure savings. The robustness of the method also enhances supply chain reliability by reducing the dependency on volatile or hazardous raw materials that are subject to strict transportation regulations. By simplifying the synthesis of complex intermediates, companies can shorten their production lead times and respond more agilely to market demands for new agrochemical active ingredients.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet stringent regulatory limits on heavy metal residues in agrochemicals. This simplification of the downstream processing workflow leads to substantial cost savings in both materials and labor. Additionally, the use of commercially available and stable N-alkoxyphthalimides as starting materials ensures a consistent and affordable supply chain for raw materials, avoiding the price volatility often associated with specialized organometallic reagents. The high atom economy of the radical coupling reaction further contributes to cost efficiency by maximizing the conversion of starting materials into the desired product.
- Enhanced Supply Chain Reliability: The operational simplicity of this photocatalytic method allows for greater flexibility in manufacturing scheduling and facility utilization. Since the reaction does not require extreme temperatures or pressures, it can be run in standard glass-lined or stainless steel reactors equipped with LED arrays, reducing the need for specialized capital investment. The stability of the reagents under ambient storage conditions also mitigates the risk of supply disruptions caused by the degradation of sensitive chemicals during transport or warehousing. This reliability ensures a continuous flow of high-purity agrochemical intermediates, supporting uninterrupted production schedules for downstream formulation plants.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but modern flow chemistry technologies and efficient LED arrays have largely solved this bottleneck, allowing for seamless translation from bench to pilot scale. The green nature of this synthesis, utilizing organic catalysts and generating minimal hazardous waste, aligns perfectly with increasingly strict environmental regulations governing chemical manufacturing. This compliance reduces the regulatory burden and potential fines associated with waste discharge, while also enhancing the corporate sustainability profile of the manufacturer. The ability to produce complex molecules with a lower environmental footprint is a significant competitive advantage in the global agrochemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or R&D pipelines.
Q: What are the primary advantages of using 4CzIPN over traditional transition metal catalysts?
A: 4CzIPN is an organic photocatalyst that eliminates the risk of heavy metal contamination in the final product, which is critical for agrochemical and pharmaceutical applications. It also operates under milder conditions compared to thermal radical initiators, improving functional group tolerance.
Q: Can this method be applied to complex natural product scaffolds?
A: Yes, the patent demonstrates successful modification of complex natural products like sclareolactone and beta-pinene derivatives. The mild radical conditions allow for late-stage functionalization without degrading sensitive structural motifs.
Q: What is the biological activity profile of the synthesized derivatives?
A: The synthesized alpha-substituted glycine derivatives exhibit significant inhibitory activity against plant pathogenic fungi, specifically Sclerotinia sclerotiorum and Rhizoctonia solani, making them valuable candidates for new fungicide development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Substituted Glycine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in the synthesis of high-value agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115611756A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications required for global agrochemical registration. We are committed to delivering high-purity agrochemical intermediates that empower our clients to develop safer and more effective crop protection solutions.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this photocatalytic route can optimize your manufacturing economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, scalable, and sustainable chemical solutions.
