Advanced Copper-Catalyzed Synthesis of Fluorinated Indole Intermediates for Commercial Scale-Up
The pharmaceutical and agrochemical industries are increasingly reliant on the strategic incorporation of fluorine atoms into organic scaffolds to enhance metabolic stability, lipophilicity, and bioavailability. Patent CN107522648B introduces a groundbreaking synthetic methodology for producing 3-α-difluoromethyl-α-trifluoromethyl-methanol indoles and their derivatives, addressing a critical gap in the availability of these high-value building blocks. This technology leverages a copper-catalyzed system to directly functionalize the indole core using pentafluoropropenyloxytrimethylsilane, a reagent that serves as a versatile source of fluorinated carbon fragments. For R&D directors and procurement specialists, this patent represents a significant opportunity to access complex fluorinated intermediates through a streamlined, cost-effective pathway that avoids the pitfalls of traditional multi-step fluorination sequences. The robustness of this chemistry suggests immediate applicability in the development of next-generation bioactive molecules where precise control over fluorine placement is paramount for efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of polyfluoroalkyl groups onto heterocyclic systems like indoles has been fraught with significant technical challenges that hinder efficient manufacturing. Conventional strategies often rely on harsh electrophilic fluorinating agents or require pre-functionalized starting materials that are expensive and difficult to source on a large scale. Many existing protocols necessitate extreme reaction conditions, such as cryogenic temperatures or high-pressure environments, which drastically increase energy consumption and capital expenditure for reactor infrastructure. Furthermore, traditional methods frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that require tedious and yield-eroding purification steps. The use of stoichiometric amounts of heavy metal reagents or toxic fluorine sources also raises substantial environmental and safety concerns, complicating waste disposal and regulatory compliance for commercial production facilities. These limitations collectively create a bottleneck in the supply chain for fluorinated pharmaceutical intermediates, driving up costs and extending lead times for drug development programs.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN107522648B offers a remarkably elegant and practical solution by utilizing a catalytic amount of inexpensive copper salts to drive the reaction. This novel approach enables the direct coupling of readily available indoles with pentafluoropropenyloxytrimethylsilane under mild thermal conditions, typically around 80°C in common organic solvents like 1,2-dichloroethane. The reaction proceeds with exceptional efficiency, often delivering isolated yields exceeding 90% for a wide range of substrates, which is a testament to its high atom economy and selectivity. By avoiding the need for exotic reagents or extreme conditions, this method significantly simplifies the operational complexity of the synthesis, making it highly attractive for process chemistry teams aiming to reduce manufacturing costs. The ability to tolerate diverse functional groups on the indole ring further enhances its utility, allowing for the late-stage functionalization of complex molecular architectures without the need for extensive protecting group strategies.

The mechanistic underpinnings of this copper-catalyzed transformation involve the activation of the silicon-carbon bond in the pentafluoropropenyloxytrimethylsilane reagent, facilitating a nucleophilic attack on the electron-rich C3 position of the indole ring. The copper catalyst likely plays a dual role in coordinating the fluorinated species and stabilizing the transition state, thereby lowering the activation energy required for the C-C bond formation. This catalytic cycle is highly efficient, minimizing the generation of metal-containing waste and ensuring that the reaction proceeds cleanly to the desired alcohol product. Following the initial fluorination, the resulting 3-α-difluoromethyl-α-trifluoromethyl-methanol indoles can be further derivatized into valuable difluoromethyldiacetonitrile compounds through a subsequent base-mediated reaction with potassium tert-butoxide. This secondary transformation expands the chemical space accessible from this platform, providing medicinal chemists with a versatile toolkit for exploring structure-activity relationships in fluorinated drug candidates.
From an impurity control perspective, the high selectivity of this copper-catalyzed system is a major advantage for ensuring product quality and consistency. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups, such as esters, ethers, and halides, which might otherwise degrade under more aggressive fluorination protocols. This inherent stability reduces the formation of side products and degradation impurities, simplifying the downstream purification process and improving the overall purity profile of the final API intermediate. For quality assurance teams, this means a more robust analytical profile with fewer unknown peaks to identify and quantify, accelerating the regulatory filing process. The use of standard chromatographic techniques for purification further ensures that the process can be easily validated and transferred between different manufacturing sites without significant re-optimization.
Mechanistic Insights into Copper-Catalyzed Fluorination
The core of this innovation lies in the unique reactivity of the copper catalyst in mediating the insertion of the fluorinated moiety. Unlike radical-based fluorination methods that can lead to indiscriminate substitution patterns, this ionic or organometallic pathway ensures precise regiocontrol at the indole C3 position. The electronic nature of the pentafluoropropenyloxy group allows for a controlled release of the fluorinated fragment, which then adds across the indole double bond in a highly concerted manner. This mechanism is supported by the broad substrate scope observed in the patent examples, where both electron-rich and electron-deficient indoles participate effectively in the reaction. Understanding this mechanistic nuance is crucial for process engineers, as it highlights the importance of maintaining strict anhydrous conditions and optimal catalyst loading to maximize turnover numbers and minimize catalyst deactivation.
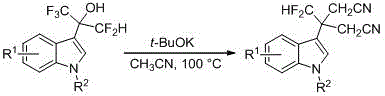
Furthermore, the subsequent conversion of the fluorinated alcohol to the dinitrile derivative showcases the versatility of the intermediate. This transformation involves a base-promoted rearrangement or substitution that installs two nitrile groups, significantly altering the electronic and steric properties of the molecule. Such structural modifications are invaluable in drug design, as nitrile groups can serve as hydrogen bond acceptors or precursors to other functional groups like amines or carboxylic acids. The ability to access these distinct chemical motifs from a common intermediate streamlines the synthesis of compound libraries, enabling rapid iteration during the lead optimization phase. For supply chain managers, this modularity means that a single bulk production run of the fluorinated alcohol can support multiple downstream projects, optimizing inventory management and reducing the risk of stockouts for critical research materials.
How to Synthesize 3-alpha-difluoromethyl-indole Efficiently
To implement this synthesis effectively, operators must adhere to precise procedural guidelines regarding reagent quality and reaction monitoring. The protocol specifies the use of ultra-dry solvents and inert atmosphere conditions to prevent hydrolysis of the silyl reagent, which is critical for maintaining high yields. Detailed standardized synthesis steps for this procedure are provided in the guide below, ensuring reproducibility across different laboratory scales.
- React substituted indoles with pentafluoropropenyloxytrimethylsilane using a copper salt catalyst in 1,2-dichloroethane at 80°C.
- Perform aqueous workup and silica gel column chromatography to isolate the fluorinated alcohol intermediate.
- Treat the intermediate with potassium tert-butoxide in dry acetonitrile at 100°C to generate difluoromethyldiacetonitrile derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The reliance on commodity chemicals like copper salts and common chlorinated solvents eliminates the dependency on specialized, high-cost fluorinating agents that are often subject to volatile market pricing and supply constraints. This shift to readily available raw materials significantly de-risks the supply chain, ensuring consistent availability of key inputs even during periods of global chemical shortages. Moreover, the mild reaction temperatures reduce energy consumption compared to high-temperature or cryogenic processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility. These factors combine to create a more resilient and cost-efficient production model that aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric fluorinating reagents and the use of catalytic amounts of cheap copper salts result in substantial raw material cost savings. Additionally, the high yields and simplified purification requirements reduce solvent usage and waste disposal costs, further enhancing the overall economic viability of the process. This cost structure allows for more competitive pricing of the final intermediates, providing a strategic advantage in tender negotiations with pharmaceutical clients.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted indoles and trimethylsilyl reagents, the process mitigates the risk of supply disruptions associated with niche chemicals. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from multiple suppliers. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for clinical trial materials.
- Scalability and Environmental Compliance: The straightforward workup procedure involving simple extraction and chromatography facilitates easy scale-up from gram to kilogram quantities without complex engineering changes. The absence of highly toxic or hazardous reagents simplifies environmental health and safety compliance, reducing the regulatory burden and permitting timelines for new production lines. This scalability ensures that the technology can seamlessly transition from R&D to commercial manufacturing, supporting the long-term growth of the product portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and scope defined within the patent documentation to provide accurate guidance for potential partners.
Q: What are the key advantages of this copper-catalyzed fluorination method?
A: The method utilizes cheap and readily available copper salts and operates under mild conditions (80°C), offering excellent functional group tolerance and high isolated yields compared to traditional harsh fluorination techniques.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the process uses common solvents like 1,2-dichloroethane and standard heating conditions, making it highly suitable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Q: What is the scope of substrates compatible with this reaction?
A: The protocol demonstrates broad compatibility with various substituted indoles, including those with electron-donating groups like methoxy and electron-withdrawing groups like halogens and nitro groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-alpha-difluoromethyl-indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed fluorination technology in advancing the development of novel therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of fluorinated indole intermediate meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this process for your specific needs, delivering high-quality materials that accelerate your drug development timeline.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the production of complex fluorinated pharmaceutical intermediates.
