Advanced Liquid-Phase Fluorination for High-Purity Ion Exchange Resin Monomers
The chemical industry is currently witnessing a significant paradigm shift in the production of high-performance fluorinated materials, driven by the urgent need for safer and more economically viable synthetic routes. Patent CN1816521A introduces a groundbreaking methodology for the production of fluorinated sulfonyl fluorides, which serve as critical precursors for advanced ion exchange resins and fluoropolymers. Unlike traditional methods that rely heavily on the hazardous handling of sulfur trioxide (SO3) or suffer from limited structural diversity, this invention leverages a sophisticated liquid-phase fluorination technique combined with oxidative sulfonation. This approach not only mitigates the severe safety risks associated with gaseous SO3 but also expands the accessible chemical space, allowing for the efficient synthesis of complex molecular architectures with multiple fluorosulfonyl groups. For R&D directors and procurement specialists alike, understanding this technology is paramount, as it represents a tangible leap forward in process safety and supply chain resilience for high-purity fluorine materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated sulfonyl fluorides has been plagued by significant operational hurdles and safety concerns that hinder large-scale industrial implementation. Traditional pathways often involve the reaction of tetrafluoroethylene with sulfur trioxide to form cyclic intermediates, which are subsequently reacted with perfluoroalkylene oxides. However, this route necessitates extreme caution in handling SO3, a corrosive and volatile reagent that poses substantial risks to personnel and equipment integrity. Furthermore, the economic viability of these legacy processes is compromised by the difficulty in synthesizing specific starting materials and the inherent limitation of the final products to compounds possessing specific side chains, such as trifluoromethyl groups. These structural constraints inevitably impact the performance characteristics of the resulting ion exchange membranes, limiting their applicability in demanding electrochemical environments where tailored membrane properties are essential for optimal efficiency and longevity.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a robust sequence starting from readily available sulfur-containing organic compounds. By employing halogen-based oxidants, such as chlorine in aqueous solvents, the process efficiently converts sulfide precursors into sulfonyl halides, which are then transformed into the desired sulfonyl fluorides. This strategy eliminates the need for direct SO3 handling entirely, thereby simplifying the reactor design and safety protocols required for production. Moreover, the subsequent liquid-phase fluorination step allows for precise control over reaction conditions, enabling the introduction of fluorine atoms into complex organic backbones with high selectivity. This flexibility empowers manufacturers to produce a diverse array of molecular structures with arbitrary numbers of fluorosulfonyl groups, directly addressing the market demand for customized high-purity fluorine materials tailored for next-generation ion exchange applications.
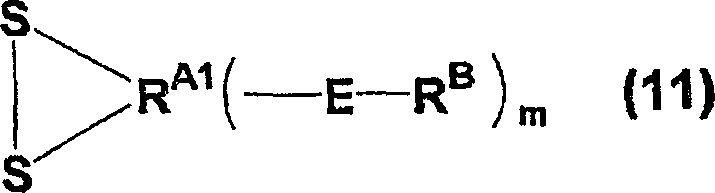
Mechanistic Insights into Oxidative Sulfonation and Liquid-Phase Fluorination
The core of this technological advancement lies in the meticulous orchestration of oxidative sulfonation followed by liquid-phase fluorination, a combination that ensures both high yield and exceptional purity. The mechanism initiates with the oxidation of a sulfur-containing precursor, such as a dithiolane derivative or a sulfide, using an oxidizing agent containing halogens as an essential component. In the presence of water, chlorine reacts with the sulfur moiety to cleave carbon-sulfur bonds or oxidize sulfides directly into sulfonyl chloride groups. This intermediate is then subjected to a halogen exchange reaction, typically utilizing potassium hydrogen fluoride or similar fluoride sources, to replace the chlorine atoms with fluorine, yielding the stable sulfonyl fluoride functionality. This step is critical as it establishes the thermal and chemical stability required for the subsequent aggressive fluorination conditions, ensuring that the sulfonyl groups remain intact while the rest of the molecule undergoes perfluorination.
Following the formation of the sulfonyl fluoride precursor, the process advances to the liquid-phase fluorination stage, where elemental fluorine gas, often diluted with inert gases like nitrogen, is introduced into a solvent system containing the substrate. This reaction is conducted under controlled temperatures, typically ranging from -20°C to +50°C, to manage the exothermic nature of fluorine addition. The solvent, such as R-113 or other fluorinated hydrocarbons, plays a pivotal role in dissipating heat and maintaining the substrate in solution, thereby preventing localized hot spots that could lead to decomposition. The result is a fully perfluorinated intermediate containing both sulfonyl fluoride and ester linkages. Finally, a thermal decomposition step at elevated temperatures, preferably between 150°C and 250°C, cleaves the ester bonds to generate the terminal acid fluoride groups. This multi-stage mechanism ensures that the final product possesses the precise structural attributes necessary for high-performance polymerization into ion exchange membranes.
How to Synthesize Fluorinated Sulfonyl Fluorides Efficiently
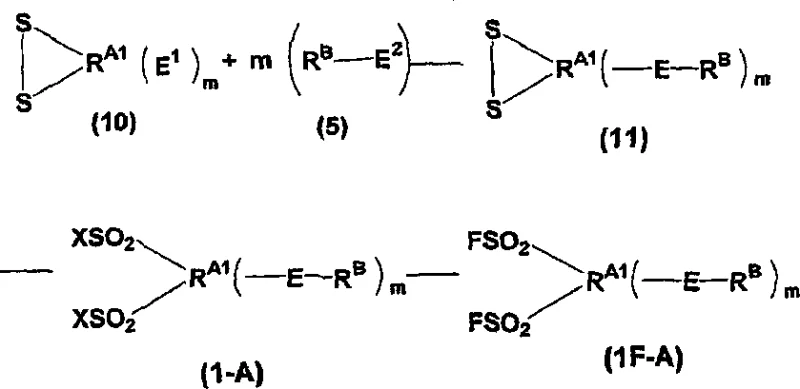
The synthesis of these specialized fluorinated compounds requires a deep understanding of reaction kinetics and hazard management to ensure consistent quality and safety. The process begins with the preparation of the sulfur-containing backbone, followed by oxidation and fluorination steps that must be tightly controlled to avoid side reactions. Detailed standard operating procedures regarding reagent stoichiometry, temperature ramping rates, and quenching protocols are essential for replicating the high yields reported in the patent examples. For technical teams looking to implement this chemistry, adherence to the specific solvent systems and catalyst loadings described is crucial for maximizing the conversion of starting materials into the desired perfluorinated intermediates.
- Oxidize sulfur-containing precursors (such as dithiolanes or sulfides) using halogen-based oxidants like chlorine in aqueous solvents to generate sulfonyl halides.
- Convert sulfonyl chlorides to sulfonyl fluorides using fluoride sources like potassium hydrogen fluoride, followed by liquid-phase fluorination with diluted fluorine gas.
- Perform thermal decomposition of the resulting perfluoro esters at elevated temperatures (150-250°C) to cleave ester bonds and generate the final acid fluoride functionality.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this manufacturing process offers profound advantages that extend well beyond mere technical feasibility, directly impacting the bottom line and supply chain stability for global buyers. The ability to utilize readily available raw materials, such as simple sulfides and common halogenating agents, significantly reduces the dependency on exotic or hard-to-source reagents that often bottleneck production schedules. This accessibility translates into a more resilient supply chain, capable of withstanding market fluctuations and geopolitical disruptions that frequently affect the availability of specialized fluorine chemicals. Furthermore, the process design inherently supports scalability, moving seamlessly from laboratory benchtop experiments to multi-ton annual commercial production without the need for prohibitively expensive specialized equipment required for handling gaseous sulfur trioxide.
- Cost Reduction in Manufacturing: One of the most compelling economic benefits of this technology is the inherent capability for by-product recycling, which drastically lowers the effective cost of raw materials. The patent explicitly demonstrates that decomposition by-products, such as perfluoro acid fluorides, can be recovered and reused as starting materials in earlier stages of the synthesis, such as the esterification step. This closed-loop approach minimizes waste generation and reduces the volume of fresh reagents required per unit of output, leading to substantial cost savings in fluorine materials manufacturing. By eliminating the need for expensive transition metal catalysts and reducing waste disposal fees associated with hazardous sulfur by-products, the overall production economics become significantly more favorable compared to conventional methods.
- Enhanced Supply Chain Reliability: The reliance on stable, storable intermediates rather than unstable gases enhances the reliability of the supply chain for high-purity fluorine materials. Traditional methods often require the just-in-time generation of reactive species, creating vulnerabilities in the production schedule. In contrast, this process generates stable sulfonyl fluoride intermediates that can be stockpiled and transported safely, decoupling the production of precursors from the final fluorination step. This flexibility allows manufacturers to maintain strategic inventory levels, ensuring continuous supply to downstream customers even during periods of maintenance or unexpected feedstock shortages. The robustness of the liquid-phase fluorination step further ensures consistent batch-to-batch quality, reducing the risk of supply interruptions due to off-specification product.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with increasingly stringent global regulations regarding industrial emissions and waste management. By avoiding the use of sulfur trioxide and minimizing the generation of acidic waste streams through efficient recycling loops, the process reduces the environmental footprint of fluorine materials production. The liquid-phase nature of the key fluorination step facilitates better containment of fluorine gas, minimizing fugitive emissions and enhancing workplace safety. This compliance advantage not only mitigates regulatory risk but also appeals to environmentally conscious end-users in the automotive and energy sectors who prioritize sustainable sourcing. The scalability of the reactor systems used in this process ensures that production capacity can be expanded to meet growing demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
Understanding the nuances of this advanced synthesis technology is critical for stakeholders evaluating its potential impact on their operations. The following questions address common inquiries regarding the technical feasibility, safety protocols, and commercial implications of the process described in CN1816521A. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees.
Q: What are the primary safety advantages of the liquid-phase fluorination method described in CN1816521A?
A: The liquid-phase method allows for better heat dissipation and temperature control compared to gas-phase reactions, significantly reducing the risk of thermal runaway when handling highly reactive elemental fluorine.
Q: How does this process improve cost efficiency for ion exchange resin manufacturing?
A: The process enables the recycling of by-products such as perfluoro acid fluorides back into the synthesis loop as starting materials, drastically reducing raw material consumption and waste disposal costs.
Q: Can this method produce compounds with multiple sulfonyl fluoride groups?
A: Yes, the patent specifically describes methods to introduce two or more fluorosulfonyl groups onto a single organic backbone, which is critical for creating cross-linked ion exchange membranes with superior mechanical properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Sulfonyl Fluoride Supplier
As the global demand for high-performance ion exchange membranes continues to surge, particularly in the fuel cell and chlor-alkali industries, securing a reliable fluorinated sulfonyl fluoride supplier is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of fluorinated intermediate meets the exacting standards required for polymerization into defect-free membranes. We understand that the complexity of fluorine chemistry demands a partner with deep technical expertise, and our team is dedicated to optimizing these advanced synthetic routes for maximum efficiency and safety.
We invite industry leaders to collaborate with us to unlock the full potential of this innovative manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive down costs and accelerate the deployment of next-generation fluoropolymer solutions, ensuring your supply chain remains robust and competitive in a rapidly evolving market.
