Advanced Palladium-Catalyzed Synthesis of Z-Bisfuryl Imines for Commercial Pharmaceutical Applications
Introduction to Novel Z-Bisfuryl Imine Synthesis Technology
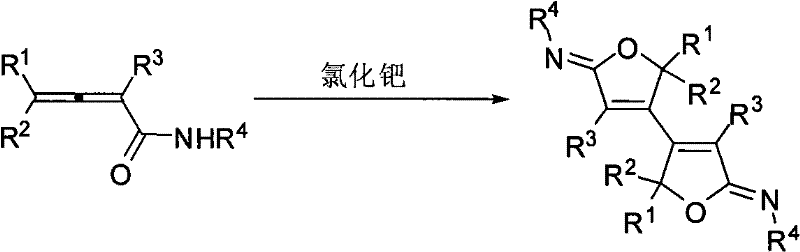
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN101508688B introduces a groundbreaking synthesis method for Z-bisfuryl imine compounds, which are recognized as one of the most important intermediates in organic synthesis and common structural units in natural products. This technology leverages a palladium-catalyzed self-ring-closing coupling reaction of 2,3-allenamides, operating under the influence of specific additives like potassium carbonate and sodium iodide. The significance of this invention cannot be overstated, as it provides a highly regioselective pathway to access these valuable compounds, which possess immense potential for development in biotechnology, medicine, and agrochemical applications. Prior to this disclosure, the industry faced a significant gap in reliable synthetic routes for this specific class of imines, making this patent a pivotal resource for R&D teams aiming to expand their library of functionalized heterocycles.
Furthermore, the operational simplicity of this method makes it particularly attractive for large-scale manufacturing environments where reproducibility and safety are paramount. The reaction utilizes readily available raw materials and reagents, such as dimethylformamide or dimethylacetamide as solvents, which are standard in industrial chemical processing. The ability to introduce various substituents, including hydrogen, benzyl, and alkyl groups ranging from C1 to C12, allows for significant structural diversity, enabling chemists to tailor the physicochemical properties of the final product to meet specific drug design requirements. This flexibility ensures that the process is not limited to a single compound but serves as a versatile platform technology for generating a wide array of substituted Z-bisfuryl imine derivatives, thereby supporting the continuous innovation needed in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Z-bisfuryl imine compounds has been plagued by a distinct lack of efficient and direct methodologies, creating a bottleneck for researchers attempting to incorporate this privileged scaffold into their target molecules. Before the advent of the technology described in CN101508688B, there was effectively no established method for synthesizing such compounds, forcing chemists to rely on convoluted multi-step sequences or less selective reactions that often resulted in complex mixtures of isomers. Traditional approaches to forming furan rings and imine linkages separately often suffered from poor regiocontrol, leading to difficult purification processes and significantly reduced overall yields. The absence of a dedicated catalytic system meant that harsh conditions were frequently required, which could degrade sensitive functional groups present on the substrate, limiting the scope of compatible starting materials. These limitations not only increased the cost of goods but also extended the timeline for project completion, hindering the rapid iteration necessary in competitive pharmaceutical development.
The Novel Approach
In stark contrast to the historical challenges, the novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed system that enables a direct self-cyclization coupling of 2,3-allenamides. This method transforms a potentially unstable allene precursor directly into the stable and highly functionalized Z-bisfuryl imine structure in a single operational step. The use of palladium chloride as the catalyst, combined with the synergistic effects of potassium carbonate and sodium iodide additives, creates a unique reaction environment that promotes high regioselectivity. This means that the reaction preferentially forms the desired Z-isomer over other possible geometric isomers, drastically simplifying the downstream purification process. The reaction conditions are relatively mild, typically conducted at 80°C, which preserves the integrity of sensitive substituents and allows for a broader substrate scope. This streamlined approach represents a paradigm shift in how these intermediates are accessed, offering a clear path from simple starting materials to high-value complex structures.
Mechanistic Insights into PdCl2-Catalyzed Cyclization
The core of this technological advancement lies in the intricate mechanistic pathway facilitated by the palladium catalyst. The reaction initiates with the coordination of the palladium species to the electron-rich allene system of the 2,3-allenamide substrate. Under the influence of the iodide ions provided by sodium iodide, the palladium center becomes more nucleophilic or electrophilic as required to activate the specific pi-system of the allene. This activation triggers an intramolecular nucleophilic attack by the amide oxygen or nitrogen onto the activated allene carbon, initiating the ring-closing event that forms the furan moiety. The presence of potassium carbonate acts as a base to neutralize any acidic byproducts generated during the cycle, maintaining the optimal pH for the catalytic turnover. The subsequent dimerization or coupling step links two of these furan-containing intermediates through an imine bond, locking the structure into the thermodynamically favorable Z-configuration. This precise control over the stereochemistry is a hallmark of the catalyst system, ensuring that the final product possesses the specific spatial arrangement required for biological activity.
Understanding the impurity profile is equally critical for R&D directors focused on product quality. The high regioselectivity of this palladium-catalyzed process inherently minimizes the formation of structural isomers and oligomeric byproducts that typically plague non-catalytic thermal cyclizations. By controlling the stoichiometry of the additives and the catalyst loading, which can be as low as 0.01 equivalents, the reaction pathway is tightly regulated to favor the desired bimolecular coupling. Any minor impurities that do form are typically structurally similar to the starting material or simple hydrolysis products, which are easily removed during the standard aqueous workup and column chromatography steps described in the patent examples. This clean reaction profile translates directly to higher purity specifications for the final API intermediate, reducing the burden on analytical teams to identify and quantify trace unknowns. The mechanism ensures that the energy barrier for the desired pathway is significantly lower than competing side reactions, providing a robust and predictable chemical transformation suitable for rigorous quality control standards.
How to Synthesize Z-Bisfuryl Imine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific ratios of reagents and the thermal profile of the reaction. The patent outlines a generalized procedure where the 2,3-allenamide substrate is dissolved in a polar aprotic solvent such as dimethylformamide (DMF) or dimethylacetamide (DMAc) to achieve a concentration of approximately 0.2 mol/L. To this solution, the catalytic system comprising palladium chloride and the additive package of potassium carbonate and sodium iodide are added. The detailed standardized synthesis steps for this process are provided in the guide below, ensuring that operators can replicate the high yields and selectivity reported in the intellectual property documentation. Adhering to these parameters is essential for maximizing the efficiency of the transformation and ensuring consistent batch-to-batch quality.
- Combine potassium carbonate, sodium iodide, palladium chloride catalyst, 2,3-allenamide substrate, and DMF or DMAc solvent in a reaction vessel.
- Heat the reaction mixture in an oil bath at 80°C and stir continuously for a duration ranging from 8 to 30 hours depending on the specific substrate substituents.
- Quench the reaction with water, extract the organic phase using diethyl ether, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers substantial strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the significant cost reduction in fine chemical manufacturing achieved through the elimination of complex multi-step sequences. By consolidating the formation of the furan rings and the imine linkage into a single catalytic pot, the process drastically reduces the consumption of solvents, reagents, and labor hours associated with intermediate isolation and purification. Furthermore, the use of a palladium catalyst at low loading levels minimizes the expense associated with precious metals, while the simple aqueous workup allows for the efficient recovery and recycling of organic solvents. These factors combine to lower the overall cost of goods sold, making the final Z-bisfuryl imine intermediates more price-competitive in the global market without sacrificing quality or performance.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the high atom economy of the self-coupling reaction and the low catalyst loading requirements. Unlike traditional methods that might require stoichiometric amounts of expensive activating agents or protecting group manipulations, this catalytic cycle turns over efficiently, meaning less waste is generated per kilogram of product. The reduction in processing steps directly correlates to lower utility costs and reduced equipment occupancy time, allowing manufacturing facilities to increase their throughput capacity. Additionally, the ease of product separation via standard extraction and chromatography techniques reduces the need for specialized distillation or crystallization equipment, further lowering capital expenditure requirements for production lines dedicated to these intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available starting materials such as 2,3-allenamides and common inorganic salts ensures a stable and resilient sourcing strategy. The robustness of the reaction conditions, which tolerate a wide range of substituents and function well in standard industrial solvents, mitigates the risk of batch failures due to minor variations in raw material quality. This reliability translates to shorter lead times for high-purity pharmaceutical building blocks, as manufacturers can confidently schedule production runs without fearing unpredictable yields or extensive rework. The ability to scale this chemistry from gram to kilogram scales with minimal optimization ensures that supply can be ramped up quickly to meet sudden increases in demand from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is notably lower than conventional alternatives, aligning with the increasing regulatory pressure for greener chemical manufacturing. The reaction generates minimal hazardous waste, and the use of recyclable solvents like DMF or DMAc fits well within existing solvent recovery infrastructure. The high selectivity of the reaction reduces the generation of toxic byproducts, simplifying wastewater treatment and disposal protocols. This compliance with environmental standards not only avoids potential regulatory fines but also enhances the corporate sustainability profile of the manufacturer. The scalability is further supported by the exothermic nature of the reaction being manageable at 80°C, allowing for safe operation in large-scale reactors without the need for extreme cooling or heating capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this Z-bisfuryl imine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the fit of this chemistry within their existing portfolios and supply chains.
Q: What represents the primary innovation in patent CN101508688B regarding Z-bisfuryl imine synthesis?
A: The primary innovation lies in the development of the first effective method for synthesizing Z-bisfuryl imine compounds via a palladium-catalyzed self-ring-closing coupling reaction of 2,3-allenamides, addressing a previous lack of synthetic routes for this important structural unit.
Q: How does the use of sodium iodide and potassium carbonate additives impact the reaction outcome?
A: The additives potassium carbonate and sodium iodide play a critical role in facilitating the catalytic cycle and enhancing the regioselectivity of the reaction, ensuring the formation of the desired Z-isomer with high purity and minimizing side product formation.
Q: What are the typical yield ranges observed for this palladium-catalyzed cyclization process?
A: Experimental data from the patent indicates that the reaction consistently achieves yields ranging from 50% to 90%, demonstrating robust efficiency across various substituted 2,3-allenamide substrates including benzyl, alkyl, and bulky tert-butyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Z-Bisfuryl Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our commitment to stringent purity specifications and the operation of rigorous QC labs guarantees that every batch of Z-bisfuryl imine supplied meets the highest international standards. We understand that consistency is key in pharmaceutical manufacturing, and our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including efficient metal scavenging and solvent recovery systems.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain partner dedicated to accelerating your time-to-market while optimizing your overall production costs through innovative chemical solutions.
