Advanced Synthesis of Substituted Amantadine Compounds for Commercial Pharmaceutical Production
Advanced Synthesis of Substituted Amantadine Compounds for Commercial Pharmaceutical Production
The pharmaceutical landscape for neuroprotective agents has been significantly transformed by the development of efficient synthetic routes for adamantane derivatives, particularly those outlined in patent CN100345819C. This pivotal intellectual property details a robust preparation method for substituted symmetrel compounds, such as Memantine hydrochloride, which serves as a critical NMDA receptor antagonist for treating Alzheimer's disease and vascular dementia. Traditional manufacturing pathways have long struggled with severe environmental burdens and operational complexities, creating a pressing demand for innovation among global reliable pharmaceutical intermediates suppliers. The disclosed technology addresses these challenges by introducing a streamlined two-step protocol that replaces hazardous sulfuric acid-mediated Ritter reactions with a safer nucleophilic substitution strategy. By shifting the paradigm from dangerous acidolysis to controlled amidation and hydrolysis, this method not only enhances the safety profile of the manufacturing plant but also drastically improves the economic viability of producing high-value antiviral and anti-dementia agents. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a stable supply chain of high-purity pharmaceutical intermediates that meet stringent regulatory standards while minimizing ecological footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of memantine and related adamantane amines relied heavily on the Ritter reaction, a process fraught with significant logistical and environmental drawbacks that hindered efficient cost reduction in pharmaceutical intermediates manufacturing. As documented in prior art such as US3391142, the conventional approach necessitates the use of massive quantities of concentrated sulfuric acid to convert bromo-adamantane into acetamido-adamantane, followed by a perilous deacetylation step in glycol ether under alkaline conditions. This legacy methodology presents a triple threat to modern chemical plants: it generates vast amounts of acidic wastewater that require expensive treatment, involves highly exothermic and dangerous reaction conditions that pose safety risks to personnel, and utilizes costly solvents that inflate the raw material budget. Furthermore, the deacetylation step often requires elevated temperatures and extended reaction times, which inadvertently promote the formation of undesirable by-products, thereby complicating the purification process and reducing the overall yield of the final active pharmaceutical ingredient. These cumulative inefficiencies render the traditional route increasingly obsolete in an era where green chemistry and process safety are paramount concerns for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
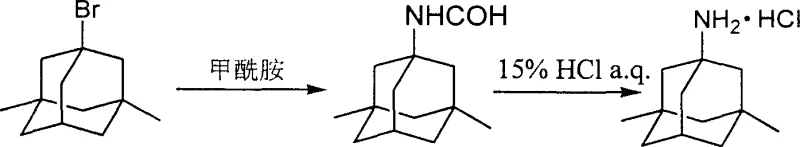
In stark contrast to the cumbersome legacy protocols, the innovative method described in CN100345819C offers a transformative solution by leveraging a direct nucleophilic substitution between a substituted halo-adamantane and a carboxamide, typically formamide. This novel pathway eliminates the need for corrosive sulfuric acid entirely, replacing it with a thermally driven reaction that can proceed at temperatures ranging from 70°C to 250°C, either neat or in appropriate organic solvents. The subsequent deprotection step utilizes mild mineral or organic acids in aqueous or hydrophilic media to cleave the formyl group, yielding the target amine salt with exceptional efficiency. This strategic shift not only simplifies the operational workflow by removing hazardous unit operations but also significantly reduces the generation of toxic waste streams, aligning perfectly with modern sustainability goals. The versatility of this approach allows for the direct use of crude intermediates in the second step or simple recrystallization, thereby accelerating the production timeline and enhancing the throughput capacity for reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Substitution and Acidic Hydrolysis
From a mechanistic perspective, the success of this synthesis relies on the unique reactivity of the bridgehead carbon in the adamantane cage system, which undergoes nucleophilic displacement under thermal activation. Although bridgehead positions are typically sterically hindered, the rigid cage structure of the diamantane framework facilitates the formation of a stabilized carbocation intermediate or allows for direct SN2-like displacement when driven by high thermal energy and the nucleophilicity of the amide nitrogen. The initial reaction between the bromo-diamantane and formamide proceeds through the attack of the amide nitrogen on the electrophilic carbon center, displacing the bromide ion and forming a stable formamido linkage. This step is critical as it installs the nitrogen functionality without the need for harsh acidic catalysts, preserving the integrity of the carbon skeleton and minimizing skeletal rearrangements that often plague acid-catalyzed processes. The careful control of temperature ensures that the reaction kinetics favor the desired substitution over elimination pathways, resulting in a clean conversion to the formyl-protected intermediate.
The second phase of the mechanism involves the acidic hydrolysis of the formamide group, a transformation that is kinetically more favorable than the hydrolysis of acetamides used in older methods. In the presence of aqueous acid, the carbonyl oxygen of the formamide is protonated, increasing the electrophilicity of the carbonyl carbon and making it susceptible to nucleophilic attack by water molecules. This leads to the formation of a tetrahedral intermediate which subsequently collapses to release formic acid and the free amine, which is immediately captured as its hydrochloride salt in the reaction medium. This mechanism is particularly advantageous because formamides are generally more labile towards hydrolysis than higher homologs, allowing the reaction to proceed under milder conditions and shorter timeframes. Consequently, the potential for thermal degradation of the sensitive adamantane core is minimized, ensuring that the final product exhibits a superior impurity profile with fewer degradation by-products, a key metric for high-purity OLED material and pharma grade chemicals alike.
How to Synthesize 3,5-Dimethyl-1-formamido-diamantane Efficiently
The practical execution of this synthesis involves a straightforward protocol that begins with the charging of formamide and the halo-adamantane precursor into a reactor equipped with efficient stirring and heating capabilities. The mixture is heated to the optimal temperature range, typically around 140°C, and maintained for several hours to ensure complete conversion, after which the reaction mass is quenched with water and extracted with a chlorinated solvent like chloroform. The organic layer is then washed to remove residual acids and salts, dried over anhydrous magnesium sulfate, and concentrated to yield the crude formamido intermediate, which can often be carried forward without further purification.
- Mix substituted halo-adamantane with formamide or substituted carboxamide, optionally using a solvent, and heat to 70-250°C for nucleophilic substitution.
- Extract the reaction mixture with an organic solvent, wash, dry, and concentrate to obtain the crude formamido-adamantane intermediate.
- Hydrolyze the intermediate using mineral or organic acid in water or a hydrophilic solvent to yield the final amantadine compound salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for adamantane-based therapeutics. By eliminating the dependency on concentrated sulfuric acid and glycol ethers, manufacturers can significantly reduce the expenditure associated with hazardous waste disposal and solvent recovery systems, leading to substantial operational cost savings. The simplified workup procedure, which often bypasses the need for complex distillation or chromatographic purification of intermediates, translates directly into reduced cycle times and lower labor costs per kilogram of produced API. Furthermore, the use of commodity chemicals like formamide and standard mineral acids ensures that the raw material supply is robust and less susceptible to market volatility compared to specialized reagents required by older methodologies. This resilience in the supply chain is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of sulfuric acid and expensive glycol ether solvents removes significant line items from the production budget, specifically regarding waste treatment and solvent procurement. The process operates with higher atom economy and reduced energy consumption due to shorter reaction times and milder hydrolysis conditions, driving down the overall cost of goods sold. Additionally, the higher yield obtained through this method means that less raw material is wasted, maximizing the output from every batch and improving the return on investment for manufacturing assets.
- Enhanced Supply Chain Reliability: Sourcing formamide and simple mineral acids is far more reliable than procuring specialized catalysts or handling regulated hazardous materials that face strict transportation restrictions. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities without requiring exotic corrosion-resistant equipment, thereby expanding the pool of qualified contract manufacturing organizations. This flexibility ensures that supply disruptions are minimized and that production can be scaled up rapidly to meet surges in market demand for antiviral or neuroprotective medications.
- Scalability and Environmental Compliance: The process generates significantly less acidic wastewater and organic solvent waste, simplifying compliance with increasingly stringent environmental regulations such as REACH and local EPA guidelines. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream facilitates easier permitting for new production lines and reduces the risk of regulatory shutdowns. This environmental stewardship not only protects the company's reputation but also future-proofs the manufacturing asset against tightening global sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational benefits and feasibility of adopting this route for large-scale production.
Q: What are the primary advantages of this new synthesis method over the traditional Ritter reaction?
A: The new method eliminates the need for large quantities of concentrated sulfuric acid, thereby reducing environmental pollution and safety hazards associated with hazardous waste disposal. It also simplifies the operational steps by avoiding complex deacetylation processes in glycol ethers, leading to a more streamlined production workflow.
Q: How does this process impact the purity and yield of Memantine HCl?
A: By utilizing a direct nucleophilic substitution followed by controlled acidic hydrolysis, the process minimizes the formation of side products common in high-temperature acid treatments. Experimental data indicates a substantial improvement in yield compared to previous methods that suffered from low conversion rates during the deprotection phase.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for industrial suitability by using readily available reagents like formamide and avoiding expensive or difficult-to-handle solvents. The simplified workup procedure, which often allows for direct use of crude intermediates or simple recrystallization, significantly enhances scalability and reduces overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Memantine HCl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying efficient and sustainable synthetic routes to meet the growing global demand for neuroprotective agents like Memantine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for downstream API synthesis, providing our partners with absolute confidence in product quality.
We invite pharmaceutical companies and chemical distributors to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your competitive advantage in the marketplace.
