Advanced Rhodium-Catalyzed Hydroformylation for High-Purity 5-Formylvalerate Esters Production
The chemical industry continuously seeks more efficient pathways for producing valuable aldehyde intermediates, particularly those serving as precursors for nylon and pharmaceutical applications. Patent CN1065522C introduces a significant breakthrough in this domain by detailing a highly selective process for preparing 5-formylvaleric acid and its corresponding esters. This innovation centers on a sophisticated hydroformylation reaction where 3-pentenoic acid or 3-pentenoate is treated with carbon monoxide and hydrogen in the presence of a specialized catalyst system. The core of this technological advancement lies in the utilization of a specific phosphite ligand structure, which forms a stable chelate complex with rhodium. Unlike traditional methods that struggle with isomer distribution and catalyst stability, this patented approach leverages a unique molecular architecture to drive the reaction overwhelmingly towards the desired linear 5-formyl product. By optimizing the coordination environment around the rhodium center, the process achieves superior regioselectivity, effectively suppressing the formation of branched isomers that complicate downstream purification. This development represents a critical step forward for manufacturers seeking reliable pharmaceutical intermediates supplier partnerships that can deliver high-purity materials with consistent quality.
The pursuit of efficiency in fine chemical manufacturing often hinges on overcoming the limitations inherent in legacy catalytic systems. Conventional methods, such as those disclosed in earlier patents like EP-A-556681, have historically faced challenges regarding the selectivity of 5-formylvalerate ester production. These older processes typically yield a significant proportion of unwanted isomers, specifically 3- and 4-formylvalerate esters, which possess boiling points very similar to the target product. This similarity makes fractional distillation energy-intensive and technically demanding, often resulting in lower overall yields and higher operational expenditures. Furthermore, the ligands employed in these prior art methods are frequently complex to synthesize, requiring multi-step procedures involving scarce substituted bis-phenols. The difficulty in preparing these ligands not only inflates the cost of the catalyst system but also introduces supply chain vulnerabilities due to the reliance on specialized starting materials. Consequently, manufacturers utilizing these conventional routes often face bottlenecks in scaling up production while maintaining economic viability.
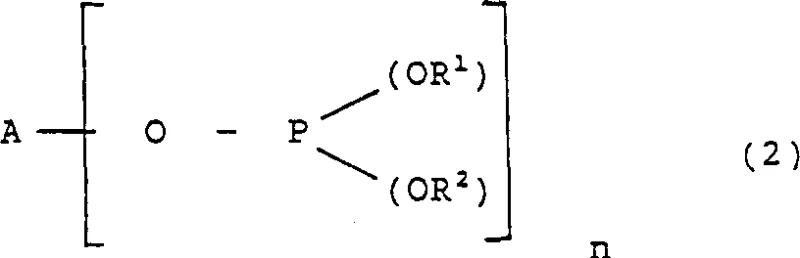
In stark contrast, the novel approach detailed in the patent data offers a transformative solution to these persistent industrial pain points. The new methodology employs a bidentate phosphite ligand represented by Formula (2), which is structurally distinct from its predecessors. A key advantage of this new ligand class is its ease of preparation; it can often be synthesized in fewer steps, sometimes even in a single vessel without isolating intermediate products. This simplification drastically reduces the complexity of catalyst manufacturing. Moreover, the ligand does not rely on hard-to-source substituted bis-phenols, instead utilizing readily available hydroxyl compounds. From a performance standpoint, the new catalyst system demonstrates markedly improved selectivity. Experimental data indicates that the ratio of the desired 5-formylvalerate ester to the undesired 3- and 4-isomers is significantly enhanced compared to prior art. This improvement translates directly into simplified downstream processing, as the reduced burden of isomer separation allows for more efficient fractionation. For procurement teams focused on cost reduction in fine chemical manufacturing, this shift represents a tangible opportunity to lower production costs through both raw material savings and reduced energy consumption during purification.
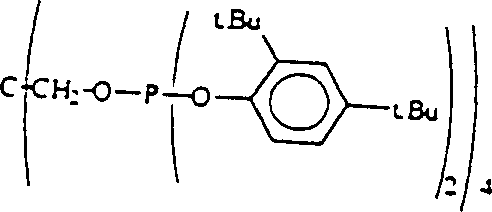
Understanding the mechanistic underpinnings of this hydroformylation process is crucial for R&D directors evaluating its feasibility for integration into existing production lines. The heart of the system is the formation of a chelating type complex between the phosphite ligand and the rhodium atom. In this configuration, the ligand molecule, which contains at least two trivalent phosphorus atoms, coordinates with a single rhodium center. This chelation effect creates a rigid and well-defined coordination sphere that profoundly influences the reaction pathway. The specific geometry imposed by the chelating ligand favors the insertion of the olefin substrate in a manner that leads to the linear aldehyde product. Additionally, the ligand structure features aromatic organic groups, preferably with ortho-substitution, such as tert-butyl groups. These bulky substituents provide significant steric hindrance around the phosphorus atoms, which serves to stabilize the trivalent phosphorus against oxidation and hydrolysis. This stabilization is vital for maintaining catalyst activity over extended reaction periods and under the elevated temperatures required for industrial throughput.
Furthermore, the electronic and steric properties of the ligand play a pivotal role in impurity control. The presence of ortho-substituents not only stabilizes the catalyst but also fine-tunes the electronic density at the rhodium center, optimizing the rate of the hydroformylation cycle relative to competing side reactions like isomerization or hydrogenation. In comparative experiments, non-chelating phosphite ligands or those lacking the specific structural features of Formula (2) showed significantly lower selectivity for the 5-formyl product, often yielding high amounts of methyl valerate or branched aldehydes. The patented system effectively suppresses these pathways, ensuring that the impurity profile of the crude reaction mixture is much cleaner. This high level of control over the reaction outcome minimizes the formation of heavy ends and polymeric by-products, which can foul reactors and complicate waste treatment. For technical teams, this means a more predictable and robust process that requires less intervention and monitoring, thereby freeing up resources for other critical R&D initiatives.
How to Synthesize 5-Formylvalerate Esters Efficiently
The synthesis of 5-formylvalerate esters using this advanced catalytic system is designed to be operationally straightforward while delivering high performance. The process begins with the preparation of the catalyst solution, where a rhodium precursor, such as Rh(CO)2(acac) or Rh(OAc)2, is mixed with the bidentate phosphite ligand in a molar excess relative to the rhodium. Typical ligand-to-rhodium ratios range from 1:1 to 100:1, with preferred embodiments utilizing ratios around 5:1 to ensure full complexation. The reaction is then conducted in a suitable solvent, such as toluene or the substrate itself, under a pressurized atmosphere of synthesis gas. The temperature is carefully controlled, generally between 50°C and 120°C, to balance reaction rate with catalyst stability. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst system by mixing a rhodium precursor, such as Rh(CO)2(acac), with the specific bidentate phosphite ligand defined in Formula (2) in a suitable solvent like toluene.
- Introduce the substrate, 3-pentenoic acid or its ester, into the reactor containing the catalyst mixture under an inert atmosphere.
- Pressurize the reactor with a synthesis gas mixture of carbon monoxide and hydrogen (H2: CO ratio between 1:2 to 5:1) and maintain temperatures between 50°C and 120°C to drive the hydroformylation reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling strategic advantages that extend beyond mere technical performance. The primary benefit lies in the substantial cost savings achievable through the simplification of the catalyst supply chain. Because the novel phosphite ligands are easier to synthesize and rely on commodity chemicals rather than exotic intermediates, the overall cost of the catalyst system is significantly reduced. This reduction in input costs directly improves the margin profile of the final 5-formylvalerate product. Additionally, the enhanced selectivity of the process means that less raw material is wasted on producing off-spec isomers. This efficiency gain reduces the volume of waste streams that require treatment or disposal, aligning with increasingly stringent environmental regulations and sustainability goals. The ability to source key components more reliably also mitigates the risk of supply disruptions, ensuring a steady flow of production.
- Cost Reduction in Manufacturing: The streamlined synthesis of the ligand eliminates several expensive and time-consuming steps associated with traditional ligand preparation. By avoiding the need for complex substituted bis-phenols, manufacturers can leverage cheaper, more abundant starting materials. Furthermore, the high selectivity of the reaction reduces the load on downstream purification units. Since less energy is required to separate the desired 5-formylvalerate from close-boiling isomers, utility costs for distillation columns are drastically lowered. This cumulative effect results in a leaner, more cost-effective manufacturing process that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials for both the substrate (3-pentenoates derived from butadiene) and the ligand components ensures a robust supply chain. Unlike processes dependent on niche specialty chemicals, this method utilizes building blocks that are produced at scale by multiple suppliers worldwide. This diversification of supply sources reduces vulnerability to geopolitical shifts or single-supplier failures. Moreover, the stability of the catalyst system allows for longer campaign runs without the need for frequent catalyst replenishment, further smoothing out production schedules and improving delivery reliability to customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in batch reactors with conditions amenable to continuous operation. The high thermal stability of the chelating ligand allows the product to be distilled off while the catalyst remains in the pot, facilitating catalyst recycling and minimizing heavy metal waste. This feature is critical for meeting modern environmental standards regarding heavy metal discharge. The reduction in by-product formation also simplifies waste management, lowering the environmental footprint of the facility and reducing the regulatory burden associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroformylation technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these details is essential for making informed decisions about process integration and long-term sourcing strategies.
Q: How does the novel phosphite ligand improve selectivity compared to prior art?
A: The novel ligand forms a stable chelating complex with rhodium, featuring ortho-substituted aromatic groups that provide steric bulk. This configuration significantly enhances regioselectivity towards the linear 5-formylvalerate ester, minimizing the formation of unwanted branched isomers like 3- and 4-formylvalerate esters.
Q: What are the advantages of the ligand synthesis method described in the patent?
A: The synthesis of the bidentate phosphite ligand is streamlined, often requiring fewer steps than conventional methods. It avoids the need for difficult-to-obtain substituted bis-phenols, allowing for preparation from readily available hydroxyl compounds and phosphorus halides, which simplifies the supply chain and reduces raw material costs.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The catalyst exhibits high thermal stability, allowing the product to be separated via simple distillation while the high-molecular-weight catalyst remains in the liquid phase for potential recycling. This robustness supports continuous processing and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Formylvalerate Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of advanced polymers and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 5-formylvalerate ester meets the exacting standards required by our global clientele. Our expertise in rhodium-catalyzed processes allows us to optimize reaction parameters for maximum yield and minimal impurity generation, delivering a product that facilitates efficient downstream synthesis for our partners.
We invite you to collaborate with us to unlock the full potential of this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your supply chain objectives. Together, we can drive innovation and efficiency in the production of essential chemical intermediates.
