Advanced Manufacturing of Neratinib Impurity D: A Robust Route for Pharmaceutical Quality Control
The pharmaceutical industry's relentless pursuit of drug safety has placed impurity profiling at the forefront of regulatory compliance, particularly for potent tyrosine kinase inhibitors like Neratinib. As detailed in patent CN111943933B, a significant technological breakthrough has been achieved in the preparation of Neratinib Impurity D, a critical reference standard required for validating the quality and safety of the final active pharmaceutical ingredient. This novel methodology addresses longstanding challenges in impurity synthesis by employing a streamlined four-step reaction sequence starting from N,N-dimethylamino trans-crotonic acid. By leveraging specific catalytic conditions involving zinc nitrate, this process not only enhances reaction efficiency but also ensures the structural integrity of sensitive functional groups that are prone to degradation in conventional synthetic routes. For R&D directors and quality control managers, access to such high-fidelity impurity standards is indispensable for meeting stringent ICH guidelines regarding the identification and quantification of trace organic impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinoline-based impurities has been plagued by inefficient reaction pathways that compromise both yield and purity. Prior art, such as the methods disclosed in WO2009052264A, often relied on direct Michael addition reactions which, while conceptually straightforward, resulted in a plethora of undesirable byproducts. These side reactions necessitated rigorous and costly purification protocols, typically involving column chromatography, which significantly reduces the overall mass recovery and increases the cost of goods sold. Furthermore, traditional acidic conditions frequently induced the hydrolysis of the critical nitrile (-CN) group on the quinoline ring and the cleavage of side chain amides, leading to structurally compromised materials that fail to serve as accurate analytical standards. The inability to consistently produce high-purity material without extensive downstream processing has long been a bottleneck for supply chains dependent on reliable reference substances.
The Novel Approach
The innovative strategy outlined in the provided patent data revolutionizes this landscape by introducing a robust, four-step synthetic route that prioritizes chemoselectivity and operational simplicity. By initiating the synthesis with N,N-dimethylamino trans-crotonic acid and progressing through carefully controlled acylation and Michael addition steps, the process builds the molecular complexity gradually and efficiently. The cornerstone of this advancement lies in the final coupling step, where the use of zinc nitrate as a specific catalyst in an acetonitrile solvent system creates an optimal environment for bond formation. This approach effectively suppresses the hydrolysis pathways that plague earlier methods, allowing for the isolation of the target Neratinib Impurity D with exceptional purity levels exceeding 99.0% simply through recrystallization. 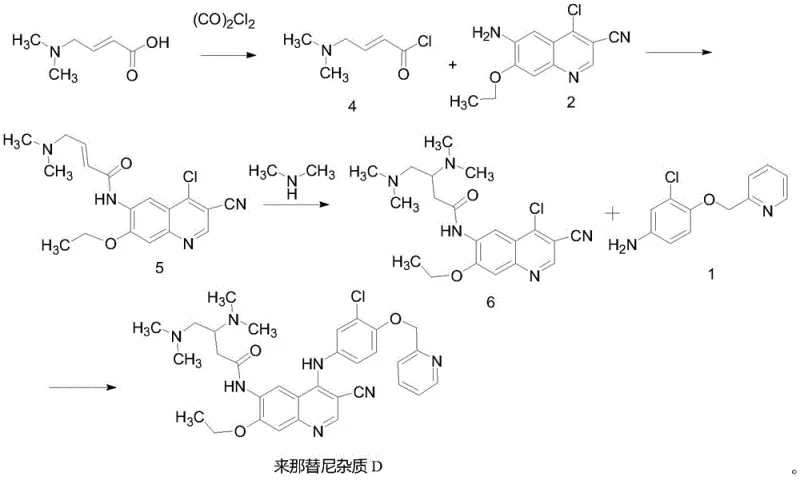
Mechanistic Insights into Zinc Nitrate Catalyzed Coupling
The mechanistic superiority of this synthesis is most evident in the final transformation, where the choice of catalyst dictates the success of the reaction. In conventional acid-catalyzed couplings, strong protic acids often protonate sensitive nitrogen atoms or activate water molecules that attack the nitrile and amide functionalities, leading to degradation. In contrast, zinc nitrate functions as a Lewis acid that likely coordinates with the carbonyl oxygen or the leaving group, activating the electrophile for nucleophilic attack by the aniline derivative without generating a highly acidic medium. This subtle yet profound difference in reaction mechanism preserves the delicate balance of functional groups within the molecule, preventing the formation of carboxylic acid byproducts from nitrile hydrolysis. The result is a cleaner reaction profile that minimizes the formation of closely related impurities, thereby simplifying the purification burden and enhancing the overall reliability of the synthetic output.
Furthermore, the thermal stability of the intermediates under the specified reaction conditions contributes significantly to the high yield observed. The process operates at a moderate reflux temperature of 60-85°C, which provides sufficient kinetic energy to drive the coupling to completion while avoiding the thermal decomposition that can occur at higher temperatures. This thermal window is critical for maintaining the stereochemical and structural integrity of the dimethylamino side chain and the chloro-substituted phenyl ring. By optimizing the solvent system to acetonitrile, the protocol ensures excellent solubility of reactants while facilitating the precipitation of the product upon cooling, a phenomenon that aids in the physical separation of the target molecule from soluble impurities. 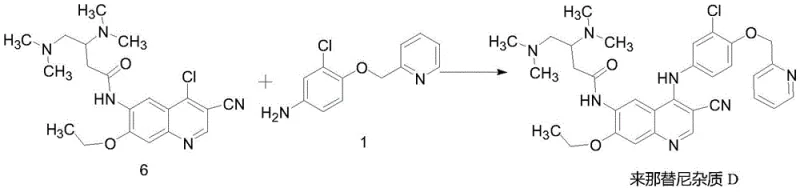
How to Synthesize Neratinib Impurity D Efficiently
The execution of this synthesis requires precise adherence to the established protocol to replicate the high yields and purity reported in the patent literature. The process begins with the activation of the carboxylic acid precursor using oxalyl chloride, followed by sequential coupling and amination steps that build the core scaffold. Each stage is designed to maximize conversion while minimizing workup complexity, culminating in a final catalytic step that locks in the molecular structure with high fidelity. Operators should note that the avoidance of aqueous workups in the early stages and the specific choice of recrystallization solvents are key to achieving the reported 99.0% purity without chromatographic intervention. Detailed standardized operating procedures for each reaction stage are essential for technology transfer and scale-up.
- React N,N-dimethylamino trans-crotonic acid with oxalyl chloride in anhydrous THF at low temperature to generate the acid chloride intermediate (Compound 4).
- Couple Compound 4 with the quinoline amine (Compound 2) in NMP solvent, followed by pH adjustment and recrystallization to isolate Compound 5.
- Perform a Michael addition reaction between Compound 5 and dimethylamine in THF at elevated temperatures (80-110°C) to form Compound 6.
- Execute the final coupling of Compound 6 with Compound 1 using zinc nitrate catalyst in acetonitrile at reflux to obtain Neratinib Impurity D.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this novel synthetic route offers transformative economic and logistical benefits that extend far beyond the laboratory bench. The elimination of column chromatography represents a massive reduction in processing time and consumable costs, as silica gel and eluent solvents are expensive and generate significant hazardous waste. By achieving high purity through simple recrystallization, manufacturers can drastically reduce the cycle time per batch, thereby increasing the throughput of existing production facilities without the need for capital-intensive equipment upgrades. This efficiency gain translates directly into a more resilient supply chain capable of responding rapidly to fluctuating market demands for high-quality pharmaceutical reference standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive purification techniques with cost-effective crystallization methods. Traditional methods relying on column chromatography consume vast quantities of solvents and stationary phases, driving up the variable cost per kilogram of product. By circumventing this step, the new method significantly lowers the direct material costs and reduces the environmental burden associated with solvent disposal. Additionally, the use of cheap and readily available starting materials like N,N-dimethylamino trans-crotonic acid ensures that raw material price volatility has a minimal impact on the final product cost, providing stable pricing structures for long-term contracts.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis; however, this streamlined four-step route mitigates risk by reducing the number of unit operations where failures can occur. The robustness of the zinc nitrate catalyzed step means that batch-to-batch variability is minimized, ensuring a consistent supply of material that meets strict quality specifications. Furthermore, the mild reaction conditions do not require specialized high-pressure or cryogenic equipment, allowing the synthesis to be performed in a wider range of manufacturing facilities, thus diversifying the potential supplier base and reducing dependency on single-source vendors.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns perfectly with green chemistry principles by reducing waste generation and energy consumption. The absence of complex purification steps reduces the volume of organic waste streams, simplifying compliance with increasingly stringent environmental regulations. The reaction conditions are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production without the need for re-optimization. This scalability ensures that as the demand for Neratinib and its associated quality control materials grows, the supply of Impurity D can be expanded proportionally to support global regulatory submissions and ongoing quality assurance programs.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this synthesis method. Understanding the nuances of the catalytic system and purification strategy is vital for laboratories aiming to adopt this protocol for in-house reference standard production or for qualifying external suppliers. The answers provided are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is Zinc Nitrate preferred over traditional acid catalysts for this synthesis?
A: Traditional strong acid catalysts like methanesulfonic acid often lead to unwanted hydrolysis of the quinoline ring nitrile group (-CN) and side chain amides. Zinc nitrate acts as a milder Lewis acid that facilitates the coupling reaction while preserving these sensitive functional groups, ensuring higher product integrity.
Q: What purity levels can be achieved with this new method?
A: The optimized protocol allows for the production of Neratinib Impurity D with a purity exceeding 99.0% directly after recrystallization. This eliminates the need for complex and costly column chromatography purification steps typically required in older methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and readily available raw materials and operates under mild reaction conditions (60-85°C) without requiring specialized large-scale equipment. The high yield and simplified purification make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Neratinib Impurity D Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your drug development pipeline depends on the quality of the reference materials you utilize. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for early-stage research or late-stage clinical validation. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Neratinib Impurity D we supply matches the structural and purity profiles defined by international pharmacopeial standards.
We invite you to collaborate with us to optimize your supply chain for critical pharmaceutical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your commitment to drug safety and quality excellence.
