Advanced Synthesis of Alkyl 6-Aminocaproate for High-Purity Caprolactam Manufacturing
The global demand for high-performance polyamides and specialized amino acid derivatives necessitates robust, scalable synthetic routes for key intermediates like alkyl 6-aminocaproate. Patent CN1264808C introduces a transformative methodology for producing alkyl 6-aminocaproate and its cyclized derivative, caprolactam, starting from 3-pentenenitrile (3PN). Unlike conventional industrial processes that rely on cyclohexane or generate substantial quantities of hexamethylenediamine (HMD) as an unavoidable co-product, this invention leverages a sophisticated sequence of hydroformylation, oxidative esterification, and selective hydrogenation. By stabilizing the reactive aldehyde intermediates through esterification prior to purification, the process overcomes historical thermal instability issues, enabling the isolation of linear isomers with exceptional purity. This technical breakthrough offers a compelling value proposition for manufacturers seeking to optimize yield and minimize byproduct management in the production of nylon-6 precursors and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of butadiene-derived 3-pentenenitrile into caprolactam precursors has been plagued by significant chemical and economic inefficiencies. Traditional pathways often involve converting 3PN to adiponitrile (ADN), followed by partial hydrogenation to 6-aminocapronitrile. A critical drawback of this ADN route is the concurrent formation of large volumes of hexamethylenediamine (HMD), a distinct chemical commodity that may not be required by the specific production facility, thereby creating inventory imbalances and necessitating complex separation protocols. Furthermore, alternative strategies attempting to utilize 5-formylvaleronitrile (5-FVN) directly have failed commercially because 5-FVN is thermally unstable. Attempts to separate the desired linear 5-FVN from its branched 3- and 4-isomers via distillation result in severe decomposition and yield losses, rendering the isolation of high-purity linear intermediates practically impossible using standard unit operations.
The Novel Approach
The methodology disclosed in CN1264808C circumvents these bottlenecks by introducing an oxidative esterification step that fundamentally alters the physical properties of the intermediate stream. Instead of attempting to distill unstable aldehydes, the process converts the mixture of 3-, 4-, and 5-formylvaleronitriles into their corresponding alkyl cyanovalerates. These esterified products possess significantly higher thermal stability and distinct boiling point differentials compared to their aldehyde precursors. Specifically, the linear alkyl 5-cyanovalerate exhibits a boiling point approximately 20°C higher than its branched isomers under vacuum conditions. This physical property enhancement allows for the use of conventional fractional distillation to effectively strip away branched impurities, yielding a refined linear feedstock that can be subsequently hydrogenated to alkyl 6-aminocaproate without the burden of HMD co-production or thermal degradation losses.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation and Oxidative Esterification
The initial stage of this synthesis involves the hydroformylation of 3-pentenenitrile using carbon monoxide and hydrogen in the presence of a Group VIII metal catalyst, with rhodium complexes being particularly preferred. The choice of ligand in this catalytic cycle is paramount for directing regioselectivity towards the desired linear aldehyde. The patent specifies the use of phosphorus-containing ligands, including multidentate phosphites, to modulate the electronic and steric environment around the rhodium center. These ligands facilitate the insertion of the olefin into the metal-hydride bond in a manner that favors the formation of the terminal aldehyde group necessary for the subsequent linear product. The precise tuning of the ligand structure, as exemplified by the general formula shown below, ensures high conversion rates while maintaining the integrity of the nitrile functionality under moderate temperatures ranging from 50°C to 150°C.
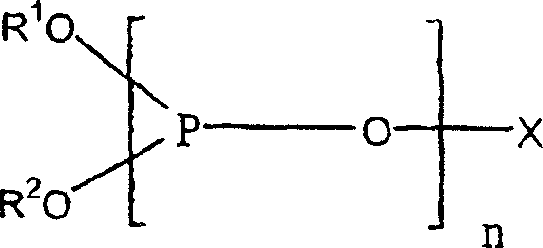
Following hydroformylation, the crude formylvaleronitrile (FVN) mixture undergoes oxidative esterification, a dual-function reaction that oxidizes the aldehyde moiety to a carboxylic acid derivative while simultaneously esterifying it with an alcohol such as methanol. This transformation is catalyzed by heterogeneous palladium-based systems, often promoted with elements like tellurium, zinc, or lead. The mechanism likely involves the activation of molecular oxygen on the palladium surface to generate a peroxy-species that attacks the carbonyl carbon, followed by nucleophilic attack by the alcohol. This step is exothermic and operates efficiently at temperatures between 40°C and 80°C. The resulting alkyl cyanovalerates are chemically robust, allowing them to withstand the thermal stress of downstream distillation columns where the critical separation of linear versus branched isomers occurs based on vapor pressure differences.
How to Synthesize Alkyl 6-Aminocaproate Efficiently
The synthesis of alkyl 6-aminocaproate via this patented route requires precise control over reaction parameters to maximize linearity and minimize side reactions. The process begins with the rigorous exclusion of air during the hydroformylation step to prevent catalyst deactivation, followed by a controlled introduction of oxygen during the oxidative esterification phase. Once the linear alkyl 5-cyanovalerate is isolated with purity exceeding 99.5%, it is subjected to hydrogenation using sponge metal catalysts. The detailed operational parameters, including specific catalyst loadings, pressure gradients, and temperature profiles required to achieve commercial-grade consistency, are outlined in the standardized synthesis guide below.
- Hydroformylate 3-pentenenitrile using a Group VIII metal catalyst (e.g., Rhodium) to generate a mixture of formylvaleronitriles (FVN).
- Perform oxidative esterification on the FVN mixture using a Palladium catalyst and alcohol to convert aldehydes into stable alkyl cyanovalerates.
- Isolate the linear alkyl 5-cyanovalerate via distillation and hydrogenate it using a Raney metal catalyst to yield alkyl 6-aminocaproate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this synthetic route offers profound logistical and economic advantages rooted in process intensification and waste reduction. By eliminating the formation of hexamethylenediamine, manufacturers remove the need to manage, store, or market a significant co-product, thereby streamlining the supply chain focus solely on caprolactam precursors. The ability to use standard distillation equipment for isomer separation, rather than requiring complex cryogenic or extractive separation technologies for unstable aldehydes, significantly lowers capital expenditure requirements for new plant installations. Furthermore, the use of heterogeneous catalysts in the oxidative esterification step facilitates easier catalyst recovery and recycling, reducing the consumption of precious metals like palladium over the lifecycle of the plant.
- Cost Reduction in Manufacturing: The elimination of HMD byproduct generation translates directly into reduced downstream processing costs, as there is no need for energy-intensive separation trains to isolate diamines from amino-nitriles. Additionally, the thermal stability of the alkyl cyanovalerate intermediates minimizes material loss during purification, leading to higher overall mass balance efficiency. The process operates at moderate pressures and temperatures compatible with standard stainless-steel reactor infrastructure, avoiding the need for exotic alloys required by highly corrosive alternative chemistries.
- Enhanced Supply Chain Reliability: The reliance on widely available feedstocks such as butadiene-derived 3-pentenenitrile and common alcohols like methanol ensures a resilient raw material supply base. The robustness of the oxidative esterification step against thermal degradation means that production campaigns can be run for extended durations without the risk of fouling reactors with decomposition polymers. This stability enhances batch-to-batch consistency, ensuring that downstream polymerization customers receive intermediates with predictable reactivity profiles.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by utilizing molecular oxygen as the oxidant, generating water as the primary byproduct of oxidation rather than stoichiometric amounts of heavy metal waste salts. The high selectivity of the hydrogenation step using Raney-type catalysts reduces the formation of secondary amines or cyclic byproducts, simplifying wastewater treatment requirements. The modular nature of the reaction steps allows for flexible capacity expansion, enabling manufacturers to scale from pilot units to multi-ton annual production with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this alkyl 6-aminocaproate synthesis pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on catalyst selection, impurity profiles, and operational safety. Understanding these nuances is essential for engineering teams evaluating the feasibility of integrating this technology into existing fine chemical manufacturing assets.
Q: Why is the oxidative esterification step critical in this process?
A: Direct isolation of 5-formylvaleronitrile (5-FVN) is impractical due to its thermal instability and difficulty in separating from branched isomers. Oxidative esterification converts these unstable aldehydes into thermally stable alkyl cyanovalerates, allowing for effective fractional distillation to isolate the linear isomer with high purity.
Q: How does this method improve upon traditional caprolactam precursors?
A: Traditional routes converting 3-pentenenitrile to adiponitrile often produce significant amounts of hexamethylenediamine (HMD) as a byproduct during partial hydrogenation. This patented process bypasses HMD formation entirely by routing through alkyl 5-cyanovalerate, simplifying downstream purification and reducing waste handling costs.
Q: What catalysts are preferred for the hydrogenation step?
A: The patent highlights sponge metal catalysts, specifically Raney Nickel or Raney Cobalt, as highly effective for hydrogenating the nitrile group to an amine while maintaining the ester functionality. Supported Ruthenium catalysts are also noted as suitable alternatives for high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl 6-Aminocaproate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of supply chain continuity and chemical purity in the production of high-value polymer precursors and pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent data to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze trace isomeric impurities, guaranteeing that every batch of alkyl 6-aminocaproate meets the exacting standards required for nylon-6 polymerization or active pharmaceutical ingredient synthesis.
We invite global partners to collaborate with us on optimizing this efficient synthetic route for their specific market needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and reduce total landed costs.
