Advanced Vinyl Sulfonic Anhydride Production for Next-Gen Fuel Cell Membranes
Advanced Vinyl Sulfonic Anhydride Production for Next-Gen Fuel Cell Membranes
The development of high-performance fluorine-based polymer electrolytes is critical for the advancement of fuel cell technology, particularly for applications requiring robust separators and catalyst binder polymers. Patent CN111954662B introduces a groundbreaking methodology centered on the synthesis and utilization of vinyl sulfonic anhydride as a pivotal intermediate. This innovation addresses long-standing challenges in producing short-chain fluorinated monomers, which are essential for achieving higher glass transition temperatures and superior mechanical strength in final polymer products. By establishing a novel pathway that bypasses traditional limitations associated with cyclization side reactions and hazardous phosphorus reagents, this technology offers a streamlined route to high-purity vinyl sulfonyl fluoride. The strategic implementation of this anhydride-mediated process represents a significant leap forward for manufacturers seeking reliable fluorine material suppliers capable of delivering complex electronic chemical intermediates at scale.
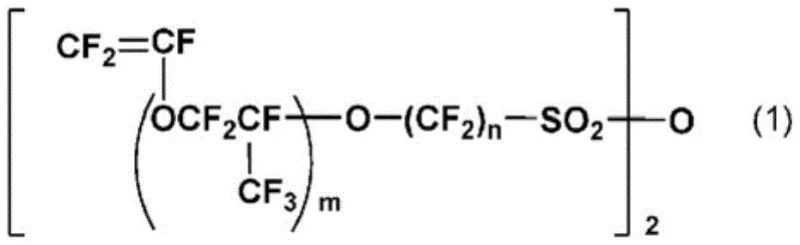
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated monomers with short spacers between the main chain and the sulfonic acid group has been plagued by significant chemical hurdles. When attempting to synthesize monomers where the spacer length parameter p equals zero, conventional decarboxylation-vinylation reactions predominantly favor intramolecular cyclization rather than the desired linear elimination. As illustrated in the prior art reaction schemes, this tendency results in the formation of stable five-membered cyclic compounds instead of the target vinyl sulfonyl fluoride, drastically reducing the yield of the useful monomer. Furthermore, alternative methods disclosed in earlier patents rely heavily on the use of phosphorus pentachloride to convert sulfonic acid salts into sulfonyl chlorides. This approach inevitably generates phosphorus oxychloride as a stoichiometric byproduct, which is highly reactive and complicates the subsequent fluorination step by limiting solvent choices and reaction conditions. The necessity to separate phosphorus oxychloride from the reaction mixture adds layers of operational complexity and cost, rendering these traditional methods less suitable for efficient industrial production.
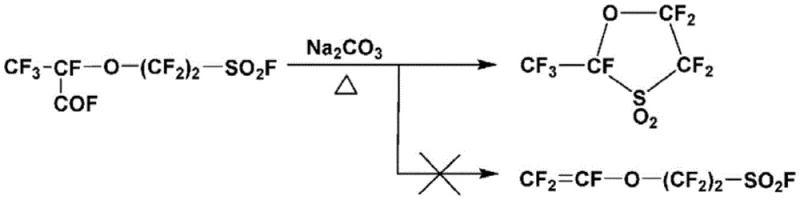
The Novel Approach
In stark contrast to these cumbersome legacy processes, the present invention leverages the unique reactivity of vinyl sulfonic anhydride to facilitate a cleaner and more efficient transformation. By first converting the vinyl sulfonic acid compound into its corresponding anhydride form using dehydrating agents such as phosphorus pentoxide or thionyl chloride, the process creates a highly activated intermediate that is primed for nucleophilic attack. This anhydride intermediate serves as a superior precursor because it reacts smoothly with fluorinating agents to yield the desired vinyl sulfonyl fluoride without generating the problematic phosphorus oxychloride byproduct. Moreover, the reaction system is designed to co-produce a vinyl sulfonic acid compound alongside the target monomer, which can be easily separated and recycled back into the process. This closed-loop strategy not only mitigates the issue of low yields caused by cyclization but also eliminates the need for hazardous chlorine-containing reagents, thereby simplifying the purification workflow and enhancing the overall safety profile of the manufacturing operation.
Mechanistic Insights into Anhydride-Mediated Fluorination
The core of this technological breakthrough lies in the precise control of the dehydration and subsequent fluorination steps, which together form a robust catalytic-like cycle for monomer production. In the initial stage, the vinyl sulfonic acid compound undergoes a condensation reaction with an acid anhydride-forming agent, effectively removing water to form the symmetric vinyl sulfonic anhydride. This transformation is critical because the anhydride linkage possesses a higher electrophilicity at the sulfur center compared to the parent acid or its salts, making it exceptionally susceptible to nucleophilic substitution by fluoride ions. When a metal fluoride such as sodium fluoride or potassium fluoride is introduced, the fluoride ion attacks the sulfur atom of the anhydride, cleaving the S-O-S bond to release one molecule of vinyl sulfonyl fluoride and one molecule of the corresponding vinyl sulfonic acid salt. This mechanism ensures that the fluorination proceeds under milder conditions than those required for sulfonyl chlorides, reducing the thermal stress on the sensitive vinyl ether moiety and preserving the integrity of the double bond.
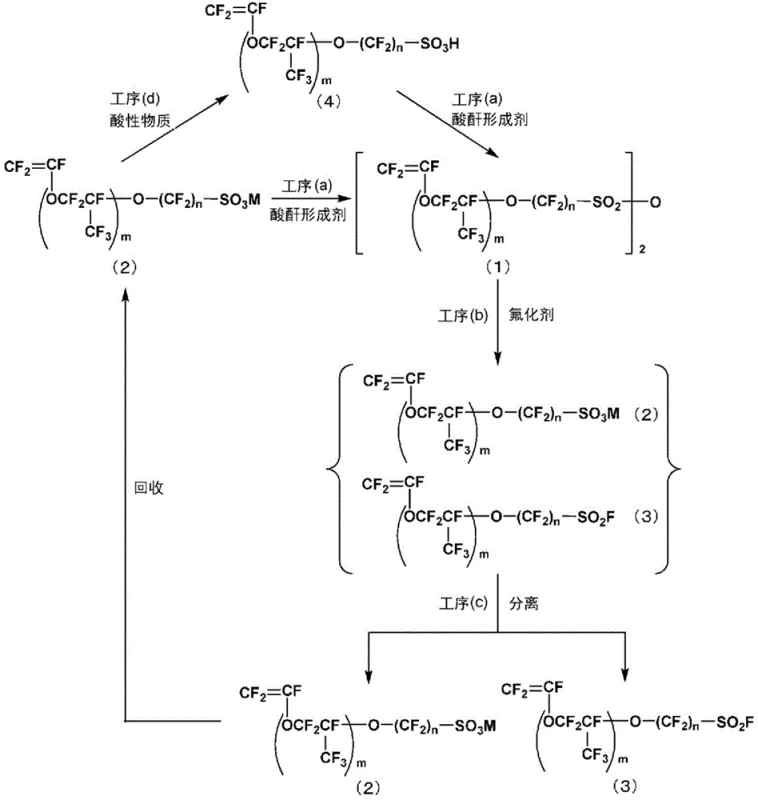
Furthermore, the mechanistic design inherently supports impurity control through the physical separation of reaction components based on their volatility and solubility profiles. Since the vinyl sulfonyl fluoride product typically has a different boiling point than the vinyl sulfonic acid salt byproduct, they can be efficiently separated via distillation or extraction, ensuring a high-purity output suitable for polymerization. The recovered acid salt is not discarded; instead, it is subjected to an acidification step using strong acids or ion-exchange resins to regenerate the free vinyl sulfonic acid. This regenerated acid is then fed back into the dehydration step, creating a continuous cycle that maximizes atom economy. By avoiding the accumulation of phosphorus-containing impurities and preventing the formation of cyclic byproducts through the use of the pre-activated anhydride, the process maintains a clean impurity profile throughout multiple cycles, which is essential for meeting the stringent quality standards required for fuel cell membrane applications.
How to Synthesize Vinyl Sulfonic Anhydride Efficiently
The practical implementation of this synthesis route involves a series of carefully controlled unit operations that transform readily available starting materials into high-value fluorinated monomers. The process begins with the preparation of the vinyl sulfonic acid precursor, which may be derived from fluorinated epoxides or other standard fluorine chemistry feedstocks. Once the acid is obtained, it is mixed with a dehydrating agent under inert atmosphere conditions to generate the anhydride intermediate, followed by the addition of a fluoride source to effect the final conversion. The detailed standardized synthesis steps, including specific temperature ramps, pressure controls, and molar ratios optimized for maximum yield, are outlined in the comprehensive guide below for technical teams evaluating process feasibility.
- Contact a vinyl sulfonic acid compound with an acid anhydride-forming agent such as phosphorus pentoxide to generate vinyl sulfonic anhydride.
- React the resulting vinyl sulfonic anhydride with a fluorinating agent like sodium fluoride to produce vinyl sulfonyl fluoride and a vinyl sulfonic acid salt byproduct.
- Separate the desired fluorinated monomer and convert the recovered acid salt back into vinyl sulfonic acid using an acidic substance for reuse in the initial step.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthetic route offers substantial advantages that directly address the cost and reliability concerns of procurement managers and supply chain directors in the specialty chemicals sector. By eliminating the reliance on phosphorus pentachloride, the process removes the need for expensive and complex downstream purification systems designed to handle corrosive phosphorus oxychloride waste. This simplification of the purification train translates into significantly reduced capital expenditure for reactor lining and waste treatment infrastructure, while also lowering the operational costs associated with hazardous waste disposal. Additionally, the ability to recycle the vinyl sulfonic acid byproduct back into the main production loop means that raw material consumption is drastically minimized, leading to a more sustainable and cost-effective manufacturing model that insulates the supply chain from fluctuations in precursor pricing.
- Cost Reduction in Manufacturing: The elimination of phosphorus pentachloride and the associated byproduct phosphorus oxychloride removes a major cost driver related to reagent consumption and waste management. Without the need for specialized separation units to remove phosphorus contaminants, the overall processing time is shortened, and energy consumption is lowered, resulting in a leaner production cost structure. Furthermore, the recycling of the acid byproduct ensures that nearly all input material is converted into valuable product or reused, maximizing the return on investment for every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: The use of common and stable reagents such as phosphorus pentoxide and alkali metal fluorides ensures a robust supply chain that is less vulnerable to the shortages often associated with specialized chlorinating agents. The modular nature of the process, which allows for the intermediate anhydride to be stored or transported if necessary, provides flexibility in production scheduling and inventory management. This flexibility enables manufacturers to respond more agilely to market demand spikes for fuel cell materials without compromising on lead times or product availability.
- Scalability and Environmental Compliance: The process operates under conditions that are amenable to large-scale batch or continuous flow reactors, facilitating the commercial scale-up of complex fluorinated monomers from pilot quantities to multi-ton annual production. The absence of chlorine gas evolution and the reduction of phosphorus waste align with increasingly stringent environmental regulations, reducing the regulatory burden and permitting risks for manufacturing facilities. This environmental compatibility ensures long-term operational continuity and enhances the corporate sustainability profile for companies adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this vinyl sulfonic anhydride technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is key to appreciating how this method overcomes the specific bottlenecks of previous generations of fluorine chemistry.
Q: Why is synthesizing short-chain fluorinated monomers (p=0) traditionally difficult?
A: Conventional decarboxylation-vinylation reactions for short-chain precursors often favor intramolecular cyclization over the desired linear monomer formation, leading to extremely low yields of the target fluorinated monomer.
Q: What is the primary disadvantage of using phosphorus pentachloride in prior art methods?
A: The use of phosphorus pentachloride generates phosphorus oxychloride as a reactive byproduct, which can inhibit the subsequent fluorination reaction and necessitates complex and costly separation procedures to ensure product purity.
Q: How does the new anhydride method improve process sustainability?
A: The process allows for the recovery and recycling of the vinyl sulfonic acid byproduct generated during fluorination, converting it back into the anhydride intermediate, thereby minimizing waste and maximizing atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Sulfonic Anhydride Supplier
As the global demand for high-efficiency fuel cells and advanced fluoropolymers continues to surge, securing a supply of high-purity intermediates is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of vinyl sulfonic anhydride or vinyl sulfonyl fluoride meets the exacting standards required for electronic grade applications. We understand the critical nature of these materials in the value chain and are committed to providing a seamless supply experience.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this anhydride-mediated process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a successful and profitable partnership in the evolving landscape of fluorine materials.
