Advanced Iron-Catalyzed Synthesis of 1-[60] Fullerene Pyrrolidine Derivatives for Electronic Materials
Advanced Iron-Catalyzed Synthesis of 1-[60] Fullerene Pyrrolidine Derivatives for Electronic Materials
The rapid evolution of organic electronics and photovoltaic technologies has intensified the demand for high-performance n-type semiconductor materials, with fullerene C60 derivatives standing at the forefront of this innovation. Patent CN112079765B discloses a groundbreaking synthesis method for 1-[60] fullerene pyrrolidine derivatives, addressing critical challenges in functionalizing the carbon cage. This technology leverages a transition metal iron salt catalyst to facilitate the reaction between fullerene C60 and gamma,delta-unsaturated oxime ester compounds under neutral redox conditions. The result is the efficient construction of rare structures where nitrogen atoms are directly linked to the carbon cage, offering superior solubility and tunable electronic properties. For R&D directors and procurement specialists in the electronic materials sector, this represents a significant leap forward in accessing reliable fullerene derivative suppliers capable of delivering complex intermediates with high purity and consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of fullerene C60 to create pyrrolidine derivatives has been plagued by significant synthetic hurdles that impede commercial viability. Traditional protocols often rely on expensive precious metal catalysts such as rhodium or palladium, which not only inflate the cost of goods sold but also introduce severe contamination risks that require rigorous and costly purification steps to meet electronic grade standards. Furthermore, many existing methods operate under harsh oxidative or reductive conditions that can degrade the delicate pi-electron system of the fullerene cage, leading to poly-addition byproducts and reduced yields. These inefficiencies create bottlenecks in the supply chain for high-purity OLED material and semiconductor precursors, forcing manufacturers to contend with long lead times and inconsistent batch quality. The reliance on multi-step sequences with low atom economy further exacerbates waste generation, conflicting with modern environmental compliance standards required by global supply chain heads.
The Novel Approach
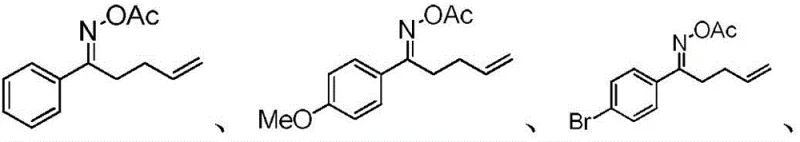
In stark contrast, the novel approach detailed in the patent utilizes an earth-abundant transition metal iron salt, specifically FeCl2·4H2O, to drive the cyclization reaction with remarkable efficiency. This method operates under a neutral oxidation-reduction state, preserving the integrity of the fullerene cage while enabling the direct formation of the nitrogen-carbon bond. The reaction proceeds smoothly in a mixed solvent system of o-dichlorobenzene and acetonitrile at moderate temperatures ranging from 70°C to 120°C. This streamlined one-pot process eliminates the need for cryogenic conditions or hazardous reagents, drastically simplifying the operational workflow. By employing gamma,delta-unsaturated oxime esters as versatile building blocks, the methodology achieves high isolated yields, exemplified by the 44% yield obtained for the phenyl-substituted derivative 2a. This robustness makes it an ideal candidate for cost reduction in electronic chemical manufacturing, providing a scalable route to diverse molecular architectures.
![General reaction scheme for the synthesis of 1-[60] fullerene pyrrolidine derivatives using iron catalysis](/insights/img/synthesis-1-60-fullerene-pyrrolidine-iron-catalysis-supplier-20260307092710-01.webp)
Mechanistic Insights into FeCl2-Catalyzed Radical Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the iron catalyst, which initiates a radical cascade sequence essential for constructing the pyrrolidine ring on the fullerene surface. The mechanism begins with the coordination of the iron(II) species to the oxime ester moiety, promoting the homolytic cleavage of the weak N-O bond. This critical step generates an iminyl radical intermediate along with an acetate anion, setting the stage for a subsequent 5-exo-trig cyclization. The resulting carbon-centered radical is highly reactive and selectively attacks the electron-deficient double bonds of the C60 cage. This addition forms a new carbon-carbon bond and a fullerene-based radical, which eventually abstracts a hydrogen atom or undergoes further termination to yield the stable 1-[60] fullerene pyrrolidine product. Understanding this radical manifold is crucial for R&D teams aiming to optimize reaction parameters, as it explains the high regioselectivity and the tolerance for various functional groups on the oxime ester substrate.
From an impurity control perspective, the neutral redox environment plays a pivotal role in minimizing side reactions that typically plague fullerene chemistry. Unlike strong oxidants that might cause cage opening or excessive functionalization, the mild iron-catalyzed conditions favor mono-addition, thereby simplifying the downstream purification process. The use of silica gel chromatography with CS2/CH2Cl2 eluents effectively separates the target mono-adducts from unreacted C60 and minor byproducts. This level of control over the impurity profile is vital for applications in display and optoelectronic materials, where trace metallic or organic impurities can severely degrade device performance. The ability to recover and reuse unreacted fullerene C60 further enhances the economic feasibility of the process, ensuring that valuable starting materials are not wasted during the synthesis of complex polymer additives or specialty chemical intermediates.
How to Synthesize 1-[60] Fullerene Pyrrolidine Derivative Efficiently
Implementing this synthesis route requires careful attention to reaction stoichiometry and solvent quality to maximize yield and reproducibility. The patent outlines a standardized protocol where fullerene C60, the specific gamma,delta-unsaturated oxime ester, and the iron catalyst are combined in a precise molar ratio of 1:2:0.2. The reaction mixture is subjected to ultrasonic treatment to ensure complete dissolution before being heated in an oil bath under a nitrogen atmosphere. Following the reaction, a straightforward workup involving filtration through a short silica plug and subsequent column chromatography affords the pure product. For detailed operational parameters and safety considerations, the standardized synthesis steps are provided below.
- Prepare the reaction mixture by adding fullerene C60, gamma,delta-unsaturated oxime ester, and FeCl2·4H2O catalyst into a dry Schlenk tube.
- Add O-dichlorobenzene and acetonitrile solvents, seal the tube, and sonicate to dissolve reactants completely before heating.
- Heat the mixture in an oil bath at 70-120°C under nitrogen, then purify the crude product using silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology offers transformative benefits that extend beyond mere technical feasibility. The shift from precious metals to inexpensive iron salts fundamentally alters the cost structure of producing high-value fullerene derivatives, removing a major barrier to entry for large-scale adoption in the electronics industry. Additionally, the robustness of the reaction conditions means that manufacturing can be scaled up without requiring exotic equipment or extreme safety measures, thereby enhancing supply chain reliability and reducing the risk of production delays. The broad substrate scope allows for the rapid generation of diverse libraries of materials, enabling faster time-to-market for new electronic formulations. This agility is a critical competitive advantage in the fast-paced sector of advanced materials.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with commercially available iron salts results in substantial cost savings on raw materials. Moreover, the simplified purification process reduces solvent consumption and labor hours associated with complex metal scavenging steps. This economic efficiency translates directly into a more competitive pricing structure for the final API intermediate or electronic chemical, allowing partners to maintain healthy margins even in volatile market conditions. The ability to recover unreacted C60 further optimizes material utilization, ensuring that every gram of input contributes to value creation.
- Enhanced Supply Chain Reliability: Sourcing high-purity catalysts and specialized reagents can often be a bottleneck; however, iron salts and oxime esters are commodity chemicals with stable global supply chains. This availability mitigates the risk of shortages that frequently affect rare earth or precious metal-dependent processes. Consequently, manufacturers can guarantee consistent delivery schedules and maintain safety stock levels with greater confidence. The resilience of this supply chain is further bolstered by the method's tolerance to minor variations in reaction conditions, ensuring consistent output quality across different production batches and facilities.
- Scalability and Environmental Compliance: The process is inherently green, operating under neutral conditions without generating hazardous heavy metal waste streams. This aligns perfectly with increasingly stringent environmental regulations globally, reducing the burden of waste disposal and compliance reporting. The simplicity of the reaction setup facilitates seamless scale-up from gram-scale laboratory optimization to ton-scale commercial production. This scalability ensures that as demand for fullerene-based electronic materials grows, the supply can expand proportionally without the need for prohibitive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fullerene derivatives. These insights are derived directly from the patent data and practical manufacturing experience, aimed at clarifying the feasibility and benefits of this technology for potential partners. Understanding these details is essential for making informed decisions about integrating these materials into your product development pipeline.
Q: What are the advantages of using iron salts over precious metal catalysts for fullerene functionalization?
A: Using transition metal iron salts significantly reduces raw material costs compared to rhodium or palladium catalysts. Furthermore, iron catalysis operates under neutral redox conditions, minimizing side reactions and simplifying the purification process by avoiding complex metal removal steps.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates broad substrate universality, accommodating various gamma,delta-unsaturated oxime esters where R can be hydrogen, alkyl, aryl, or heteroaryl groups. This flexibility allows for the creation of diverse fullerene derivatives tailored for specific electronic applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes simple reaction conditions (70-120°C), common solvents (ODCB/MeCN), and avoids sensitive reagents, making it highly adaptable for commercial scale-up from laboratory to industrial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-[60] Fullerene Pyrrolidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fullerene derivatives play in the next generation of organic electronics and photovoltaic devices. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance electronic materials. We are committed to delivering not just a chemical product, but a comprehensive solution that supports your innovation goals with reliability and technical excellence.
We invite you to collaborate with us to leverage this cutting-edge iron-catalyzed technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in commercial scale-up of complex electronic chemical manufacturing can drive value for your organization.
