Industrial Scale Synthesis of 3,3-Diaminobiphenol Hydrochloride for High-Performance Polymers
The global demand for high-performance polymers, particularly poly(p-phenylene benzobisoxazole) or PBO, has necessitated the development of robust and scalable synthetic routes for their key monomers. Patent CN110156613B, published in late 2022, introduces a groundbreaking synthetic method for 3,3-diaminobiphenol hydrochloride, a critical building block for hydroxyl-modified PBO polymers used in aerospace and heat-resistant applications. This technology addresses long-standing challenges in the fine chemical industry by offering a pathway that combines high atom utilization with stringent safety controls. By shifting away from hazardous traditional reagents towards a more controlled nitration-reduction sequence, this innovation provides a reliable foundation for the commercial scale-up of complex polymer additives. The structural integrity of the target molecule, as depicted below, is paramount for the thermal stability of the resulting polymers.
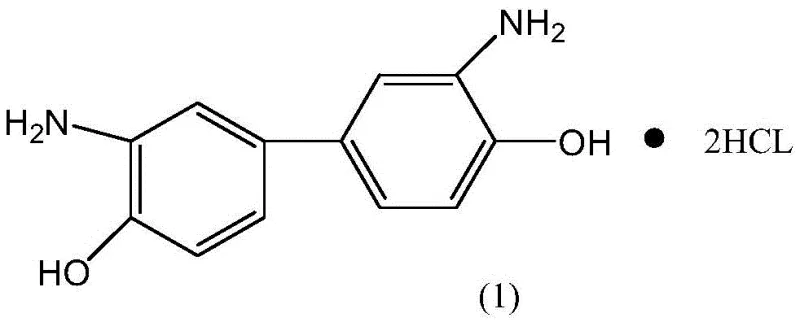
For R&D directors and procurement managers alike, the implications of this patent extend beyond mere chemical synthesis; it represents a strategic opportunity for cost reduction in polymer synthesis additives manufacturing. The ability to produce high-purity 3,3-diaminobiphenol hydrochloride with minimal toxic byproducts aligns perfectly with modern environmental compliance standards while ensuring a steady supply of critical raw materials. As we delve into the technical specifics, it becomes clear that this method offers a superior alternative to legacy processes, promising enhanced supply chain reliability and significant operational efficiencies for downstream manufacturers of advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-diaminobiphenol hydrochloride has been plagued by significant technical hurdles that hinder large-scale adoption. Prior art, such as the methods cited by Samuel Guieu, often relies on reaction conditions that pose severe safety risks due to the generation of unstable intermediates and highly toxic byproducts. These conventional routes frequently suffer from poor reaction selectivity, leading to a complex mixture of impurities that are notoriously difficult to separate, thereby drastically reducing the overall yield and purity of the final product. Furthermore, the reliance on expensive or hard-to-source materials in some existing literature, such as the work by Qiang Liu, creates a bottleneck for industrial production capacity. The combination of low yields, difficult purification processes, and safety concerns makes these traditional methods economically unviable for meeting the growing global demand for high-performance polymer precursors.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN110156613B leverages a streamlined two-step process that prioritizes both safety and efficiency. By utilizing 4,4-biphenol as a readily available starting material and employing a carefully controlled nitration followed by a specific reduction protocol, this method eliminates the need for exotic reagents. The process is characterized by its mild reaction conditions and high selectivity, which inherently minimizes the formation of unwanted side products. This shift in methodology not only simplifies the post-treatment workflow but also ensures that the final product meets the rigorous purity specifications required for aerospace and electronic applications. The result is a synthesis route that is not only chemically elegant but also commercially robust, offering a clear path toward reducing lead time for high-purity polymer synthesis additives.
Mechanistic Insights into Nitration-Reduction Sequence
The core of this innovative synthesis lies in the precise execution of the nitration step, where 4,4-biphenol is reacted with concentrated nitric acid in a mixed solvent system of toluene and glacial acetic acid. The reaction is conducted under strict ice bath conditions, maintaining a temperature between 0°C and 5°C, which is critical for controlling the exothermic nature of the nitration. This thermal management prevents local overheating that could otherwise lead to polynitration or oxidative degradation of the aromatic rings. The molar ratio of 4,4-biphenol to nitric acid is optimized between 1:2 and 1:2.2, ensuring complete conversion while minimizing excess acid waste. Following the reaction, the intermediate 3,3-dinitrobiphenol is isolated through a sophisticated purification process involving washing with water and methanol, followed by recrystallization in a toluene and N,N-dimethylformamide mixture, yielding a yellow solid with purity exceeding 98%.
The subsequent reduction step transforms the dinitro intermediate into the final diamine product using a green chemistry approach involving hydrazine hydrate and ferrous sulfate heptahydrate in absolute ethanol. Refluxing at 80°C for approximately 9 to 10 hours allows for the complete reduction of the nitro groups to amino groups without affecting the phenolic hydroxyl groups. The inclusion of activated carbon and ferrous sulfate acts as a catalytic system that enhances the efficiency of hydrazine while suppressing the formation of azo-byproducts. Post-reaction processing involves hot filtration and dissolution in dilute hydrochloric acid, followed by pH adjustment to precipitate the 3,3-diaminobiphenol hydrochloride as a white solid. This mechanism ensures high atom economy and avoids the use of heavy metal catalysts that would require costly removal steps, thereby streamlining the entire production workflow.
How to Synthesize 3,3-Diaminobiphenol Hydrochloride Efficiently
To implement this synthesis effectively in a pilot or production plant, operators must adhere to the specific parameter ranges defined in the patent to ensure reproducibility and safety. The process begins with the careful preparation of the solvent system and the slow addition of nitric acid to manage heat evolution, followed by the precise stoichiometric addition of reducing agents in the second step. Detailed standard operating procedures regarding temperature ramping, stirring rates, and filtration techniques are essential to maintain the high purity profiles observed in the experimental examples. For a comprehensive guide on the exact operational steps and safety protocols required to execute this synthesis, please refer to the standardized procedure outlined below.
- Perform nitration of 4,4-biphenol using concentrated nitric acid in a toluene and glacial acetic acid solvent system under strict ice bath conditions (0-5°C) to form 3,3-dinitrobiphenol.
- Purify the intermediate 3,3-dinitrobiphenol via filtration, washing with water and methanol, and recrystallization using a toluene and DMF mixed solvent.
- Execute reduction of the dinitro intermediate using hydrazine hydrate and ferrous sulfate heptahydrate in absolute ethanol at reflux (80°C), followed by acidification to isolate the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers transformative benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for advanced material intermediates. The elimination of expensive and specialized catalysts in favor of commodity chemicals like ferrous sulfate and hydrazine hydrate translates directly into substantial cost savings in raw material procurement. Furthermore, the use of common solvents such as toluene and ethanol simplifies solvent recovery and recycling processes, further driving down operational expenditures. The robustness of the reaction parameters means that the process is less susceptible to batch-to-batch variability, ensuring a consistent supply of quality material that reduces the risk of production downtime for downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by replacing costly reagents with inexpensive, widely available alternatives like iron salts and hydrazine. By avoiding the use of precious metal catalysts, the method eliminates the need for complex and expensive metal scavenging steps, which traditionally add considerable cost to the manufacturing process. Additionally, the high yield of the nitration step, reported to be over 90%, maximizes the utilization of the starting 4,4-biphenol, reducing waste disposal costs and improving the overall material balance of the production line.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the raw materials required, including 4,4-biphenol and concentrated nitric acid, are commodity chemicals with established global supply chains. This reduces the dependency on single-source suppliers for niche reagents, thereby mitigating the risk of supply disruptions. The simplified post-treatment process also shortens the overall production cycle time, allowing for faster turnaround on orders and enabling manufacturers to respond more agilely to fluctuations in market demand for PBO monomers and related aerospace materials.
- Scalability and Environmental Compliance: The method is inherently designed for scale-up, with reaction conditions that are easily manageable in large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The absence of highly toxic byproducts simplifies wastewater treatment and waste management, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces the regulatory burden on the manufacturer but also enhances the sustainability credentials of the final product, which is a growing priority for end-users in the automotive and aerospace sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 3,3-diaminobiphenol hydrochloride based on the proprietary data found in patent CN110156613B. These insights are derived directly from the experimental examples and comparative analysis provided in the documentation, offering clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is crucial for technical teams evaluating the integration of this monomer into their own polymer synthesis workflows.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: Unlike conventional methods which suffer from poor safety profiles and difficult separation, this patented route utilizes easily controllable parameters, avoids highly toxic byproducts, and achieves significantly higher yields suitable for industrial application.
Q: What purity levels can be achieved with this process?
A: The process demonstrates exceptional selectivity, with the intermediate 3,3-dinitrobiphenol reaching purity levels above 98% and the final 3,3-diaminobiphenol hydrochloride product consistently exceeding 90% purity as verified by HPLC analysis.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method is designed for high industrial feasibility, utilizing common solvents like toluene and ethanol and avoiding expensive catalysts, which simplifies the scale-up process from laboratory to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Diaminobiphenol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We are committed to maintaining stringent purity specifications and operate rigorous QC labs to verify that every batch of 3,3-diaminobiphenol hydrochloride meets the exacting standards required for aerospace and high-performance material applications. Our expertise in process optimization allows us to deliver this complex intermediate with the consistency and reliability that global supply chains demand.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the advanced materials sector.
