Scalable Production of High-Purity 1,3-Propanediol Intermediates via Novel Hydroformylation
Scalable Production of High-Purity 1,3-Propanediol Intermediates via Novel Hydroformylation
The chemical industry continuously seeks more efficient pathways for producing critical diols like 1,3-propanediol (PDO), a vital monomer for high-performance polyesters. Patent CN1074405C introduces a transformative approach to synthesizing 1,3-alkanediols and 3-hydroxyaldehydes through the hydroformylation of oxiranes, specifically ethylene oxide. This technology addresses long-standing challenges in catalyst recovery and product stability that have historically hindered the economic viability of PDO production. By utilizing a Group VIII metal-based hydroformylation catalyst system, preferably unmodified cobalt carbonyls, the process achieves high yields while maintaining the integrity of the reactive intermediates. For procurement specialists and R&D leaders seeking a reliable agrochemical intermediate supplier or partners in fine chemical synthesis, understanding the nuances of this low-concentration, aqueous-extraction-based methodology is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 1,3-propanediol via hydroformylation faced significant technical barriers related to product isolation and catalyst management. Prior art, such as that disclosed in US-A-3687981, relied on separating the catalyst from cyclic hemiacetal products through phase separation, a method proven to be complicated and inadequate for commercial scale-up. Furthermore, alternative processes attempted direct preparation using excess phosphine-modified catalysts, which introduced prohibitive costs due to the expensive ligands and difficulties in retaining them during recycling. Perhaps most critically, traditional thermal separation methods like distillation often resulted in the degradation of the highly reactive 3-hydroxyaldehyde intermediates into heavy ends, drastically reducing overall yield and increasing waste disposal costs. These inefficiencies created a bottleneck for manufacturers aiming for cost reduction in electronic chemical manufacturing or polymer precursor production.
The Novel Approach
The patented process overcomes these hurdles by fundamentally rethinking the reaction environment and separation strategy. Instead of relying on thermal distillation, the invention employs a dilute reaction regime where the ethylene oxide concentration is maintained below 15 wt% at the start of the reaction. This specific constraint minimizes side reactions and allows for the formation of a reaction mixture that is ideally suited for aqueous extraction. By extracting the 3-hydroxyaldehyde into a water phase at moderate temperatures, the process avoids the thermal stress that causes degradation. Simultaneously, the cobalt catalyst remains largely in the organic solvent phase, enabling straightforward recycling without complex purification steps. This dual benefit of protecting the product and preserving the catalyst represents a significant leap forward in the commercial scale-up of complex polymer additives and intermediates.
Mechanistic Insights into Cobalt-Catalyzed Hydroformylation
The core of this technological advancement lies in the precise manipulation of the catalytic cycle using Group VIII metals, with cobalt carbonyls being the preferred species. The reaction proceeds under a stable carbon monoxide atmosphere, typically at pressures ranging from 3.5 to 34.6 MPa, which facilitates the formation of the active hydrido-carbonyl species necessary for inserting CO into the epoxide ring. A critical innovation in this mechanism is the optional use of lipophilic promoters, which accelerate the reaction rate without rendering the catalyst water-soluble. These promoters, which can include specific lipophilic tertiary amines or phosphine oxides, interact with the cobalt center to enhance turnover frequency (TOF) while maintaining the phase preference of the metal complex. This ensures that when the aqueous extraction step occurs, the valuable catalyst stays behind in the organic layer, ready for the next cycle.
To visualize the structural complexity of the promoters that enable this selective acceleration, consider the class of pyridine-substituted pyridines described in the patent documentation. 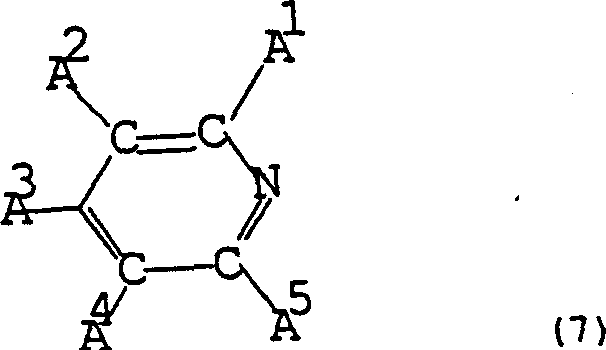 . These structures, characterized by their specific substitution patterns (where substituents A are selected to avoid steric hindrance), exemplify the type of lipophilic amines that can increase reaction rates significantly—up to 1.5 times faster in some embodiments—without compromising the separation efficiency. The ability to tune the electronic and steric properties of these promoters allows for fine control over the impurity profile, ensuring that by-products like acetaldehyde are minimized. This level of mechanistic control is paramount for R&D directors focused on purity specifications and impurity谱 analysis in high-value intermediate synthesis.
. These structures, characterized by their specific substitution patterns (where substituents A are selected to avoid steric hindrance), exemplify the type of lipophilic amines that can increase reaction rates significantly—up to 1.5 times faster in some embodiments—without compromising the separation efficiency. The ability to tune the electronic and steric properties of these promoters allows for fine control over the impurity profile, ensuring that by-products like acetaldehyde are minimized. This level of mechanistic control is paramount for R&D directors focused on purity specifications and impurity谱 analysis in high-value intermediate synthesis.
How to Synthesize 1,3-Propanediol Efficiently
Implementing this synthesis route requires careful attention to the sequential steps of hydroformylation, extraction, and hydrogenation to maximize yield and catalyst life. The process begins with the introduction of ethylene oxide and synthesis gas into a reactor containing the cobalt catalyst and a suitable solvent like methyl tert-butyl ether (MTBE). Following the reaction, the mixture undergoes a gentle aqueous extraction to isolate the intermediate, which is then hydrogenated to the final diol.
- Conduct hydroformylation of ethylene oxide using a Group VIII metal catalyst, preferably cobalt carbonyl, at temperatures below 100°C and controlled pressures.
- Separate the 3-hydroxyaldehyde intermediate from the reaction mixture using aqueous extraction to avoid thermal degradation and facilitate catalyst recycling.
- Hydrogenate the concentrated aqueous 3-hydroxyaldehyde solution over a nickel catalyst to yield high-purity 1,3-propanediol.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the shift from thermal separation to aqueous extraction offers profound logistical and financial benefits. The elimination of high-temperature distillation steps not only reduces energy consumption but also mitigates the risk of equipment fouling caused by heavy end formation. This translates directly into lower operational expenditures and reduced maintenance downtime for production facilities. Furthermore, the ability to recycle the cobalt catalyst efficiently means a drastic reduction in the consumption of expensive transition metals, which are subject to volatile market pricing. By adopting this methodology, manufacturers can achieve substantial cost savings in fine chemical manufacturing while ensuring a more stable and predictable supply of raw materials.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive phosphine ligands in many embodiments, relying instead on unmodified cobalt carbonyls which are more economical. Additionally, the high selectivity of the reaction reduces the burden on downstream purification units, lowering the overall cost per kilogram of produced 1,3-propanediol. The avoidance of thermal degradation means higher effective yields from the same amount of feedstock, further driving down the unit cost of production without requiring capital-intensive new infrastructure.
- Enhanced Supply Chain Reliability: By simplifying the separation process to a liquid-liquid extraction, the technology reduces the complexity of the manufacturing workflow. This simplicity enhances operational robustness, making it easier to maintain continuous production schedules and meet tight delivery windows. The reduced dependency on specialized, high-cost ligands also insulates the supply chain from disruptions in the availability of niche chemical reagents, ensuring a more consistent flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The use of moderate reaction temperatures and the absence of harsh thermal treatments align well with modern environmental, health, and safety (EHS) standards. The process generates fewer heavy waste streams associated with degraded by-products, simplifying waste management and disposal. This environmental efficiency facilitates easier regulatory compliance and supports the scaling of production from pilot plants to multi-ton commercial facilities with minimal ecological footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this hydroformylation technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the process feasibility.
Q: How does this process improve catalyst recovery compared to traditional distillation?
A: Traditional distillation often leads to thermal degradation of the reactive 3-hydroxyaldehyde intermediate. This patented method utilizes aqueous extraction at moderate temperatures (25-55°C), allowing over 90% of the cobalt catalyst to remain in the organic phase for direct recycling, significantly reducing metal loss.
Q: What are the optimal reaction conditions for maximizing selectivity?
A: The process achieves high selectivity by maintaining ethylene oxide concentrations below 15 wt% and operating at temperatures between 75-85°C. The use of lipophilic promoters further enhances reaction rates without compromising the phase separation required for catalyst recovery.
Q: Can this method be scaled for industrial polymer-grade production?
A: Yes, the process is designed for commercial scalability. By eliminating complex phase separation steps and avoiding high-temperature distillation of sensitive intermediates, the method supports continuous operation suitable for producing metric ton quantities of polymer-grade 1,3-propanediol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Propanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN1074405C can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,3-propanediol or related hydroformylation product adheres to the highest quality standards required by international clients.
We invite you to collaborate with us to explore how this advanced technology can optimize your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to support your R&D and sourcing decisions, ensuring a partnership built on transparency, technical excellence, and mutual growth.
