Advanced Catalytic Hydrogenation for High-Purity Alicyclic Oxetane Monomers
Advanced Catalytic Hydrogenation for High-Purity Alicyclic Oxetane Monomers
The landscape of advanced polymer chemistry is continually evolving, driven by the demand for materials with superior optical and thermal properties. A pivotal development in this sector is detailed in patent CN1922164A, which discloses a robust method for producing alicyclic oxetane compounds. These compounds are characterized by their exceptional light transmittance in the near-ultraviolet region and their utility as cationic polymerizable monomers. Unlike traditional aromatic counterparts, the alicyclic structures derived from this process offer enhanced heat resistance, toughness, and adhesiveness while minimizing curing shrinkage. For R&D directors and procurement specialists in the electronic materials sector, understanding this catalytic hydrogenation pathway is crucial for sourcing next-generation monomers that enable high-performance coatings, adhesives, and dental materials. The technology represents a significant leap forward in creating stable, high-reactivity monomers from commercially accessible aromatic precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in the patent data, the synthesis of polyfunctional oxetane compounds often relied on cumbersome and inefficient chemical pathways. Traditional methods frequently involved the conversion of hydroxymethyl oxetanes into sulfonate intermediates, which then required reaction with diols in the presence of alkali bases. This multi-step approach not only introduced significant complexity to the manufacturing process but also necessitated rigorous purification steps, such as distillation under reduced pressure, to isolate the final product. Furthermore, alternative routes utilizing chloromethyl oxetanes reacting with alicyclic diols often suffered from low reaction efficiency and prevalent side reactions. These inefficiencies resulted in suboptimal yields of the target compounds, thereby driving up production costs and creating supply chain vulnerabilities. The reliance on such convoluted synthetic routes hindered the widespread adoption of high-performance oxetane monomers in cost-sensitive industrial applications.
The Novel Approach
The methodology presented in the patent data offers a transformative solution by employing a direct ring hydrogenation strategy. Instead of building the alicyclic structure from scratch through complex coupling reactions, this novel approach starts with readily available aromatic oxetane compounds. By subjecting these aromatic precursors to a catalytic hydrogenation reaction under high-pressure hydrogen, the aromatic rings are efficiently saturated to form the desired alicyclic residues. This single-step transformation drastically simplifies the synthetic route, eliminating the need for hazardous sulfonate intermediates or low-yield substitution reactions. The process allows for the production of compounds like 4,4'-bis[(3-ethyl-oxetan-3-yl)methoxymethyl]bicyclohexane with high purity and structural integrity. This streamlined approach not only enhances the overall economic viability of the monomer production but also ensures a more consistent quality profile, which is essential for downstream applications in precision electronics and optics.
Mechanistic Insights into Catalytic Ring Hydrogenation
The core of this technological advancement lies in the precise execution of the catalytic hydrogenation mechanism. The reaction utilizes a heterogeneous catalyst system containing noble metals such as Ruthenium (Ru), Palladium (Pd), Platinum (Pt), or other transition metals like Nickel (Ni) and Cobalt (Co). In the preferred embodiments, a Ruthenium-based catalyst supported on carbon or alumina is employed to facilitate the addition of hydrogen across the aromatic double bonds. The reaction is conducted under elevated pressures, typically exceeding 1 MPa, and at temperatures ranging from 50°C to 250°C, with an optimal window between 100°C and 160°C. Under these conditions, the aromatic rings of the precursor molecule undergo saturation without compromising the integrity of the sensitive oxetane rings. This selectivity is paramount, as the preservation of the oxetane functionality is required for the subsequent cationic polymerization capabilities of the final monomer. The mechanism ensures that the resulting alicyclic structure retains the necessary steric and electronic properties for high reactivity.
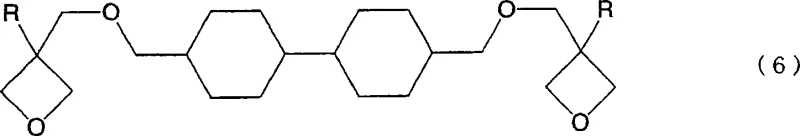
Controlling the impurity profile is another critical aspect of this mechanistic process. The hydrogenation conditions are tuned to minimize side reactions such as ring-opening of the oxetane moiety or over-reduction of other functional groups. The patent data indicates that the resulting products can achieve high degrees of hydrogenation, preferably 90% or more, ensuring that the final material possesses the desired lack of UV-absorbing aromatic chromophores. The removal of aromaticity is what grants the material its superior transparency in the near-ultraviolet spectrum, a key selling point for optical applications. Furthermore, the process allows for the formation of specific stereoisomers, such as trans-trans or trans-cis configurations, which can influence the physical properties like viscosity and crystallinity of the final cured resin. Understanding these mechanistic nuances allows manufacturers to tailor the material properties to specific end-use requirements, whether for flexible displays or rigid dental composites.
How to Synthesize Alicyclic Oxetane Compounds Efficiently
Implementing this synthesis route requires careful attention to reactor design and safety protocols due to the high-pressure hydrogen environment. The process begins with the charging of the aromatic oxetane precursor into a pressure-resistant vessel, followed by the addition of the solvent and catalyst. Detailed operational parameters regarding temperature ramping and pressure maintenance are critical for maximizing yield and safety. The standardized synthesis steps below outline the general procedure derived from the patent examples, providing a roadmap for scaling this chemistry from laboratory to commercial production.
- Load the aromatic oxetane precursor, such as 4,4'-bis[(3-ethyl-oxetan-3-yl)methoxymethyl]biphenyl, into a high-pressure autoclave equipped with agitation.
- Add a supported noble metal catalyst, specifically Ruthenium on carbon or alumina, along with a compatible solvent like ethyl acetate.
- Pressurize the system with hydrogen gas to approximately 6 MPa and maintain a temperature range of 100-160°C until hydrogen absorption ceases.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift to this hydrogenation-based manufacturing process offers profound advantages for supply chain stability and cost management. By simplifying the synthetic route from a multi-step derivation to a direct catalytic transformation, manufacturers can significantly reduce the number of unit operations required. This reduction translates directly into lower capital expenditure on equipment and reduced operational overheads associated with intermediate handling and purification. For procurement managers, this means a more resilient supply base capable of responding to demand fluctuations without the bottlenecks inherent in complex, multi-stage chemical syntheses. The ability to utilize common aromatic precursors, which are often produced at massive scales for other industries, further secures the raw material supply chain against volatility.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, such as sulfonyl chlorides used in traditional sulfonate pathways, leads to substantial cost savings in raw material procurement. Additionally, the simplified work-up procedure, which primarily involves catalyst filtration and solvent removal, reduces energy consumption and waste disposal costs. The high efficiency of the noble metal catalysts, which can often be recovered and reused, further contributes to a lower cost per kilogram of the final active ingredient. These factors combine to create a highly competitive cost structure for alicyclic oxetane monomers, making them viable for broader industrial adoption beyond niche high-end applications.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood hydrogenation technology ensures that production can be scaled reliably across different manufacturing sites. Unlike specialized coupling reactions that may require unique reagents with long lead times, hydrogen and supported metal catalysts are commodities with established global supply networks. This universality reduces the risk of supply disruptions caused by single-source dependencies. Furthermore, the shorter overall processing time associated with the one-step hydrogenation allows for faster turnaround times from order to delivery, enabling manufacturers to maintain leaner inventory levels while still meeting customer deadlines effectively.
- Scalability and Environmental Compliance: The process is inherently scalable, as high-pressure hydrogenation is a standard unit operation in the fine chemical industry, easily transferred from pilot plants to large-scale reactors. From an environmental standpoint, the atom economy of the hydrogenation reaction is superior to substitution reactions that generate stoichiometric amounts of salt waste. The reduction in solvent usage and the avoidance of halogenated intermediates align with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and mitigates the risk of production shutdowns due to environmental non-compliance, ensuring long-term business continuity for partners relying on these materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of alicyclic oxetane compounds. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry stakeholders. Understanding these details helps in making informed decisions regarding material selection and supplier qualification.
Q: What are the primary advantages of alicyclic oxetanes over aromatic analogues?
A: Alicyclic oxetanes exhibit significantly improved light transmission in the near-ultraviolet region due to the absence of aromatic chromophores, making them ideal for optical applications.
Q: Which catalysts are most effective for this ring hydrogenation process?
A: Catalysts containing metals such as Ruthenium (Ru), Palladium (Pd), or Platinum (Pt) supported on carbon or alumina provide high activity and selectivity for the ring saturation.
Q: How does this method improve supply chain reliability compared to traditional synthesis?
A: By utilizing a direct one-step hydrogenation of readily available aromatic precursors, the process eliminates complex multi-step derivatizations, reducing lead times and production bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alicyclic Oxetane Supplier
As the demand for high-performance optical and electronic materials continues to surge, securing a reliable supply of advanced monomers like alicyclic oxetanes is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing catalytic hydrogenation processes to meet stringent purity specifications, ensuring that every batch delivers the consistent performance required for critical applications in display technologies and precision coatings. With rigorous QC labs and a commitment to process excellence, we guarantee the structural integrity and optical clarity of our alicyclic oxetane offerings.
We invite R&D directors and procurement leaders to collaborate with us to unlock the full potential of these advanced materials. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific formulation needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing processes can enhance your product portfolio while reducing overall material costs. Let us be your strategic partner in navigating the complexities of modern chemical supply chains.
