Advanced Oxidative Dimerization For Commercial Scale Tetraalkylbisphenol Production
The chemical manufacturing landscape is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for high-value intermediates. Patent CN100339346C introduces a groundbreaking method for producing 3,3',5,5'-tetraalkyl-4,4'-bisphenol, a critical compound used extensively in the formulation of epoxy resins, polycarbonates, and specialized polymer additives. This technology addresses a long-standing industrial challenge regarding the recovery and reuse of unreacted 2,6-dialkylphenol without compromising reaction kinetics or product purity. By implementing a controlled oxidative dimerization process in an aqueous solvent system, manufacturers can achieve significant improvements in yield consistency and waste reduction. The core innovation lies in the precise management of aromatic aldehyde impurities that typically accumulate during recycling loops, which historically led to catalyst deactivation and prolonged processing times. For R&D Directors and Procurement Managers, understanding this mechanism is vital for securing a reliable supply chain of high-purity fine chemical intermediates. This report analyzes the technical merits of this patent to demonstrate how it translates into tangible commercial advantages for global chemical enterprises seeking cost reduction in electronic chemical manufacturing and polymer additive production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for tetraalkylbisphenols often suffer from significant inefficiencies when attempting to scale up production or implement raw material recycling protocols. In conventional oxidative dimerization processes, the reaction liquid remaining after solid-liquid separation contains substantial amounts of unreacted 2,6-dialkylphenol alongside the metal catalyst and various by-products. Historically, the industry standard involved disposing of this reaction liquid or attempting to recycle it without adequate purification, which led to the progressive accumulation of specific impurities. These impurities, particularly aromatic aldehydes formed by the oxidation of alkyl chains, act as potent catalyst poisons that coordinate with the active metal centers. As the concentration of these aldehydes increases over multiple batches, the reaction rate slows down drastically, requiring longer processing times and higher energy inputs to achieve the same conversion levels. Furthermore, the inability to effectively separate these impurities meant that a large volume of valuable unreacted raw material was often discarded as waste, driving up the overall cost of goods sold and creating environmental disposal burdens. This inefficiency creates a bottleneck for commercial scale-up of complex polymer additives, as the process becomes economically unviable when raw material utilization drops below critical thresholds.
The Novel Approach
The method disclosed in the patent data offers a sophisticated solution by integrating a controlled distillation and impurity management strategy directly into the synthesis workflow. Instead of blindly recycling the entire recovered fraction of unreacted dialkylphenol, this novel approach dictates that the amount of aromatic aldehydes relative to the metal catalyst must be adjusted to 60-fold mol or less before reintroduction into the reactor. This is achieved through a strategic distillation step where the aqueous slurry or the recovered reaction liquid is processed to separate the azeotropic mixture of water and unreacted raw material from the heavier impurity fractions. By selectively supplying only a portion of this recovered fraction back into the oxidative dimerization reaction, the system prevents the saturation of the catalyst with inhibitory aldehyde species. This ensures that the catalytic activity remains high and the reaction time remains consistent across multiple production cycles. Consequently, this approach allows for the repeated recovery and reuse of unreacted dialkylphenol without the typical degradation in performance, leading to a drastic simplification of the waste treatment process. For supply chain heads, this translates to a more robust manufacturing process that reduces lead time for high-purity intermediates by eliminating the variability associated with catalyst poisoning.
Mechanistic Insights into Cu-Catalyzed Oxidative Dimerization
The core of this synthesis relies on the oxidative dimerization of 2,6-dialkylphenol in the presence of a water solvent and a transition metal catalyst, typically a copper compound. The reaction proceeds through a radical mechanism where the phenolic hydroxyl group is activated by the basic aqueous environment, facilitating the coupling of two phenol units at the para-positions to form the 4,4'-bisphenol structure. Oxygen or an oxygen-containing gas serves as the oxidant, regenerating the active catalyst species and driving the equilibrium towards the desired dimer. However, a side reaction occurs where the alkyl groups on the phenol ring undergo oxidation, generating aromatic aldehydes as by-products. These aldehydes possess functional groups that are chemically similar to the phenolic substrates but interact differently with the metal catalyst. Specifically, the carbonyl oxygen in the aldehyde can coordinate strongly with the copper center, forming stable complexes that block the active sites required for phenol activation. This competitive inhibition is the primary reason for the observed increase in reaction time when impurities accumulate. Understanding this interaction is crucial for R&D teams aiming to optimize the process parameters for maximum efficiency and minimal by-product formation.
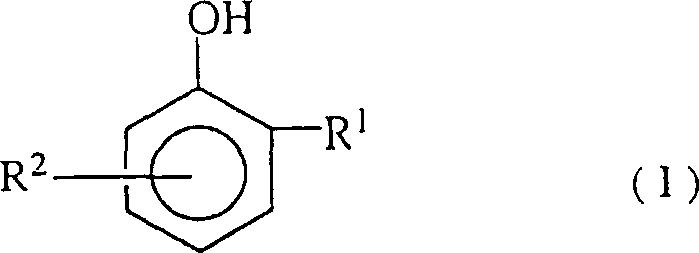
Controlling the impurity profile is therefore not just a matter of purification but a fundamental aspect of maintaining catalytic turnover. The patent specifies that the aromatic aldehydes, represented by General Formula (I) where R1 is a carbonyl group and R2 is an alkyl group, must be kept below a critical threshold relative to the catalyst concentration. When the molar ratio of these aldehydes to the metal catalyst exceeds 60:1, the inhibition effect becomes pronounced, leading to incomplete conversion and the need for extended reaction periods. By implementing a distillation step that concentrates these aldehydes in a waste stream while returning the purified dialkylphenol to the reactor, the process maintains the delicate balance required for high-speed catalysis. This mechanism ensures that the impurity spectrum remains stable, preventing the formation of heavy polymeric by-products like polyphenylene ethers that can contaminate the final product. For quality control teams, this means a more consistent impurity profile in the final tetraalkylbisphenol, reducing the need for extensive downstream purification and ensuring that stringent purity specifications are met for sensitive applications in the electrical and electronic fields.
How to Synthesize 3,3',5,5'-Tetraalkyl-4,4'-bisphenol Efficiently
Implementing this synthesis route requires careful attention to the sequence of reaction, separation, and recycling steps to maximize the benefits of the impurity control strategy. The process begins with the oxidative dimerization in an alkaline aqueous medium, followed by a pH adjustment and heating treatment to reduce quinone by-products before solid-liquid separation. The key to efficiency lies in the subsequent distillation of the mother liquor, where the unreacted 2,6-dialkylphenol is recovered as an azeotrope with water. Operators must monitor the concentration of aromatic aldehydes in this recovered stream and adjust the feed ratio to ensure the critical catalyst-to-impurity ratio is never exceeded in the subsequent batch. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Conduct oxidative dimerization of 2,6-dialkylphenol in aqueous solvent with metal catalyst.
- Separate tetraalkylbisphenol via solid-liquid separation from the aqueous slurry.
- Distill recovered liquid to control aromatic aldehyde impurities relative to catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial benefits that directly impact the bottom line and operational resilience of chemical manufacturing facilities. The primary advantage is the significant reduction in raw material consumption, as the ability to recycle unreacted 2,6-dialkylphenol without performance loss means less fresh feedstock is required per unit of output. This efficiency gain is compounded by the reduction in waste disposal costs, as the volume of reaction liquid requiring treatment is drastically minimized through the closed-loop recycling system. For procurement managers, this translates to a more stable cost structure that is less susceptible to fluctuations in the market price of dialkylphenol precursors. Furthermore, the process enhances supply chain reliability by reducing the risk of batch failures due to catalyst deactivation, ensuring that delivery schedules can be met consistently. The elimination of expensive heavy metal removal steps, often required when catalyst performance degrades and side reactions increase, further contributes to cost reduction in fine chemical intermediates manufacturing. Overall, this technology provides a competitive edge by aligning economic efficiency with environmental sustainability goals.
- Cost Reduction in Manufacturing: The ability to reuse unreacted raw materials repeatedly without the need for extensive purification or fresh catalyst addition leads to substantial cost savings over the lifecycle of the production campaign. By preventing the accumulation of catalyst-poisoning impurities, the process avoids the need for frequent catalyst replenishment, which is a significant cost driver in metal-catalyzed reactions. Additionally, the reduction in waste volume lowers the operational expenses associated with environmental compliance and hazardous waste disposal. This qualitative improvement in material efficiency ensures that the manufacturing process remains economically viable even when raw material prices fluctuate. The streamlined workflow also reduces energy consumption associated with heating and processing larger volumes of waste liquid, contributing to a lower overall carbon footprint for the facility.
- Enhanced Supply Chain Reliability: Consistency in reaction time and yield is critical for maintaining a reliable supply of high-purity intermediates to downstream customers. By controlling the impurity levels that cause reaction delays, this method ensures that production cycles are predictable and can be scheduled with high precision. This reliability reduces the need for safety stock and allows for a more lean inventory management strategy, freeing up working capital for other strategic investments. For supply chain heads, the ability to scale production without the risk of process instability means that large-volume orders can be fulfilled with confidence. The robustness of the process against impurity accumulation also means that the supply of critical raw materials is less likely to be disrupted by technical issues, ensuring continuity of supply for key accounts in the polymer and electronics industries.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as distillation and filtration that are easily adapted to large-scale reactors. The reduction in waste generation aligns with increasingly stringent environmental regulations, making it easier to obtain and maintain operating permits in regulated jurisdictions. By minimizing the discharge of organic contaminants and heavy metals, the facility can demonstrate a commitment to sustainable manufacturing practices, which is a growing requirement for global corporate customers. The simplified waste stream also reduces the complexity of effluent treatment plants, lowering capital expenditure for environmental infrastructure. This scalability ensures that the technology can be deployed across multiple production sites to meet growing global demand for tetraalkylbisphenol derivatives without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative dimerization technology. These answers are derived directly from the patent specifications to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this process in existing manufacturing setups. The focus is on clarifying the mechanism of impurity control and the practical implications for production planning.
Q: How does aromatic aldehyde accumulation affect the catalyst?
A: Aromatic aldehydes coordinate with the metal catalyst, reducing its activity and extending reaction time significantly.
Q: What is the critical molar ratio for impurity control?
A: The molar ratio of aromatic aldehydes to the metal catalyst must be maintained at 60:1 or less to ensure efficiency.
Q: Can unreacted raw materials be recycled safely?
A: Yes, by distilling the recovered fraction and managing impurity levels, unreacted dialkylphenol can be reused effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',5,5'-Tetraalkyl-4,4'-bisphenol Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patent methodologies like CN100339346C into robust, commercial-scale manufacturing processes. Our CDMO team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of catalyst management and impurity control are perfectly executed. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of tetraalkylbisphenol meets the exacting standards required for high-performance polymer and electronic applications. Our commitment to process optimization means we can deliver the cost and efficiency benefits described in this report to our global partners. We understand the critical nature of supply chain continuity and are dedicated to being a long-term strategic partner for your chemical sourcing needs.
We invite you to discuss how our manufacturing capabilities can support your specific project requirements and help you achieve your cost reduction goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to supply high-purity intermediates reliably. Let us help you optimize your supply chain with our advanced synthesis technologies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
