Revolutionizing 4-Hydroxy-2-ketobutyric Acid Production via Novel Aldolase Screening and Metabolic Engineering
Introduction to Breakthrough Biocatalytic Synthesis
The landscape of fine chemical manufacturing is undergoing a paradigm shift with the advent of advanced biocatalytic strategies, specifically highlighted in the recent patent CN116286761A. This intellectual property discloses a groundbreaking screening method for aldolase biological components capable of catalyzing the condensation of formaldehyde and pyruvic acid to synthesize 4-hydroxy-2-ketobutyric acid (4-HKB). Unlike traditional chemical synthesis which often relies on harsh conditions and toxic reagents, this biological approach leverages the specificity of enzymes belonging to the HpcH functional domain family. The invention identifies specific protein sequences, ranging from 240 to 320 amino acids, that exhibit remarkable stereoselectivity and catalytic efficiency under mild physiological conditions. For R&D directors and procurement specialists, this represents a pivotal opportunity to transition towards greener, more sustainable supply chains for high-value intermediates used in the production of homoserine, 3-hydroxypropionic acid, and 1,3-propanediol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biological utilization of one-carbon compounds like formaldehyde and methanol has been constrained by the inefficiencies of natural metabolic pathways. The Ribulose Monophosphate (RuMP) pathway, while widely studied, suffers critically from the slow regeneration rate of the Ru5P receptor, which acts as a bottleneck for carbon flux. This limitation severely restricts the ability of microbial hosts to assimilate methanol or formaldehyde at rates sufficient for industrial-scale chemical production. Furthermore, conventional chemical routes to 4-hydroxy-2-ketobutyric acid often involve multi-step organic synthesis requiring protecting groups, extreme temperatures, and expensive transition metal catalysts. These traditional methods generate significant hazardous waste and struggle to achieve the high stereochemical purity required for downstream pharmaceutical applications, thereby inflating purification costs and complicating regulatory compliance for global supply chains.
The Novel Approach
The novel approach detailed in the patent circumvents these metabolic bottlenecks by engineering a non-natural homoserine cycle. By screening vast protein libraries from databases like Uniprot and NCBI, researchers identified promiscuous aldolases that can accept formaldehyde as a substrate alongside pyruvate. This strategy effectively decouples carbon assimilation from the restrictive RuMP cycle, allowing for a direct, streamlined condensation reaction. The implementation of this pathway in engineered Escherichia coli strains, specifically those with knocked-out frmA genes to prevent formaldehyde detoxification, creates a highly efficient cellular factory. This method not only simplifies the metabolic route but also capitalizes on the high reactivity of formaldehyde in a controlled enzymatic environment, transforming a toxic waste product into a valuable building block for complex organic molecules with minimal byproduct formation.
Mechanistic Insights into HpcH-Catalyzed Condensation
The core of this technological advancement lies in the specific mechanistic action of the screened aldolases, particularly those belonging to the HpcH functional domain family. These enzymes facilitate a carbon-carbon bond formation between the carbonyl carbon of formaldehyde and the alpha-carbon of pyruvate. The patent highlights several high-performing candidates, such as CsAld from Curvibacter sp. and BsAld from Byssochlamys spectabilis, which demonstrate conversion rates exceeding 95% in vitro. The catalytic cycle relies on the formation of a Schiff base intermediate or a metal-dependent enolate mechanism, typical of class I or II aldolases, ensuring precise stereocontrol during the bond formation. This level of enzymatic precision eliminates the formation of racemic mixtures common in chemical catalysis, thereby reducing the burden on downstream chiral separation processes and ensuring a consistent, high-purity product stream suitable for sensitive pharmaceutical syntheses.
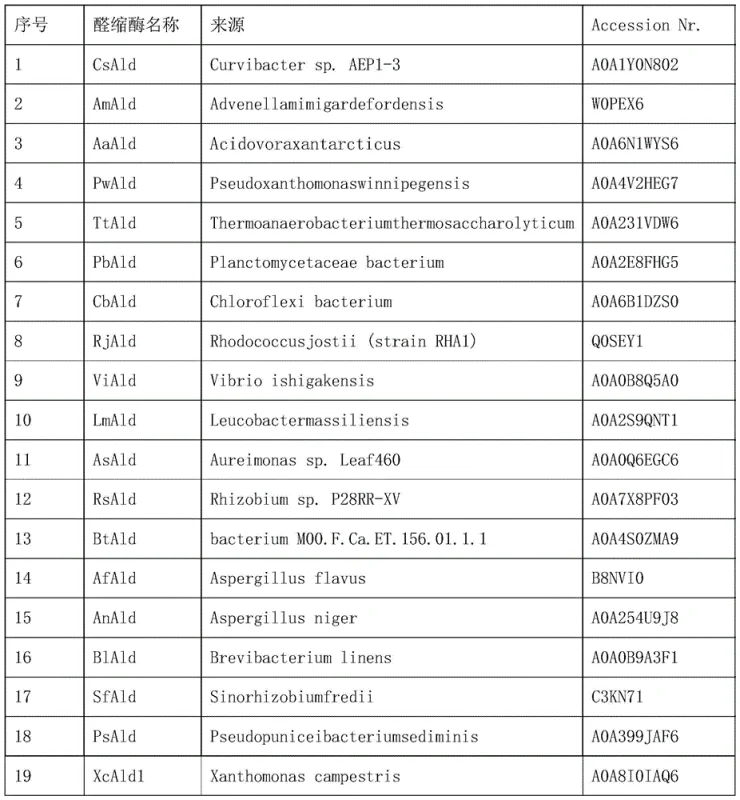
Furthermore, the impurity profile of this biocatalytic process is inherently cleaner due to the substrate specificity of the enzymes. Unlike broad-spectrum chemical catalysts that may react with various functional groups present in the fermentation broth, these aldolases are tuned to recognize specific ketone and aldehyde motifs. The patent data indicates that by optimizing the host strain to weaken the native glutathione-dependent formaldehyde detoxification system (frmRAB), intracellular formaldehyde accumulation is maximized for the desired condensation reaction rather than being oxidized to formic acid or carbon dioxide. This metabolic channeling ensures that the carbon flux is directed almost exclusively towards the target 4-hydroxy-2-ketobutyric acid, minimizing the generation of hard-to-remove side products and simplifying the overall purification workflow for commercial manufacturing.
How to Synthesize 4-Hydroxy-2-ketobutyric Acid Efficiently
To implement this synthesis route effectively, manufacturers must follow a precise fermentation and induction protocol optimized for recombinant protein expression. The process begins with the cultivation of seed cultures in LB medium supplemented with appropriate antibiotics to maintain plasmid stability, followed by transfer to a defined inorganic salt medium. Critical control points include the timing of IPTG induction relative to cell density (OD600) and the temperature shift to 30°C to balance enzyme activity with cell viability. The detailed standardized synthesis steps, including specific media compositions, feeding strategies for methanol or sarcosine substrates, and downstream processing parameters, are outlined in the technical guide below to ensure reproducible high-yield production.
- Cultivate recombinant E. coli W3110△frmA strains carrying aldolase and methanol dehydrogenase genes in LB medium with carbenicillin at 37°C overnight.
- Transfer seed culture to inorganic salt medium with glucose and methanol, inducing gene expression with IPTG when OD600 reaches 0.5.
- Maintain fermentation at 30°C for 24 hours with periodic substrate feeding, followed by centrifugation and HPLC analysis for product quantification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers profound strategic advantages beyond mere technical feasibility. The shift from petrochemical-derived precursors to bio-based feedstocks like methanol or sarcosine insulates the supply chain from volatile oil prices and geopolitical instability associated with fossil fuel sourcing. Additionally, the elimination of heavy metal catalysts and harsh organic solvents drastically reduces the environmental footprint of the manufacturing process, aligning with increasingly stringent global ESG (Environmental, Social, and Governance) mandates. This compliance advantage translates into smoother regulatory approvals and enhanced brand reputation for downstream customers in the pharmaceutical and nutraceutical sectors who are under pressure to green their own supply chains.
- Cost Reduction in Manufacturing: The biocatalytic route significantly lowers operational expenditures by removing the need for expensive transition metal catalysts and complex purification steps associated with removing metal residues. The mild reaction conditions, operating at ambient pressure and moderate temperatures (30°C), reduce energy consumption for heating and cooling compared to traditional high-pressure chemical synthesis. Furthermore, the high stereoselectivity of the aldolase enzymes minimizes the loss of raw materials to unwanted isomers, improving overall atom economy and reducing the cost of goods sold through higher effective yields of the desired active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Utilizing robust microbial hosts like Escherichia coli ensures a scalable and reliable production platform that is less susceptible to the supply disruptions common in specialty chemical markets. The ability to produce 4-hydroxy-2-ketobutyric acid from simple, widely available substrates like methanol or glycine derivatives (sarcosine) diversifies the raw material base. This flexibility allows manufacturers to switch feedstock sources based on availability and price without retooling the entire production line, thereby securing continuity of supply for critical downstream customers even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The fermentation-based process is inherently scalable, leveraging decades of established infrastructure in the industrial biotechnology sector. Scaling from laboratory benchtop to multi-ton commercial bioreactors follows well-understood kinetic models, reducing the technical risk associated with process scale-up. Moreover, the aqueous nature of the reaction medium and the biodegradability of the biological catalysts simplify wastewater treatment protocols. This reduces the capital investment required for effluent treatment plants and ensures compliance with environmental discharge regulations, avoiding potential fines and operational shutdowns related to pollution control.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aldolase-based synthesis pathway. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on substrate selection, strain engineering, and process optimization. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing portfolios for fine chemicals and pharmaceutical intermediates.
Q: How does this novel aldolase screening method overcome traditional formaldehyde utilization limitations?
A: Traditional RuMP pathways are limited by inefficient Ru5P receptor regeneration. This patent introduces a non-natural homoserine cycle using promiscuous aldolases (HpcH family) that directly condense formaldehyde with pyruvate, bypassing complex regeneration steps and enabling higher efficiency.
Q: What are the specific advantages of using sarcosine over methanol as a substrate in this pathway?
A: While both substrates work, experimental data indicates that using sarcosine as a formaldehyde precursor yields significantly higher titers of 4-hydroxy-2-ketobutyric acid (e.g., 5.33mM vs 0.95mM with methanol) due to more efficient intracellular formaldehyde generation and reduced toxicity issues.
Q: Is the enzymatic process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard E. coli fermentation hosts (W3110) and common expression vectors (pTrc99a), ensuring compatibility with existing industrial bioreactor infrastructure. The mild reaction conditions (30°C, neutral pH) further facilitate easy scale-up without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-2-ketobutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic pathways described in patent CN116286761A for the production of high-value intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 4-hydroxy-2-ketobutyric acid meets the exacting standards required for pharmaceutical grade applications. We are committed to leveraging our expertise in metabolic engineering and fermentation technology to deliver consistent, high-quality supply solutions.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced biocatalysis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this sustainable and efficient synthesis method into your product portfolio. Together, we can drive the next generation of green chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
