Advanced Amino Protection Reagent Cpac-Bt for Scalable Pharmaceutical Synthesis
The landscape of fine chemical synthesis, particularly within the realm of peptide and pharmaceutical intermediate manufacturing, is constantly evolving to meet the demands for higher purity and greater process efficiency. A significant advancement in this field is documented in patent CN101139324B, which introduces a novel amino-protecting reagent known as N-benzotriazole phenacyl carbonate, or Cpac-Bt. This innovative reagent addresses several longstanding challenges associated with traditional protecting groups by offering a combination of mild reaction conditions, high stability, and a unique deprotection mechanism. For R&D directors and process chemists, the introduction of Cpac-Bt represents a strategic opportunity to streamline synthetic routes, particularly for complex molecules where orthogonal protection is critical. The reagent is characterized by its white crystalline form and molecular formula C15H11N3O3, indicating a robust structure that facilitates easy handling and storage compared to more volatile or sensitive alternatives currently available in the market.
The utility of Cpac-Bt extends beyond mere protection; it enables the synthesis of benzoylmethoxycarbonyl-protected amine compounds with exceptional fidelity. In the context of industrial scale-up, the ability to utilize such a reagent can significantly impact the overall yield and purity profile of the final active pharmaceutical ingredient (API). By providing a method that operates effectively at temperatures ranging from -5°C to 25°C, the patent outlines a process that is not only energy-efficient but also safer to operate on a large scale. This technical breakthrough positions Cpac-Bt as a vital tool for manufacturers seeking to optimize their production workflows while maintaining stringent quality control standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the protection of amino groups in organic synthesis has relied heavily on reagents such as benzyl chloroformate (Cbz-Cl), di-tert-butyl dicarbonate (Boc anhydride), and trityl chloride. While these reagents have served the industry well for decades, they possess inherent limitations that can complicate modern synthetic strategies. For instance, the removal of the Cbz group typically necessitates catalytic hydrogenation using palladium on carbon, a process that poses safety risks due to the use of hydrogen gas and can be problematic when the molecule contains other hydrogenation-sensitive functionalities like olefins or benzyl esters. Similarly, Boc and Trt groups require acidic conditions for deprotection, which can lead to the degradation of acid-labile moieties elsewhere in the complex molecular architecture. These constraints often force chemists into convoluted synthetic sequences to avoid incompatibility, thereby increasing both the time and cost associated with drug development and manufacturing.
The Novel Approach
In stark contrast to these conventional methods, the novel approach utilizing N-benzotriazole phenacyl carbonate (Cpac-Bt) offers a paradigm shift in amino protection chemistry. The preparation of this reagent involves the reaction of alpha-hydroxyacetophenone with N,N'-carbonyldibenzotriazole (DBC) in the presence of a base, a process that proceeds smoothly at mild temperatures to yield the reagent in high purity. 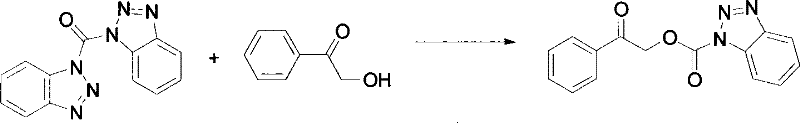 The resulting Cpac-Bt reagent is a stable solid that can be easily stored and handled, eliminating the logistical challenges associated with liquid or unstable protecting agents. Furthermore, the application of Cpac-Bt allows for the protection of amines under gentle alkaline conditions, producing crystalline protected intermediates that are exceptionally easy to purify via recrystallization. This ease of purification is a critical advantage in industrial settings, as it reduces the reliance on resource-intensive chromatographic separations, thereby enhancing the overall throughput and economic viability of the manufacturing process.
The resulting Cpac-Bt reagent is a stable solid that can be easily stored and handled, eliminating the logistical challenges associated with liquid or unstable protecting agents. Furthermore, the application of Cpac-Bt allows for the protection of amines under gentle alkaline conditions, producing crystalline protected intermediates that are exceptionally easy to purify via recrystallization. This ease of purification is a critical advantage in industrial settings, as it reduces the reliance on resource-intensive chromatographic separations, thereby enhancing the overall throughput and economic viability of the manufacturing process.
Mechanistic Insights into Cpac Protection and Deprotection
The mechanistic pathway for the formation and subsequent removal of the Cpac group is central to understanding its value proposition for complex synthesis. During the protection phase, the carbonyl carbon of the Cpac-Bt reagent acts as a potent electrophile, susceptible to nucleophilic attack by the lone pair of electrons on the amine nitrogen. This reaction is facilitated by the presence of a base, which deprotonates the amine or scavenges the proton released during the acylation event, driving the equilibrium towards the formation of the stable urethane linkage. The benzotriazole moiety serves as an excellent leaving group, ensuring that the reaction proceeds rapidly and to completion even at low temperatures, minimizing the risk of side reactions such as racemization in chiral amino acid derivatives. This high level of chemoselectivity ensures that the integrity of the substrate is maintained throughout the protection step.
Perhaps the most distinctive feature of the Cpac strategy is its deprotection mechanism, which relies on a reductive cleavage rather than hydrolysis or hydrogenolysis. 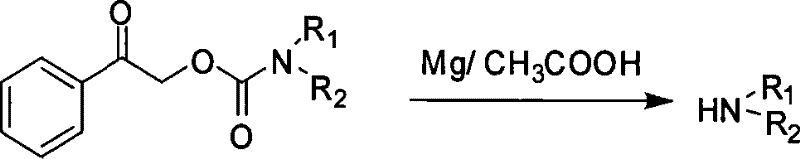 As illustrated in the reaction scheme, the protected amine is treated with a metal powder, such as magnesium or zinc, in the presence of glacial acetic acid. This system generates a mild reducing environment that specifically targets the benzylic ketone functionality within the protecting group. The reduction triggers a fragmentation cascade that releases the free amine along with acetophenone and carbon dioxide byproducts. Crucially, this reductive condition is orthogonal to many other common protecting groups; it does not affect methyl, benzyl, or tert-butyl esters, nor does it interfere with Cbz, Boc, or Fmoc groups. This orthogonality provides synthetic chemists with a powerful tool for selective deprotection in multi-functional molecules, allowing for precise control over the synthetic sequence without compromising other sensitive structural elements.
As illustrated in the reaction scheme, the protected amine is treated with a metal powder, such as magnesium or zinc, in the presence of glacial acetic acid. This system generates a mild reducing environment that specifically targets the benzylic ketone functionality within the protecting group. The reduction triggers a fragmentation cascade that releases the free amine along with acetophenone and carbon dioxide byproducts. Crucially, this reductive condition is orthogonal to many other common protecting groups; it does not affect methyl, benzyl, or tert-butyl esters, nor does it interfere with Cbz, Boc, or Fmoc groups. This orthogonality provides synthetic chemists with a powerful tool for selective deprotection in multi-functional molecules, allowing for precise control over the synthetic sequence without compromising other sensitive structural elements.
How to Synthesize N-Benzotriazole Phenacyl Carbonate Efficiently
The synthesis of Cpac-Bt is designed to be straightforward and scalable, making it accessible for both laboratory research and industrial production. The process begins with the dissolution of alpha-hydroxyacetophenone in a suitable organic solvent such as acetonitrile, nitromethane, or dichloromethane. A base, such as diisopropylethylamine or pyridine, is added to the mixture to maintain the necessary alkalinity for the reaction. Subsequently, N,N'-carbonyldibenzotriazole (DBC) is introduced slowly while maintaining the temperature between -5°C and 25°C to control the exotherm and ensure high selectivity. The reaction typically proceeds to completion within 3 to 4 hours, yielding the crude reagent which can be isolated and purified to obtain white crystals with a yield exceeding 85%. For detailed operational parameters and specific stoichiometric ratios optimized for different scales, please refer to the standardized synthesis guide below.
- Synthesize Cpac-Bt by reacting alpha-hydroxyacetophenone with N,N'-carbonyldibenzotriazole (DBC) in an organic solvent like acetonitrile or dichloromethane with a base at -5 to 25°C.
- Protect the target amine by reacting it with Cpac-Bt in the presence of a base at mild temperatures (-5 to 30°C) to form the benzoylmethoxycarbonyl protected derivative.
- Remove the Cpac protecting group by treating the protected amine with magnesium or zinc powder in glacial acetic acid within an organic solvent, preserving other sensitive functional groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of Cpac-Bt technology offers compelling advantages that translate directly into operational efficiency and cost optimization. The primary driver for cost reduction lies in the simplicity of the reagent's synthesis and the accessibility of its raw materials. Alpha-hydroxyacetophenone and DBC are commercially available commodity chemicals, ensuring a stable and reliable supply chain that is not subject to the volatility often seen with specialized catalysts or rare reagents. Furthermore, the elimination of precious metal catalysts like palladium, which are traditionally required for Cbz deprotection, removes a significant cost center from the manufacturing budget. This shift not only reduces direct material costs but also simplifies the waste management process, as there is no need for the recovery or disposal of heavy metal residues, leading to substantial savings in environmental compliance and waste treatment.
- Cost Reduction in Manufacturing: The implementation of Cpac-Bt significantly lowers the overall cost of goods sold (COGS) by streamlining the purification process. Since the protected amine intermediates tend to crystallize readily from the reaction mixture, manufacturers can rely on filtration and recrystallization rather than expensive and time-consuming column chromatography. This reduction in downstream processing requirements leads to faster batch turnover times and lower labor costs. Additionally, the high yields reported in the patent data indicate minimal material loss, maximizing the utilization of starting materials and further enhancing the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The stability of the Cpac-Bt reagent is a critical factor for supply chain resilience. Unlike some liquid protecting agents that may degrade over time or require cold chain logistics, Cpac-Bt is a stable solid that can be stored under ambient conditions without significant loss of potency. This characteristic allows for larger inventory buffers and reduces the risk of production delays caused by reagent spoilage. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing, ensuring consistent product quality across different batches and manufacturing sites, which is essential for maintaining a reliable supply of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling up the Cpac-Bt process is inherently safer and more environmentally friendly compared to traditional methods. The avoidance of high-pressure hydrogenation equipment reduces capital expenditure and operational risk, while the use of magnesium or zinc for deprotection generates benign byproducts that are easier to manage. The solvents used in the process, such as acetonitrile or ethyl acetate, are standard industrial solvents with well-established recovery and recycling protocols. This alignment with green chemistry principles not only supports corporate sustainability goals but also facilitates smoother regulatory approvals, as the impurity profile is cleaner and easier to characterize, reducing the burden on quality control laboratories.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating Cpac-Bt into their existing workflows, we have compiled a set of frequently asked questions based on the technical specifications and experimental data provided in the patent literature. These answers address common concerns regarding compatibility, stability, and operational parameters, offering a clear overview of what process engineers and chemists can expect when adopting this technology. Understanding these nuances is crucial for making informed decisions about process development and for anticipating potential challenges during the technology transfer phase.
Q: What is the primary advantage of Cpac-Bt over traditional Cbz protecting groups?
A: Unlike Cbz groups which typically require catalytic hydrogenation (H2/Pd) for removal, the Cpac group can be cleaved under mild reductive conditions using magnesium or zinc powder in acetic acid. This orthogonality allows Cpac to be removed without affecting other hydrogenation-sensitive groups like benzyl esters or unsaturated bonds.
Q: Is the Cpac-Bt reagent stable for long-term storage?
A: Yes, N-benzotriazole phenacyl carbonate (Cpac-Bt) is described as a stable white crystalline solid that is easy to preserve. Its stability simplifies inventory management and ensures consistent reactivity over time compared to more labile activating agents.
Q: Can the Cpac protecting group be used alongside Boc or Fmoc groups?
A: Absolutely. The deprotection conditions for Cpac (Mg/AcOH or Zn/AcOH) are chemically orthogonal to the acidic conditions required for Boc removal and the basic conditions for Fmoc removal. This makes Cpac-Bt highly valuable for multi-step synthetic strategies requiring orthogonal protection schemes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzotriazole Phenacyl Carbonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced protecting group chemistry like Cpac-Bt in accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-benzotriazole phenacyl carbonate or protected intermediate we deliver meets the highest international standards. We are committed to supporting your R&D efforts with high-quality materials that enable reproducible results and faster time-to-market for your critical pharmaceutical projects.
We invite you to explore how our expertise in orthogonal protection strategies can optimize your synthetic routes and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to the Cpac-Bt system. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us collaborate to build a more efficient, sustainable, and cost-effective supply chain for your next-generation therapeutics.
