Advanced Synthesis of 2-Chlorobenzimidazole Derivatives for Scalable Agrochemical Production
Advanced Synthesis of 2-Chlorobenzimidazole Derivatives for Scalable Agrochemical Production
The development of efficient synthetic routes for heterocyclic intermediates is a cornerstone of modern agrochemical manufacturing, particularly for the production of potent fungicides. Patent CN1425665A discloses a novel and highly effective process for preparing 2-chlorobenzimidazole derivatives, specifically targeting compounds with microbiocidal activity. This technology represents a significant leap forward from prior art methods, addressing critical bottlenecks in yield and scalability that have long plagued the synthesis of these valuable scaffolds. By utilizing 1,3-dihydrobenzimidazol-2-ones or their thione analogs as starting materials and reacting them with phosphoryl chloride, the process achieves unexpectedly high conversion rates. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages and commercial viability of this patented route is essential for optimizing supply chains and reducing overall manufacturing costs in the competitive fungicide market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industry practice for synthesizing 2-chlorobenzimidazole derivatives often relied on the halogen exchange reaction of corresponding 2-bromoimidazoles with hydrochloric acid, as described in documents like WO-A 97-06171. While chemically feasible, this legacy approach suffers from severe inefficiencies that make it economically unviable for large-scale production. The primary drawback is the notoriously low yield; comparative data indicates that converting 2-bromo-6,6-difluoro-[1,3]dioxolo[4,5-f]benzimidazole to its chloro-analog via HCl in dimethylformamide results in a theoretical yield of merely roughly 21.5%. Such poor efficiency not only drives up the cost of goods sold due to wasted raw materials but also creates significant waste disposal challenges. Furthermore, the requirement for specific bromo-precursors adds an extra layer of complexity and expense to the supply chain, as brominated intermediates are generally more costly and less stable than their oxygenated counterparts. These limitations necessitate a robust alternative that can deliver high purity without the economic penalty of low conversion rates.
The Novel Approach
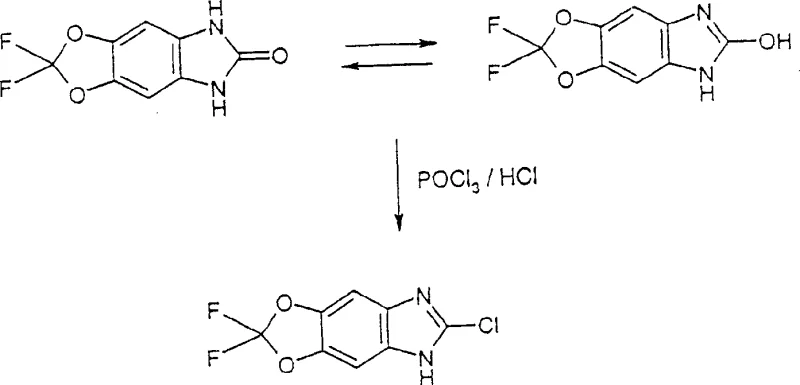
The method disclosed in CN1425665A fundamentally shifts the synthetic paradigm by employing 1,3-dihydrobenzimidazol-2-ones (or thiones) as the direct precursors for chlorination. Instead of struggling with difficult halogen exchanges on unstable bromo-species, this novel route leverages the reactivity of the carbonyl or thiocarbonyl group at the 2-position of the benzimidazole ring. By treating these ketones or thiones with phosphoryl chloride (POCl3), optionally in the presence of hydrogen chloride gas or phosphorus pentachloride, the process facilitates a smooth and high-yielding transformation into the desired 2-chloro derivative. Experimental embodiments demonstrate that this approach can achieve theoretical yields as high as 90%, a drastic improvement over the conventional 21% benchmark. This efficiency gain is not merely incremental; it transforms the economic model of producing these intermediates, making them accessible for cost-sensitive agrochemical applications while maintaining the rigorous purity standards required for downstream biological activity.
Mechanistic Insights into POCl3-Mediated Chlorination
The success of this synthetic route lies in the specific activation mechanism facilitated by phosphoryl chloride. In the reaction medium, the 1,3-dihydrobenzimidazol-2-one exists in equilibrium with its lactim tautomer, although the lactam form is generally predominant. Phosphoryl chloride acts as a powerful dehydrating and chlorinating agent, attacking the oxygen atom of the carbonyl group to form a reactive imidoyl chloride intermediate. This activation effectively converts the poor leaving group (hydroxyl, in the tautomeric sense) into an excellent leaving group, allowing for nucleophilic substitution by chloride ions present in the solution. The presence of hydrogen chloride gas further enhances the reaction kinetics by protonating the nitrogen atoms or the oxygen species, increasing the electrophilicity of the central carbon and driving the equilibrium towards the chlorinated product. This mechanism is particularly advantageous for substrates containing sensitive functional groups, such as the difluoromethylene dioxole moiety found in many modern fungicides, as the conditions are sufficiently mild to preserve these structures while being aggressive enough to effect complete chlorination at the 2-position.
From an impurity control perspective, this mechanism offers distinct advantages over radical halogenation or exchange methods. Because the reaction proceeds through a well-defined ionic pathway involving the carbonyl activation, side reactions such as ring halogenation or decomposition of the fused dioxole ring are minimized. The use of excess phosphoryl chloride serves a dual purpose: it acts as both the reagent and the solvent, ensuring homogeneous reaction conditions that prevent localized hot spots which could lead to degradation. Upon completion, the workup involves a simple quench into ice water, where the excess POCl3 hydrolyzes harmlessly to phosphoric acid and HCl, facilitating easy separation of the organic product. This clean reaction profile ensures that the resulting 2-chlorobenzimidazole derivatives possess a high purity profile, reducing the burden on downstream purification steps and ensuring consistent quality for the final active pharmaceutical or agrochemical ingredient.
How to Synthesize 2-Chlorobenzimidazole Derivatives Efficiently
The practical implementation of this synthesis begins with the preparation of the key precursor, the 1,3-dihydrobenzimidazol-2-one. As illustrated in the patent data, these precursors can be efficiently generated by reacting the corresponding phenylenediamines with phosgene, carbonyldiimidazole, or urea under controlled thermal conditions. For instance, reacting 5,6-diamino-2,2-difluoro-benzo[1,3]dioxole with phosgene in an aqueous or organic medium yields the cyclic urea in high purity. Once the precursor is secured, the chlorination step is straightforward: the solid ketone is suspended or dissolved in phosphoryl chloride and heated to reflux temperatures ranging from 70°C to 130°C. The reaction progress can be monitored via HPLC, and upon completion, the mixture is carefully poured onto crushed ice to precipitate the product. This operational simplicity makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to streamline production workflows.
- Prepare the 1,3-dihydrobenzimidazol-2-one precursor by reacting the corresponding phenylenediamine with phosgene or carbonyldiimidazole.
- Reflux the precursor with excess phosphoryl chloride (POCl3) at temperatures between 50°C to 150°C, optionally with HCl gas.
- Quench the reaction mixture into ice water, neutralize, and filter the resulting solid precipitate to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this POCl3-mediated synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The primary value driver is the substantial reduction in raw material costs associated with eliminating the need for expensive brominated intermediates. By starting from readily available diamines and converting them to the cyclic urea before chlorination, manufacturers can bypass the volatile pricing and supply constraints often associated with specialized bromo-heterocycles. Furthermore, the high yield of the process means that less raw material is required per kilogram of finished product, directly lowering the variable cost of production. This efficiency translates into a more competitive pricing structure for the final agrochemical intermediate, allowing buyers to secure better margins or pass savings on to their own customers in the formulation sector.
- Cost Reduction in Manufacturing: The elimination of low-yield halogen exchange steps drastically reduces the consumption of starting materials and solvents. Since the process utilizes phosphoryl chloride as both reagent and solvent, the need for additional exotic solvents is minimized, simplifying the bill of materials. The high conversion rate ensures that the throughput of the reactor is maximized, meaning that the same capital equipment can produce significantly more product over time compared to legacy methods. This operational efficiency leads to a lower cost per unit without compromising on the quality or purity specifications required for regulatory compliance in the agrochemical industry.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, specifically the substituted phenylenediamines, are commodity chemicals with robust global supply chains. Unlike specialized bromo-intermediates which may have limited suppliers, the diamines and phosgene equivalents used here are produced at scale by multiple vendors worldwide. This diversification of the supply base mitigates the risk of shortages and price spikes, ensuring a continuous flow of materials for production schedules. Additionally, the stability of the 1,3-dihydrobenzimidazol-2-one intermediates allows for easier storage and inventory management, providing flexibility in production planning and reducing the pressure on just-in-time delivery models.
- Scalability and Environmental Compliance: The workup procedure for this reaction is inherently scalable, relying on aqueous quenching and filtration rather than complex chromatographic separations. This simplicity facilitates the transition from pilot plant batches to multi-ton commercial production with minimal re-engineering of the process. From an environmental standpoint, while phosphoryl chloride requires careful handling, its hydrolysis products are manageable within standard wastewater treatment protocols for chemical plants. The high atom economy of the reaction, driven by the high yields, means less chemical waste is generated per unit of product, aligning with modern green chemistry initiatives and reducing the costs associated with hazardous waste disposal and environmental remediation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 2-chlorobenzimidazole derivatives. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making.
Q: What is the primary advantage of the POCl3 method over the bromo-imidazole route?
A: The POCl3 method offers significantly higher yields (up to 90% theoretical) compared to the traditional bromo-imidazole route, which often suffers from low conversion rates around 21%.
Q: Can this process be scaled for industrial fungicide production?
A: Yes, the process utilizes common reagents like phosphoryl chloride and simple aqueous workups, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: What are the typical reaction conditions for this chlorination?
A: The reaction typically proceeds at temperatures between 70°C to 130°C under reflux, often utilizing the phosphoryl chloride itself as both reactant and solvent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chlorobenzimidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the POCl3-mediated chlorination route described in CN1425665A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand the nuances of handling reactive reagents like phosphoryl chloride and the critical importance of maintaining stringent purity specifications throughout the synthesis. Our rigorous QC labs are equipped to monitor every stage of the reaction, from the initial diamine condensation to the final chlorination and isolation, ensuring that every batch meets the exacting standards required for high-performance fungicide manufacturing.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized process parameters and supply chain networks, we can help you achieve significant reductions in your cost of goods while securing a reliable source of high-purity intermediates. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us demonstrate how our commitment to innovation and quality can become a strategic asset in your supply chain, driving efficiency and reliability for your agrochemical portfolios.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
