Scalable Manufacturing of Chiral Cyclopropane R-Proline Derivatives for Advanced Organocatalysis
Introduction to Advanced Chiral Building Blocks
The landscape of asymmetric organocatalysis has been revolutionized by the introduction of constrained amino acid derivatives, specifically those featuring rigid cyclopropane structures that enhance stereochemical induction. Patent CN103435535A discloses a groundbreaking methodology for the preparation of R-proline analogs possessing a fused cyclopropane ring, addressing long-standing challenges in catalyst design and availability. This innovation is particularly significant for the development of next-generation pharmaceutical intermediates where precise spatial arrangement of functional groups dictates biological activity. The disclosed route transforms readily available R-pyroglutamic acid into highly valuable bicyclic scaffolds through a logical seven-step sequence that prioritizes atom economy and stereocontrol. By leveraging this technology, manufacturers can access high-purity organocatalysts that were previously limited by poor selectivity or prohibitive costs associated with chiral chromatography. The versatility of these compounds extends beyond simple proline mimics, serving as robust ligands for metal catalysis or standalone organocatalysts in complex bond-forming reactions such as Michael additions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
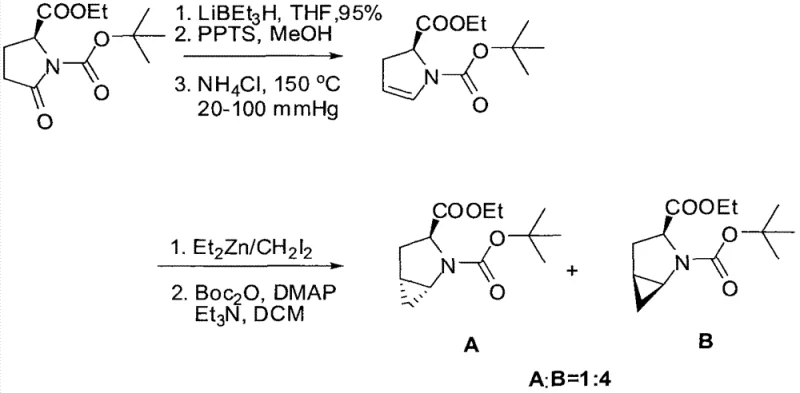
Historically, the synthesis of cyclopropane-fused proline derivatives relied heavily on methodologies that suffered from intrinsic inefficiencies and environmental hazards. Early approaches, such as those utilizing trimethylstannyl methyl lithium reagents for cyclopropanation, introduced severe toxicity concerns and required rigorous removal of heavy metal residues, complicating downstream processing for pharmaceutical applications. Furthermore, these legacy routes often exhibited poor diastereoselectivity, typically yielding mixtures of isomers in ratios as unfavorable as 1:4, necessitating extensive and yield-eroding purification steps like preparative HPLC or repeated recrystallization. The reliance on such non-selective transformations not only inflated the cost of goods but also created significant bottlenecks in supply chains due to the inability to predictably scale these erratic processes. Additionally, the use of sensitive organometallic reagents demanded stringent anhydrous conditions and specialized equipment, further limiting the accessibility of these valuable chiral building blocks for broader industrial adoption.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a streamlined strategy starting from inexpensive and abundant R-pyroglutamic acid, effectively bypassing the need for external chiral sources or resolution steps. By employing a diisobutylaluminum hydride (DIBAL-H) reduction followed by a controlled elimination, the synthesis generates a reactive alkene intermediate that is perfectly poised for stereoselective functionalization. The cornerstone of this improvement is the application of a modified Simmons-Smith cyclopropanation using diethylzinc and chloroiodomethane, which delivers the desired bicyclic framework with exceptional diastereomeric ratios exceeding 25:1. This dramatic improvement in selectivity essentially renders the separation of unwanted isomers unnecessary, thereby simplifying the workflow and significantly enhancing the overall mass balance of the process. The method replaces toxic tin reagents with safer zinc-based chemistry, aligning with modern green chemistry principles while simultaneously improving the economic viability of producing these complex chiral intermediates for the fine chemical industry.
Mechanistic Insights into Simmons-Smith Cyclopropanation and Stereocontrol
The mechanistic elegance of this synthesis lies in the precise orchestration of electronic and steric factors during the cyclopropanation step, which dictates the final stereochemical outcome. The reaction proceeds via the generation of a zinc carbenoid species from diethylzinc and chloroiodomethane, which acts as a methylene transfer agent to the electron-deficient alkene generated in the preceding elimination step. The presence of the adjacent nitrogen atom, protected as a bulky tert-butoxycarbonyl (Boc) group, exerts a powerful directing effect, shielding one face of the double bond and forcing the zinc carbenoid to approach from the less hindered trajectory. This facial selectivity is further amplified by the rigid conformation of the five-membered pyrrolidine ring, which locks the substituents into specific orientations that favor the formation of the (1R,3R,5R) isomer over its diastereomeric counterparts. The use of low temperatures, specifically maintaining the reaction between -20°C and -15°C, is critical to suppress non-selective background reactions and ensure that the kinetic product dominates the distribution. Such rigorous control over the transition state geometry allows for the direct isolation of the target enantiomer with high optical purity, eliminating the entropy-driven losses associated with thermodynamic equilibration or post-synthetic resolution.
Impurity control is meticulously managed throughout the seven-step sequence through the strategic selection of reagents and workup conditions that minimize side reactions. For instance, the initial esterification with thionyl chloride is conducted at sub-zero temperatures to prevent racemization of the alpha-center, while the subsequent DIBAL-H reduction is quenched carefully to avoid over-reduction to the amine or alcohol. The elimination step utilizes trifluoroacetic anhydride under basic conditions to generate the alkene cleanly without inducing polymerization or isomerization of the double bond. During the final hydrolysis and deprotection stages, the use of lithium hydroxide and trifluoroacetic acid respectively ensures that the sensitive cyclopropane ring remains intact, as harsh acidic or basic conditions could potentially lead to ring-opening degradation. This comprehensive attention to reaction parameters ensures that the final product meets the stringent purity specifications required for use as a catalyst in sensitive asymmetric transformations, where even trace impurities can poison the catalytic cycle or erode enantioselectivity.
How to Synthesize Cyclopropane R-Proline Efficiently
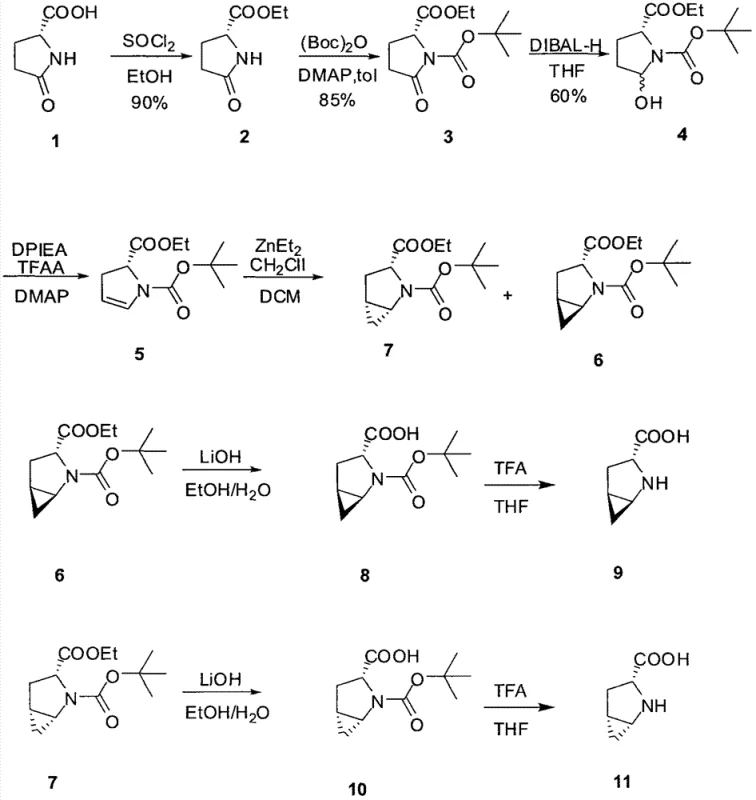
The synthesis of these high-value chiral intermediates follows a robust linear sequence that begins with the activation of R-pyroglutamic acid and culminates in the liberation of the free amino acid functionality. The process is designed to be operationally simple, relying on standard unit operations such as extraction, distillation, and crystallization that are familiar to process chemists and plant operators alike. Each step has been optimized to maximize yield and minimize the accumulation of byproducts, ensuring that the material flowing to the subsequent stage is of sufficient quality to drive the next transformation efficiently. While the specific stoichiometric ratios and temperature profiles are critical for success, the general workflow provides a reliable template for manufacturing that can be adapted to various batch sizes. For detailed operational parameters including exact molar equivalents, addition rates, and safety protocols, please refer to the standardized synthesis guide provided below which outlines the critical process parameters for each stage.
- Esterification of R-pyroglutamic acid followed by N-Boc protection.
- Selective reduction with DIBAL-H and subsequent elimination to form the alkene intermediate.
- Stereoselective Simmons-Smith cyclopropanation using ZnEt2 and CH2I2 to establish the bicyclic core.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by decoupling the production of high-purity chiral intermediates from the volatility of natural product sourcing or complex enzymatic processes. The reliance on commodity chemicals such as thionyl chloride, di-tert-butyl dicarbonate, and zinc salts ensures a stable and predictable supply chain, mitigating the risks associated with single-source biological materials or exotic reagents. The elimination of chiral resolution steps, which typically consume half of the produced material and require expensive chiral stationary phases, results in a drastic reduction in raw material consumption and waste generation. This efficiency translates directly into lower manufacturing costs, allowing buyers to secure high-quality organocatalysts at a price point that makes their inclusion in commercial drug synthesis economically feasible. Furthermore, the robustness of the chemistry reduces the likelihood of batch failures, ensuring consistent delivery schedules and enhancing the overall reliability of the supply chain for downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for preparative chiral chromatography or enzymatic kinetic resolution, which are traditionally the most expensive steps in producing optically pure amino acid derivatives. By achieving high diastereoselectivity directly through chemical synthesis, the method maximizes the yield of the desired isomer from the starting material, effectively doubling the theoretical output compared to racemic synthesis followed by resolution. The use of standard solvents like dichloromethane, toluene, and ethanol further reduces operational expenses, as these are readily available in bulk quantities at competitive prices globally. Additionally, the avoidance of toxic organotin reagents eliminates the costly waste disposal and environmental compliance burdens associated with heavy metal contamination, contributing to a leaner and more sustainable cost structure.
- Enhanced Supply Chain Reliability: The synthetic pathway is constructed entirely from commercially available off-the-shelf reagents, ensuring that production is not held hostage by the lead times of custom-synthesized catalysts or fermentation-derived precursors. The linear nature of the seven-step sequence allows for flexible manufacturing strategies, where intermediates can be stockpiled at various stages to buffer against demand fluctuations or raw material shortages. This modularity enhances the resilience of the supply chain, enabling suppliers to respond rapidly to urgent requests from pharmaceutical clients without compromising on quality or purity. The stability of the Boc-protected intermediates also facilitates storage and transport, reducing the logistical complexities often associated with handling sensitive chiral amines.
- Scalability and Environmental Compliance: The reaction conditions employed, such as moderate temperatures and atmospheric pressure, are inherently scalable from kilogram to multi-tonne production without requiring specialized high-pressure or cryogenic infrastructure. The replacement of hazardous tin-based chemistry with zinc-mediated cyclopropanation aligns with increasingly strict global environmental regulations regarding heavy metal discharge and worker safety. This compliance advantage simplifies the permitting process for manufacturing facilities and reduces the long-term liability associated with environmental remediation. The ability to run this process in standard glass-lined or stainless steel reactors makes it accessible to a wide network of contract manufacturing organizations, fostering a competitive supply market that benefits the end purchaser.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cyclopropane-fused proline derivatives. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's potential. Understanding these details is crucial for R&D teams assessing the feasibility of incorporating these catalysts into their own synthetic routes and for procurement specialists evaluating the long-term viability of the supply source. The information covers aspects ranging from stereochemical purity to the practicalities of large-scale implementation.
Q: What is the key advantage of this synthesis over traditional methods?
A: The primary advantage is the exceptional diastereoselectivity (>25:1) achieved during the Simmons-Smith cyclopropanation step, which eliminates the need for costly and time-consuming chiral resolution or separation of isomers required in older tin-based methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard, scalable reagents such as thionyl chloride, Boc anhydride, and diisobutylaluminum hydride, avoiding exotic or highly toxic organotin compounds, making it highly amenable to commercial scale-up in standard stainless steel reactors.
Q: What represents the critical control point in the reaction sequence?
A: Temperature control during the DIBAL-H reduction (-50°C to -40°C) and the subsequent cyclopropanation (-20°C to -15°C) is critical to prevent over-reduction and ensure the formation of the desired (1R,3R,5R) stereoisomer with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropane R-Proline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organocatalysts play in accelerating the discovery and development of new therapeutic agents. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to pilot plant is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the stereochemical integrity of every batch. We understand that in the competitive landscape of pharmaceutical intermediates, consistency and reliability are just as important as price, and our robust quality management systems are designed to deliver exactly that level of assurance to our global partners.
We invite you to engage with our technical team to discuss how this innovative synthesis can be tailored to meet your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this route might optimize your overall manufacturing budget. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than speculation. Let us collaborate to bring your next generation of chiral drugs to market faster and more cost-effectively.
