Advanced Synthesis of Cyclopropane R-Proline Derivatives for High-Efficiency Organocatalysis
The landscape of asymmetric synthesis is undergoing a transformative shift, driven by the urgent demand for more efficient and environmentally benign chiral catalysts. Patent CN103450072A introduces a groundbreaking methodology for the preparation of pyrrole derivatives featuring a cyclopropane structure based on R-proline. This innovation addresses critical bottlenecks in organocatalysis by providing a robust synthetic route that eliminates the need for complex chiral resolution steps. For R&D Directors and technical decision-makers, this patent represents a significant leap forward in accessing high-purity chiral building blocks essential for modern drug discovery. The disclosed method leverages the inherent rigidity of the cyclopropane ring to enhance steric hindrance, thereby solving long-standing issues related to poor cis-selectivity and limited enantioselectivity found in conventional proline-derived catalysts. By streamlining the production of these sophisticated molecules, the technology offers a reliable pathway for scaling up the manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral proline derivatives has been plagued by inefficiencies that hinder large-scale commercial adoption. Prior art, such as the methods described by Barbas III and H. Stephen, often relied on natural products as chiral sources, which inherently limited structural diversity and catalytic versatility. A significant drawback in existing technologies is the reliance on expensive and toxic cyclization reagents, such as trimethylsulfonium salts, which not only drive up costs but also pose severe environmental and safety challenges during waste disposal. Furthermore, conventional routes frequently suffer from low enantiomeric excess, typically yielding product ratios around 4:1, which necessitates additional, costly purification and resolution steps to achieve the optical purity required for active pharmaceutical ingredients. These multi-step resolution processes result in substantial material loss and extended lead times, creating friction in the supply chain for critical fine chemical intermediates.
The Novel Approach
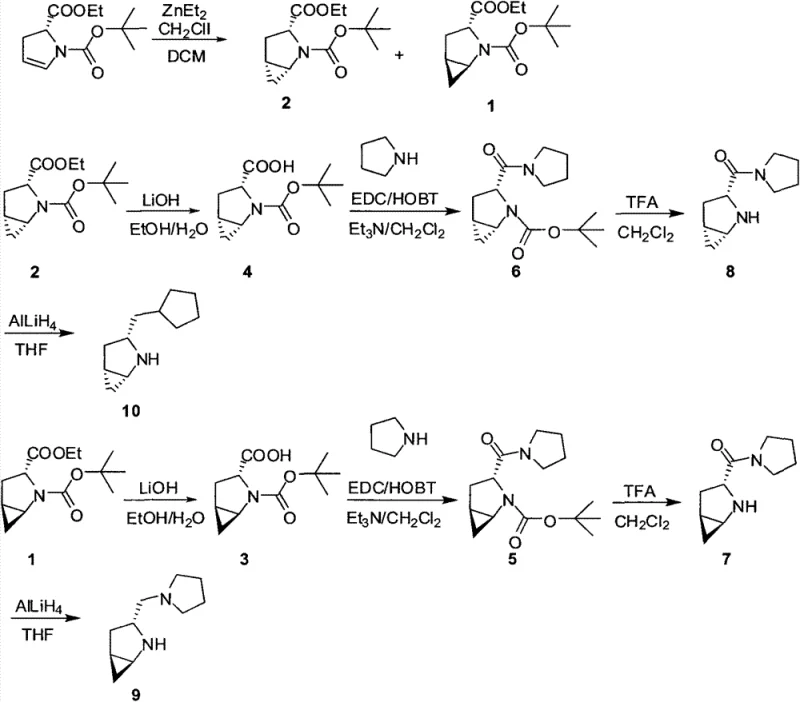
In stark contrast, the methodology outlined in CN103450072A presents a streamlined, five-step synthetic strategy that bypasses these traditional hurdles through the strategic application of the Simmons-Smith cyclopropanation reaction. This novel approach utilizes zinc ethyl and chloroiodomethane under controlled low-temperature conditions to construct the rigid cyclopropane framework with exceptional stereocontrol. The process achieves an impressive enantiomer ratio exceeding 25:1, effectively rendering downstream chiral separation unnecessary and drastically simplifying the overall workflow. By integrating ester hydrolysis, amide condensation, and selective reduction into a cohesive sequence, the method ensures high yields while maintaining operational simplicity. This technological advancement allows manufacturers to produce optically pure R-proline pyrrole derivatives with significantly reduced processing time and resource consumption.
Mechanistic Insights into Simmons-Smith Cyclopropanation and Catalytic Activity
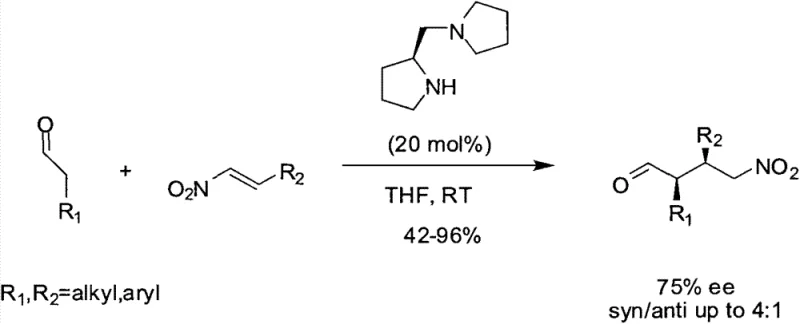
The core of this technological breakthrough lies in the precise manipulation of molecular geometry to enhance catalytic performance. The introduction of the cyclopropane ring at the ortho position of the pyrrole structure imparts a high degree of rigidity to the catalyst backbone. This structural constraint is pivotal because it restricts the conformational freedom of the molecule, thereby creating a well-defined chiral environment around the active catalytic site. When employed in asymmetric Michael addition reactions, this rigid framework effectively shields specific faces of the substrate, guiding the approach of electrophilic reagents with high precision. The result is a dramatic improvement in both enantioselectivity and diastereoselectivity, allowing for the synthesis of complex chiral molecules that were previously difficult to access with standard proline catalysts. This mechanistic advantage translates directly into higher quality outputs for pharmaceutical synthesis, reducing the burden on downstream purification processes.
Furthermore, the impurity profile of the final product is meticulously controlled through the optimization of reaction conditions and reagent stoichiometry. The use of specific aqueous workup procedures involving EDTA and amine solutions ensures the effective removal of zinc residues and other metal byproducts, which is critical for meeting stringent regulatory standards in drug manufacturing. The subsequent steps, including the use of EDCI and HOBT for amide coupling, are conducted under mild conditions that minimize the formation of side products and racemization. This attention to detail in the synthetic design ensures that the final R-proline derivative maintains its optical integrity throughout the process. For quality assurance teams, this means a more consistent and reliable supply of chiral catalysts that perform predictably in sensitive synthetic transformations.
How to Synthesize Cyclopropane R-Proline Derivative Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent addition rates to maximize the benefits of the Simmons-Smith reaction. The process begins with the preparation of the cyclopropane intermediate at temperatures between -20°C and -15°C, followed by a series of functional group transformations that preserve the chiral centers. Detailed standard operating procedures for each stage, including specific molar ratios and solvent systems, are essential for reproducing the high enantiomeric excess reported in the patent data. Manufacturers looking to adopt this technology should focus on maintaining anhydrous conditions during the organozinc steps and ensuring efficient mixing during the exothermic reduction phases. The following guide outlines the critical operational parameters required to achieve successful scale-up.
- Perform Simmons-Smith cyclopropane reaction on (R)-1-N-Boc-2,3-dihydro-2-pyrrolecarboxylic acid ethyl ester using ZnEt2 and CH2ClI at -20°C.
- Conduct ester hydrolysis using LiOH under alkaline conditions to obtain the corresponding carboxylic acid intermediates.
- Execute amide condensation with pyrrolidine using EDCI/HOBt coupling agents, followed by Boc deprotection with TFA and final reduction with LiAlH4.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this novel synthetic route offers compelling economic advantages that extend beyond simple raw material costs. By eliminating the need for chiral resolution columns and the associated solvents and consumables, the overall cost of goods sold is substantially reduced. The simplified process flow also means fewer unit operations are required, which lowers energy consumption and reduces the footprint needed for production. For supply chain managers, the robustness of this method translates into enhanced reliability, as the risk of batch failure due to poor selectivity is minimized. The ability to produce high-purity intermediates in fewer steps significantly shortens the manufacturing cycle time, allowing for faster response to market demands and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes a major cost driver associated with low-yield separation processes and expensive chromatography media. Additionally, the use of readily available reagents like zinc ethyl and chloroiodomethane, combined with high overall yields, ensures that the cost per kilogram of the final catalyst is optimized. This efficiency allows for competitive pricing strategies without compromising on the quality or purity specifications required by top-tier pharmaceutical clients.
- Enhanced Supply Chain Reliability: The streamlined nature of the five-step synthesis reduces the number of potential failure points in the production line, leading to more consistent batch-to-batch quality. The high enantioselectivity achieved directly from the reaction means that supply continuity is less dependent on variable purification outcomes. This stability is crucial for long-term supply agreements, ensuring that downstream customers receive their critical chiral building blocks on schedule without unexpected delays caused by reprocessing off-spec material.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard chemical engineering unit operations that can be easily transferred from pilot plant to commercial scale. Furthermore, the avoidance of toxic heavy metal catalysts and the implementation of efficient aqueous workups align with green chemistry principles, reducing the environmental burden and simplifying waste treatment compliance. This makes the technology attractive for companies aiming to meet increasingly strict sustainability goals while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this patented technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this catalyst into their existing synthetic workflows. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the technology's capabilities and limitations.
Q: How does this new synthesis route improve enantioselectivity compared to prior art?
A: The novel method utilizes a rigid cyclopropane structure introduced via an optimized Simmons-Smith reaction, achieving an enantiomer ratio greater than 25:1, significantly surpassing the 4:1 ratio reported in previous literature.
Q: Does this process require chiral resolution steps?
A: No, the process is designed to produce optically pure enantiomer products directly without the need for further chiral preparation or resolution, thereby simplifying the workflow and reducing material loss.
Q: What are the primary applications of this R-proline derivative?
A: This compound serves as a highly effective chiral organocatalyst for asymmetric Michael addition reactions, offering broad-spectrum stereoselectivity for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Proline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-performance chiral catalysts in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our expertise in handling sensitive organometallic reactions and asymmetric synthesis allows us to guarantee the consistent quality and optical purity of every batch of R-proline derivatives we supply to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you leverage this advanced synthetic technology to accelerate your drug development timelines and secure a competitive edge in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
