Revolutionizing Fluorine-Containing Aryl Compound Synthesis with Solvent-Free Technology
The chemical industry is constantly seeking more efficient and sustainable methods for synthesizing fluorine-containing aryl compounds, which serve as critical building blocks in the development of advanced pharmaceuticals and agrochemicals. Patent CN112939782B discloses a groundbreaking preparation method that addresses the long-standing challenges associated with traditional halogen exchange fluorination reactions. This innovation utilizes a solvent-free reaction system combined with high-boiling point phase transfer catalysts, specifically crown ethers, to achieve superior reaction control and product quality. By eliminating the need for conventional high-boiling solvents such as DMF or DMSO, the process avoids the complex separation issues that often plague industrial fluorination, resulting in significantly improved distillation yields and product purity. The technical breakthrough lies in the ability of the catalyst to function effectively as both a phase transfer agent and a reaction medium, facilitating the interaction between solid alkali metal fluorides and organic aryl halides without external solvent assistance. This approach not only streamlines the post-reaction workup but also enhances the overall economic viability of producing high-value fluorinated intermediates for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for preparing fluorine-containing aromatic compounds often rely on halogen exchange fluorination using alkali metal fluorides in the presence of high-boiling polar aprotic solvents. Solvents such as dimethylformamide (DMF), dimethyl sulfoxide (DMSO), N-methyl-2-pyrrolidone (NMP), and sulfolane are commonly employed to dissolve the reactants and facilitate the reaction kinetics. However, these solvents present significant downstream processing challenges because their boiling points are often very close to that of the target fluorinated products. This similarity in volatility makes it extremely difficult to separate the product from the solvent during rectification, leading to co-distillation and contamination that compromises the final purity of the compound. Furthermore, the recovery and reuse of these expensive solvents are technically difficult and energy-intensive, often requiring complex vacuum distillation setups that increase operational costs. In many cases, the solvent becomes contaminated with byproducts or degradation products, rendering it unsuitable for reuse and generating substantial hazardous waste that requires specialized treatment. The accumulation of tar and inorganic salts in the reaction residue further complicates the disposal process, creating environmental compliance burdens for manufacturing facilities.
The Novel Approach
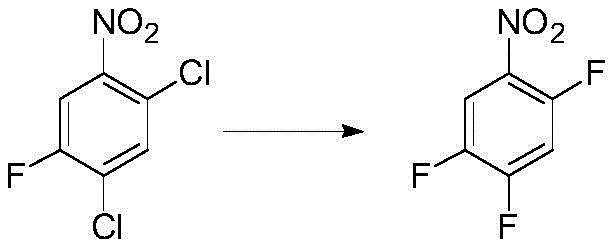
The novel approach described in the patent overcomes these limitations by implementing a solvent-free reaction system that relies on the unique properties of specific phase transfer catalysts. In this method, the reaction is carried out by reacting an aryl halide compound of formula 2 with an alkali metal fluoride in the presence of a crown ether catalyst without any additional organic solvent. The phase transfer catalyst, such as 18-crown-6 or 15-crown-5, possesses a relatively high boiling point that is distinct from the target product, ensuring that it remains in the reaction residue during the final distillation step. This physical property difference allows for a clean separation where the target fluorine-containing aryl compound of formula 1 can be distilled off with high purity, free from solvent interference. The absence of bulk solvent simplifies the reaction mixture, reducing the volume of material that needs to be heated and processed, which in turn lowers energy consumption and improves throughput. Additionally, the catalyst can be recycled directly from the pot residue for multiple reaction cycles or recovered via short distillation, significantly reducing the consumption of auxiliary materials. This streamlined process design translates directly into operational efficiency and cost savings for large-scale manufacturing operations.
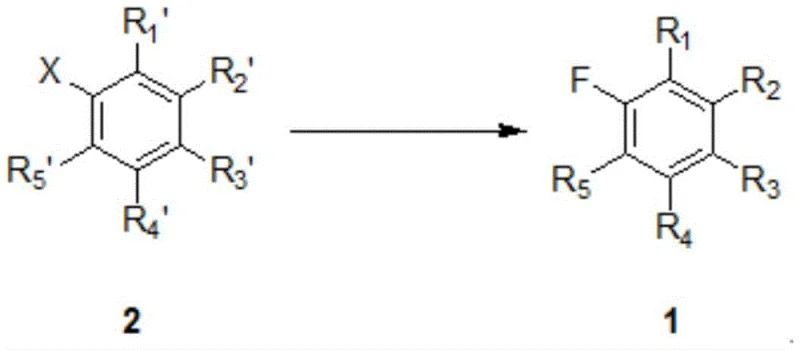
Mechanistic Insights into Crown Ether-Catalyzed Halogen Exchange Fluorination
The core mechanism of this synthesis involves the activation of the alkali metal fluoride by the crown ether phase transfer catalyst under anhydrous conditions. Crown ethers are cyclic polyethers that have a cavity size capable of complexing with specific metal cations, such as potassium or sodium ions from the fluoride salt. By encapsulating the cation, the crown ether effectively solubilizes the fluoride anion in the organic phase, increasing its nucleophilicity and reactivity towards the aryl halide substrate. This phase transfer process allows the reaction to proceed efficiently even in the absence of a traditional solvent, as the catalyst-reactant complex forms a liquid phase that facilitates molecular collision and reaction progression. The reaction temperature is typically maintained between 70°C and 180°C, which is sufficient to melt the catalyst and drive the fluorination forward while maintaining the thermal stability of the system. The anhydrous environment is critical to prevent the hydrolysis of the fluoride source and to avoid side reactions that could generate hydroxylated byproducts, ensuring that the halogen exchange proceeds selectively to form the carbon-fluorine bond. This mechanistic understanding allows for precise control over reaction parameters to optimize yield and minimize the formation of impurities.
Impurity control in this process is achieved through the careful selection of reaction conditions and the inherent cleanliness of the solvent-free system. Since there are no high-boiling solvents to co-distill with the product, the primary impurities are limited to unreacted starting materials and isomeric byproducts, which can be effectively separated during the rectification step. The solid byproduct generated is primarily an alkali metal halide, such as potassium chloride or sodium chloride, which is separated via filtration or pulping before distillation. This solid waste is significantly cleaner than the tar-heavy residues produced in conventional solvent-based methods, making it easier to handle and potentially recyclable by specialized waste management companies. The thermal stability of the crown ether catalyst ensures that it does not degrade significantly at the reaction temperatures, preventing the formation of catalyst-derived impurities that could contaminate the product stream. Furthermore, the ability to recycle the catalyst residue multiple times without significant loss of activity demonstrates the robustness of the system against impurity buildup. This high level of control over the reaction environment results in products with HPLC purities often exceeding 98%, meeting the stringent requirements of downstream pharmaceutical and agrochemical applications.
How to Synthesize Fluorine-Containing Aryl Compounds Efficiently
The synthesis of these valuable intermediates follows a streamlined protocol that emphasizes operational simplicity and material efficiency. The process begins with the charging of the aryl halide substrate, anhydrous alkali metal fluoride, and the crown ether phase transfer catalyst into a reaction vessel under inert atmosphere. The mixture is then heated to the specified temperature range to initiate the fluorination reaction, which proceeds without the need for solvent addition or complex mixing protocols. Detailed standardized synthesis steps see the guide below.
- Mix aryl halide substrate with alkali metal fluoride and a crown ether phase transfer catalyst under anhydrous conditions.
- Heat the reaction mixture to 70-180°C to facilitate halogen exchange fluorination without additional solvents.
- Perform solid-liquid separation, purify the liquid phase via distillation, and recycle the catalyst residue for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free fluorination technology offers substantial strategic advantages in terms of cost structure and supply reliability. The elimination of expensive high-boiling solvents removes a significant variable cost from the manufacturing process, as there is no longer a need to purchase, recover, or dispose of large volumes of DMF, DMSO, or NMP. This reduction in auxiliary material consumption directly lowers the bill of materials for each batch, contributing to a more competitive pricing structure for the final fluorinated intermediates. Furthermore, the simplified workup procedure reduces the processing time and energy required for solvent recovery and product purification, allowing for faster turnaround times and higher production capacity within existing infrastructure. The ability to recycle the phase transfer catalyst multiple times further amplifies these cost savings by minimizing the consumption of this specialized reagent. From a supply chain perspective, the reduced dependency on solvent supply chains mitigates the risk of disruptions caused by raw material shortages or price volatility in the solvent market. The generation of cleaner solid waste also simplifies environmental compliance and reduces the costs associated with hazardous waste disposal, enhancing the overall sustainability profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The removal of high-boiling solvents from the reaction system eliminates the need for complex and energy-intensive solvent recovery processes, leading to direct operational cost savings. By avoiding the use of solvents that are difficult to separate from the product, the process reduces product loss during purification and improves the overall mass balance of the reaction. The recyclability of the crown ether catalyst means that the effective cost per kilogram of catalyst used is drastically reduced over multiple production cycles. These factors combine to create a manufacturing process with a significantly lower cost base compared to traditional solvent-based fluorination methods. The reduction in waste treatment costs due to cleaner byproducts further contributes to the overall economic efficiency of the process.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of critical raw materials required for production, thereby decreasing the complexity of the supply chain and the potential points of failure. With fewer solvents to source and manage, the procurement team can focus on securing high-quality aryl halide substrates and fluoride salts, which are generally more stable and available commodities. The robustness of the reaction conditions allows for consistent production output, minimizing the risk of batch failures that could disrupt supply to downstream customers. Additionally, the ability to scale the process from laboratory to commercial production without significant re-engineering ensures that supply can be ramped up quickly to meet market demand. This reliability is crucial for maintaining long-term partnerships with pharmaceutical and agrochemical companies that require consistent quality and delivery performance.
- Scalability and Environmental Compliance: The solvent-free nature of the reaction makes it inherently safer and easier to scale, as there are no large volumes of flammable or toxic solvents to manage in large reactors. The reduced generation of hazardous liquid waste simplifies the environmental permitting process and lowers the ongoing costs of waste management and compliance reporting. The solid byproducts generated are primarily inorganic salts, which are easier to handle and dispose of compared to the complex organic waste streams from conventional methods. This alignment with green chemistry principles enhances the corporate social responsibility profile of the manufacturer and meets the increasing demand for sustainable chemical production. The process is designed to be compatible with standard chemical processing equipment, facilitating easy integration into existing manufacturing facilities without the need for specialized solvent-handling infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived from the specific technical disclosures and experimental data provided in the patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders evaluate the feasibility and benefits of adopting this method for their specific production needs. The information covers aspects of reaction mechanism, process optimization, and waste management to provide a comprehensive overview.
Q: How does the solvent-free method improve product purity compared to traditional methods?
A: Traditional methods use high-boiling solvents like DMF or DMSO which are difficult to separate from the product during distillation. This solvent-free method eliminates solvent interference, allowing for cleaner distillation and significantly higher product purity.
Q: Can the phase transfer catalyst be recycled in this process?
A: Yes, the crown ether catalyst remains in the pot residue after distillation due to its high boiling point. It can be directly reused in subsequent fluorination reactions multiple times or recovered via short distillation, reducing raw material costs.
Q: What are the environmental benefits of this fluorination technique?
A: By eliminating organic solvents and generating clean alkali metal halide byproducts instead of tar-heavy waste, this method significantly reduces three-waste output and simplifies waste treatment compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Aryl Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced technologies like the solvent-free fluorination process to deliver high-quality intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorine-containing aryl compound meets the exacting standards required by the pharmaceutical and agrochemical industries. Our commitment to process excellence allows us to offer products with consistent quality and reliability, supporting our partners in their own drug development and manufacturing pipelines. By integrating cutting-edge patent technologies into our production capabilities, we continue to drive value and efficiency for our customers.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain requirements. Our team is ready to discuss how this solvent-free technology can optimize your raw material costs and improve your production efficiency. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. We are committed to providing transparent technical support and collaborative solutions to help you achieve your strategic sourcing goals. Let us partner with you to build a more efficient and sustainable supply chain for fluorinated chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
