Advanced Synthetic Route for Nilotinib Intermediate Enhancing Commercial Scalability and Purity
The global demand for tyrosine kinase inhibitors continues to surge as targeted cancer therapies become the standard of care for chronic myeloid leukemia (CML). At the heart of this therapeutic class lies Nilotinib, a potent second-generation inhibitor marketed as Tasigna, which requires highly specialized chemical building blocks for its manufacture. A critical breakthrough in the supply chain for this life-saving medication is documented in Chinese Patent CN103694176A, which discloses a novel preparation method for the key intermediate 3-(4-methyl-1H-imidazolyl)-5-trifluoromethyl phenylamine. This technical insight report analyzes the proprietary synthetic strategy outlined in the patent, focusing on how reordering reaction sequences can fundamentally solve long-standing yield and purity issues. By shifting from traditional reduction-first pathways to a condensation-first approach, manufacturers can achieve superior impurity profiles and operational efficiency. The structural relationship between the target intermediate (I) and the downstream acid intermediate (II) is illustrated below, highlighting the precise molecular architecture required for effective kinase inhibition.
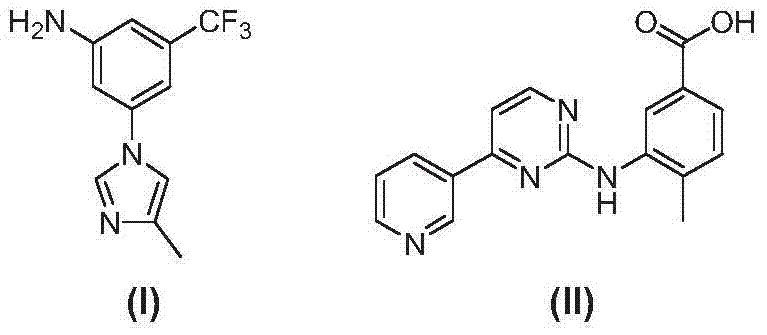
For procurement leaders and supply chain directors, understanding the nuances of this patent is not merely an academic exercise but a strategic imperative for securing reliable API intermediate supplier partnerships. The ability to produce this specific trifluoromethyl-substituted aniline derivative with high consistency directly impacts the cost of goods sold (COGS) and the continuity of drug supply. As we delve into the technical specifics, it becomes evident that the innovations protected under CN103694176A offer a viable pathway to mitigate the risks associated with complex heterocyclic synthesis. This report serves as a comprehensive guide for R&D Directors evaluating process feasibility and Procurement Managers seeking cost reduction in pharmaceutical manufacturing through intelligent route selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
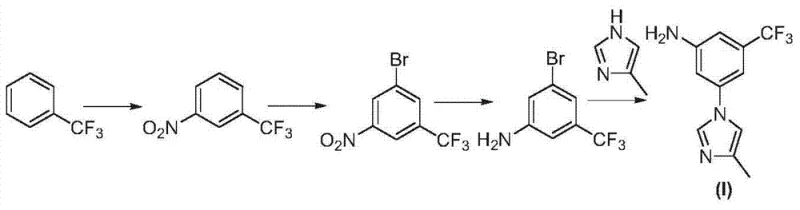
Historical approaches to synthesizing this critical nilotinib precursor have been plagued by significant chemical inefficiencies that hinder large-scale commercialization. Prior art methods, such as those described in WO2004/00528 and WO2006/135640, often rely on starting materials that are either scarce or require multi-step preparation themselves, introducing unnecessary supply chain vulnerabilities. A particularly common conventional strategy involves reducing the nitro group to an amine early in the synthesis, followed by a nucleophilic substitution to install the imidazole ring. While conceptually straightforward, this reduction-first sequence creates a highly reactive amino group that is prone to undesirable self-coupling reactions with the bromine functionality on the aromatic ring. Literature and experimental data indicate that this side reaction can generate coupled impurities in amounts exceeding 50%, drastically reducing the yield of the desired product and complicating downstream purification. Furthermore, alternative routes involving Curtius rearrangements or multiple nitration steps add layers of operational complexity and safety hazards, such as the handling of unstable acyl azides or the management of exothermic nitration events. These legacy processes, depicted in various prior art schemes, often suffer from low atom economy and generate substantial chemical waste, making them economically unviable for modern, environmentally conscious manufacturing facilities.
The Novel Approach
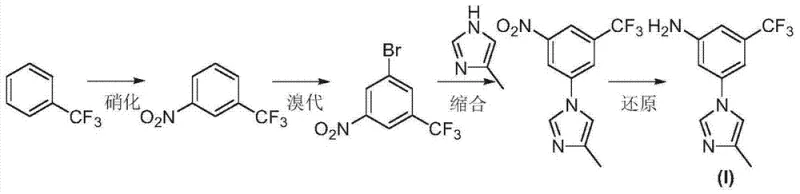
In stark contrast to these problematic legacy methods, the technology disclosed in CN103694176A introduces a paradigm shift by reversing the order of key transformations to prioritize chemoselectivity. The core innovation lies in performing the condensation reaction between the aryl halide and the imidazole moiety while the nitro group is still present on the benzene ring. This strategic sequencing leverages the electron-withdrawing nature of the nitro group to activate the aromatic ring towards nucleophilic aromatic substitution, thereby facilitating the coupling reaction without the need for harsh conditions that might degrade sensitive functionalities. Crucially, by delaying the reduction of the nitro group until after the imidazole ring is securely attached, the process completely eliminates the risk of amino-bromo self-coupling that plagues the conventional routes. The new pathway utilizes trifluorotoluene, a widely available and cost-effective commodity chemical, as the starting material, subjecting it to a logical sequence of nitration, bromination, condensation, and finally reduction. This streamlined four-step sequence not only simplifies the operational workflow but also significantly enhances the overall yield and purity of the final intermediate. The visual representation of this optimized route clearly demonstrates the linear progression from simple feedstocks to the complex target molecule, underscoring its suitability for industrial scale-up.
Mechanistic Insights into CuI-Catalyzed Condensation
The success of this novel synthetic route hinges on the meticulous optimization of the condensation step, where the aryl bromide reacts with 4-methylimidazole. In traditional nucleophilic aromatic substitutions, the presence of a nitro group ortho or para to the leaving group is beneficial; however, the reaction conditions must be carefully tuned to overcome the inherent deactivation of the ring and ensure complete conversion. The patent specifies the use of a copper-based catalytic system, specifically cuprous iodide (CuI), which acts as a powerful mediator for the C-N bond formation. This transition metal catalyst likely operates through a coordination mechanism that lowers the activation energy for the displacement of the bromine atom by the nitrogen of the imidazole ring. To further enhance the reaction kinetics, the process incorporates specific promoters such as 8-hydroxyquinoline or rigid bicyclic amines like DBU. These additives serve to stabilize the catalytic species and scavenge protons generated during the reaction, driving the equilibrium towards the product. The choice of solvent, preferably polar aprotic solvents like DMF or DMSO, is also critical to solubilize the inorganic bases and facilitate the interaction between the organic substrates and the catalyst. This sophisticated catalytic manifold ensures that the condensation proceeds with high selectivity, minimizing the formation of regioisomers or homocoupled byproducts that would otherwise compromise the quality of the intermediate.
Beyond the primary coupling event, the control of impurities is a paramount concern for R&D Directors responsible for regulatory filings and quality assurance. The mechanistic design of this process inherently suppresses the formation of the dreaded self-coupled dimers that arise from amino-bromo interactions in other routes. By maintaining the nitro group throughout the condensation phase, the aromatic ring remains sufficiently electron-deficient to prevent unwanted electrophilic attacks on the amine, which does not yet exist. Furthermore, the subsequent reduction step, whether performed using hydrazine hydrate with iron salts or via catalytic hydrogenation with palladium on carbon, is conducted under mild conditions that preserve the integrity of the newly formed imidazole linkage. The patent data reports impressive yields for individual steps, such as 91.8% for the bromination and over 90% for the final reduction, indicating a robust and forgiving process window. This high level of control translates directly to a cleaner crude product, reducing the burden on purification units and lowering the consumption of chromatography media or recrystallization solvents. For a commercial manufacturer, this means a more predictable process with fewer batch failures and a consistent supply of high-purity material suitable for GMP production of the final active pharmaceutical ingredient.
How to Synthesize 3-(4-methyl-1H-imidazolyl)-5-trifluoromethyl phenylamine Efficiently
Implementing this advanced synthetic methodology requires a disciplined approach to process parameters and reagent quality to fully realize the benefits outlined in the patent literature. The following guide summarizes the critical operational phases derived from the experimental examples, providing a framework for technical teams to evaluate the feasibility of adoption within their existing infrastructure. It is essential to note that while the general sequence is robust, specific conditions such as temperature ramps and addition rates may require fine-tuning based on the specific reactor geometry and scale of operation. The detailed standardized synthesis steps see the guide below.
- Initiate the process by nitrating trifluorotoluene to form m-nitrobenzotrifluoride under controlled low-temperature conditions.
- Perform bromination on the nitro-intermediate to generate 3-bromo-5-nitrobenzotrifluoride using bromine and sulfuric acid.
- Execute the key condensation reaction with 4-methylimidazole using a CuI catalyst system before reducing the nitro group to avoid side reactions.
- Complete the synthesis by reducing the nitro group to an amine using hydrazine hydrate or catalytic hydrogenation to yield the final target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical superiority of this synthetic route translates into tangible business value that extends far beyond the laboratory bench. The shift to a condensation-first strategy fundamentally alters the economic model of producing this nilotinib intermediate by addressing the root causes of yield loss and process instability found in older methods. By eliminating the formation of high levels of self-coupled impurities, the process reduces the need for extensive rework or discard of off-spec batches, leading to a more predictable and reliable supply stream. This reliability is crucial for maintaining the continuity of API production schedules, preventing costly downtime that can ripple through the entire pharmaceutical supply network. Furthermore, the use of trifluorotoluene as a starting material leverages a well-established global supply chain for fluorinated chemicals, mitigating the risk of raw material shortages that often plague specialty syntheses relying on exotic precursors. The overall simplification of the process flow also implies a reduced footprint for manufacturing, as fewer unit operations and isolation steps are required to reach the final intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the significant improvement in overall yield and the elimination of expensive purification steps required to remove coupling impurities. By avoiding the use of complex protecting groups or hazardous rearrangement reagents found in prior art, the direct material costs are substantially lowered. The high efficiency of the copper-catalyzed condensation ensures that valuable starting materials are converted into product rather than waste, maximizing the return on investment for every kilogram of input. Additionally, the ability to use standard industrial solvents and readily available catalysts further contributes to a leaner cost structure, allowing for competitive pricing in the generic drug market without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commodity chemicals like trifluorotoluene and common inorganic reagents ensures a stable and resilient supply base. Unlike routes that depend on custom-synthesized building blocks with long lead times, this method allows for rapid scaling of production in response to market demand fluctuations. The robustness of the chemistry means that production can be distributed across multiple manufacturing sites with minimal technology transfer friction, diversifying supply risk. This flexibility is invaluable for pharmaceutical companies seeking to dual-source critical intermediates to safeguard against geopolitical disruptions or single-vendor bottlenecks, ensuring that patient access to medication is never compromised by upstream supply issues.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, featuring reaction conditions that are manageable in large-scale reactors without requiring extreme pressures or cryogenic temperatures. The reduction in side reactions directly correlates to a lower environmental burden, as less chemical waste is generated per unit of product. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals, reducing the costs associated with waste treatment and disposal. The high atom economy of the sequence means that the process is not only commercially viable but also environmentally responsible, positioning the manufacturer as a leader in green chemistry practices within the fine chemical sector.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent technology, we have compiled answers to the most pressing questions regarding its implementation and advantages. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their supply chain.
Q: Why is the condensation step performed before reduction in this new route?
A: Performing condensation before reduction prevents the self-coupling side reactions that typically occur when an amino group is present during nucleophilic substitution, significantly improving yield and purity.
Q: What catalysts are recommended for the condensation reaction?
A: The patent specifies cuprous iodide (CuI) as the preferred catalyst, often used in conjunction with promoters like 8-hydroxyquinoline to overcome the deactivating effect of the nitro group.
Q: How does this method improve industrial scalability compared to prior art?
A: By utilizing readily available starting materials like trifluorotoluene and eliminating complex protection-deprotection steps, the process reduces operational complexity and enhances atom economy for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-methyl-1H-imidazolyl)-5-trifluoromethyl phenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team of process chemists has extensively analyzed the route disclosed in CN103694176A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this optimized synthesis to life. We understand that achieving the theoretical yields described in the lab requires precise control over reaction parameters, and our rigorous QC labs are equipped to monitor every critical quality attribute, ensuring stringent purity specifications are met for every batch. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your API manufacturing workflow, reducing your time to market and regulatory risk.
We invite you to engage with our technical procurement team to discuss how this advanced intermediate can enhance your product portfolio and drive efficiency in your operations. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this superior synthetic route. We encourage you to contact us to obtain specific COA data from our pilot runs and to request comprehensive route feasibility assessments tailored to your specific volume requirements. Let us collaborate to build a more resilient and cost-effective supply chain for next-generation oncology therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
