Scalable Synthesis of Optically Active Dioxan Acetic Acid Derivatives for Statin Production
Scalable Synthesis of Optically Active Dioxan Acetic Acid Derivatives for Statin Production
The pharmaceutical industry continuously seeks robust and cost-effective pathways for the production of critical intermediates, particularly those serving as the backbone for HMG-CoA reductase inhibitors, commonly known as statins. Patent CN1162422C, granted in 2004, introduces a groundbreaking methodology for preparing optically active 2-[6-(hydroxymethyl)-1,3-dioxan-4-yl]acetic acid derivatives. This specific chemical architecture is pivotal in the synthesis of cholesterol-lowering agents, representing a high-value segment within the fine chemical market. The innovation lies in its ability to bypass the stringent and costly requirements of traditional synthetic routes, such as ultra-low temperature reactions and high-pressure hydrogenation. By leveraging a combination of mild chemical conditions and biocatalytic steps, this process offers a compelling value proposition for manufacturers aiming to optimize their production lines. The target molecule, characterized by its specific dioxan ring structure and optical activity, is essential for ensuring the efficacy and safety of the final pharmaceutical product.
![Chemical structure of optically active 2-[6-(hydroxymethyl)-1,3-dioxan-4-yl]acetic acid derivative (Formula I)](/insights/img/optically-active-dioxan-acetic-acid-pharma-supplier-20260315041109-01.png)
For procurement specialists and supply chain directors, the implications of this technology extend far beyond the laboratory. The ability to produce such complex chiral intermediates without specialized cryogenic infrastructure translates directly into reduced capital expenditure and lower operational risks. Traditional methods often rely on hazardous reagents and energy-intensive cooling systems, which can create bottlenecks in large-scale production. In contrast, the methodology described in CN1162422C utilizes readily available raw materials and standard reaction conditions, facilitating a smoother transition from pilot scale to commercial manufacturing. This reliability is crucial for maintaining the continuity of supply for downstream pharmaceutical clients who demand consistent quality and timely delivery. As a reliable pharmaceutical intermediate supplier, understanding these technological nuances allows us to offer partners a more resilient and cost-efficient sourcing strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing 2-(6-methylol-1,3-diox-4-yl) acetogenin derivatives have historically been plagued by significant technical and economic hurdles. Many established routes require reaction temperatures as low as -80°C, necessitating the use of specialized equipment capable of maintaining such extreme cryogenic conditions. This not only increases the initial investment in plant infrastructure but also escalates ongoing energy costs. Furthermore, conventional processes often depend on expensive and sensitive reagents, such as 1,6-hexylidene bis-lithium amides and diethyl methoxy borane, which add to the raw material costs and complicate waste management. Some methods also involve high-pressure hydrogenation steps, introducing additional safety risks and requiring robust containment systems. These factors collectively render traditional synthesis routes less suitable for efficient industrial application, particularly in a market environment where cost reduction in pharmaceutical intermediate manufacturing is a primary driver of competitiveness.
The Novel Approach
The novel approach detailed in the patent data presents a paradigm shift by eliminating the dependency on ultra-low temperature reactions and expensive chiral reagents. By operating at temperatures not less than -30°C, the process significantly lowers the thermal energy requirements and allows for the use of standard industrial reactors. The integration of a microbial reduction step replaces complex chemical stereoselective reductions, offering a more environmentally friendly and cost-effective alternative. This biocatalytic step achieves high optical purity without the need for precious metal catalysts or intricate chiral auxiliaries. Additionally, the subsequent chemical steps, including acetalization and acyloxylation, are designed to proceed under mild conditions using common acid catalysts and oxidizing reagents. This streamlined workflow reduces the number of purification steps required, thereby improving overall yield and minimizing solvent consumption. For supply chain heads, this translates to a more predictable production schedule and reduced lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Microbial Reduction and Acyloxylation
The core of this synthetic innovation lies in its multi-step mechanism, which cleverly combines chemical and biological transformations to achieve the desired stereochemistry. The process begins with the formation of an enolate from an acetic acid derivative using alkali or 0-valent metals, which then reacts with a hydroxybutyric acid ester derivative. This step is critical for establishing the carbon skeleton of the molecule. Following this, the key stereoselective reduction is performed using specific microorganism strains, such as those from the Candida or Cryptococcus genera. These microbes act as biocatalysts, reducing the keto group to a hydroxyl group with high diastereoselectivity, ensuring the formation of the (3R, 5S) configuration. This biological step is advantageous as it occurs under ambient pressure and moderate temperatures, avoiding the harsh conditions associated with chemical hydride reductions. The resulting dihydroxy compound is then protected via acetalization, forming the stable dioxan ring structure that is characteristic of the final product.
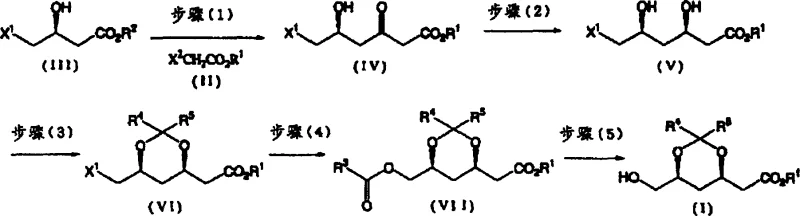
Following the formation of the dioxan ring, the process involves an acyloxylation step where a halomethyl group is converted into an acyloxymethyl group. This transformation is facilitated by the use of carboxylic acid quaternary ammonium salts or a mixture of quaternary ammonium salts and carboxylate salts. This specific reagent system allows for the substitution reaction to proceed efficiently without the need for expensive phase transfer catalysts or aggressive conditions. The final step involves solvolysis in the presence of a base, which removes the acyl protecting group to reveal the free hydroxymethyl functionality, yielding the target optically active 2-[6-(hydroxymethyl)-1,3-dioxan-4-yl]acetic acid derivative. Throughout this sequence, the control of impurities is paramount. The use of microbial reduction minimizes the formation of unwanted stereoisomers, while the mild chemical conditions reduce the risk of side reactions such as elimination or over-oxidation. This rigorous control over the reaction pathway ensures that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Optically Active Dioxan Acetic Acid Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to maximize yield and optical purity. The process is divided into five distinct stages, each optimized for industrial feasibility. The initial enolate formation must be carefully managed to prevent self-condensation, utilizing specific solvents like glycol dimethyl ether to enhance reactivity. The microbial reduction step demands the selection of appropriate strains and culture conditions to ensure high conversion rates and stereoselectivity. Subsequent chemical transformations, including acetalization and acyloxylation, are conducted under controlled temperatures to maintain the integrity of the chiral centers. The detailed standardized synthesis steps see the guide below for specific operational parameters and reagent ratios.
- React an acetic acid derivative with an enolate prepared using alkali or 0-valent metal at temperatures not less than -30°C to obtain a hydroxyketocaproic acid derivative.
- Reduce the obtained hydroxyketocaproic acid derivative using specific microorganism strains to achieve high stereoselectivity and obtain a dihydroxyacetic acid derivative.
- Treat the dihydroxy compound with an acetalizing agent in the presence of an acid catalyst, followed by acyloxylation and final solvolysis to yield the target optically active derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patent technology offers substantial benefits for procurement managers and supply chain leaders. The primary advantage is the significant reduction in manufacturing costs driven by the elimination of cryogenic infrastructure. By avoiding the need for -80°C reactors, companies can utilize existing standard equipment, thereby lowering capital expenditure and maintenance costs. Furthermore, the use of inexpensive raw materials and common reagents reduces the overall cost of goods sold. The microbial reduction step not only enhances stereoselectivity but also simplifies the purification process, leading to higher overall yields and less waste generation. These efficiencies contribute to a more competitive pricing structure, allowing suppliers to offer better value to their pharmaceutical clients without compromising on quality. The process is designed to be robust and scalable, ensuring that production can be ramped up to meet market demand without significant technical barriers.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive cryogenic equipment and specialized reagents, leading to substantial cost savings in both capital investment and operational expenses. By utilizing mild reaction conditions and readily available raw materials, the overall production cost is significantly lowered. The high efficiency of the microbial reduction step minimizes waste and reduces the need for extensive purification, further contributing to cost optimization. This economic advantage allows for more flexible pricing strategies and improved profit margins in a competitive market.
- Enhanced Supply Chain Reliability: The reliance on common chemicals and standard equipment reduces the risk of supply chain disruptions associated with specialized reagents or custom machinery. The scalability of the microbial fermentation process ensures a consistent and reliable supply of the intermediate, even during periods of high demand. Additionally, the simplified workflow reduces the lead time for production, enabling faster response to customer orders. This reliability is crucial for maintaining long-term partnerships with pharmaceutical companies that require uninterrupted supply for their drug manufacturing processes.
- Scalability and Environmental Compliance: The process is inherently scalable, allowing for seamless transition from laboratory scale to commercial production volumes. The use of biocatalysts and mild chemical conditions aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. Lower energy consumption and reduced waste generation facilitate compliance with increasingly stringent environmental regulations. This sustainability aspect not only mitigates regulatory risks but also enhances the corporate image of the manufacturer as a responsible and forward-thinking partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of optically active dioxan acetic acid derivatives. These answers are derived from the detailed technical specifications and beneficial effects outlined in the patent data. They provide clarity on the process advantages, scalability, and quality control measures that define this manufacturing route. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this technology into their supply chain.
Q: How does this patent process improve upon conventional cryogenic methods?
A: The patent eliminates the need for ultra-low temperature reactions (e.g., -80°C) and expensive reagents like 1,6-hexylidene bis-lithium amides, significantly reducing equipment costs and operational complexity.
Q: What is the role of microbial strains in this synthesis?
A: Microorganism strains are used for the stereoselective reduction of the hydroxyketocaproic acid derivative, providing high optical purity without the need for chiral chemical catalysts.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids special equipment and uses inexpensive raw materials, making it highly suitable for industrial scale-up and continuous supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Dioxan Acetic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging the innovative processes described in patents like CN1162422C, we can offer cost-effective solutions that do not compromise on quality or reliability. Our expertise in chiral synthesis and biocatalysis positions us as a strategic partner for pharmaceutical companies seeking to optimize their supply chains.
We invite you to engage with our technical procurement team to discuss how we can support your specific requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to our optimized manufacturing routes. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Our goal is to establish a long-term partnership based on trust, quality, and mutual success, ensuring that your drug development projects proceed without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
