Advanced Microbial Reduction Technology for High-Purity Chiral Alcohol Intermediates
Advanced Microbial Reduction Technology for High-Purity Chiral Alcohol Intermediates
The pharmaceutical industry continuously seeks robust and scalable methods for producing chiral intermediates with exceptional optical purity, particularly for complex therapeutic areas such as neurodegenerative disorders. Patent CN1498273A introduces a groundbreaking stereoselective process for the preparation of (S)-1-arylethanols through the microbial reduction of corresponding keto group-containing compounds. This technology represents a significant leap forward in biocatalysis, offering a reliable pathway to synthesize key intermediates used in the production of gamma-secretase inhibitors for the treatment of Alzheimer's disease. By leveraging specific oxidoreductase enzymes found in selected microorganisms, this method achieves high enantiomeric excess and reaction yields under mild conditions. The implications for commercial manufacturing are profound, as it addresses critical challenges related to stereocontrol and impurity management that often plague traditional chemical synthesis routes. This report analyzes the technical merits and commercial viability of this patented approach for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical methods for reducing substituted acetophenones to chiral alcohols often rely on stoichiometric chiral reducing agents or transition metal catalysts, which present significant drawbacks for large-scale pharmaceutical manufacturing. These conventional routes frequently require harsh reaction conditions, including extreme temperatures and pressures, which can compromise the stability of sensitive functional groups on the aromatic ring. Furthermore, achieving high enantiomeric purity typically necessitates complex resolution steps or the use of expensive chiral auxiliaries, leading to substantial material loss and increased waste generation. The presence of heavy metal residues from catalysts also poses severe regulatory hurdles, requiring rigorous and costly purification processes to meet stringent pharmaceutical quality standards. Additionally, the scalability of these chemical processes is often limited by safety concerns associated with reactive reagents and the difficulty in controlling exothermic reactions during batch operations.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes whole-cell microorganisms or isolated oxidoreductases to catalyze the stereoselective reduction of ketones under ambient conditions. This biocatalytic strategy eliminates the need for hazardous chemical reducing agents and heavy metal catalysts, thereby simplifying the safety profile and environmental footprint of the manufacturing process. The use of specific microbial strains, such as those from the genera Pichia, Candida, and Rhodotorula, ensures high stereoselectivity, directly producing the desired (S)-enantiomer with minimal formation of the unwanted (R)-isomer. 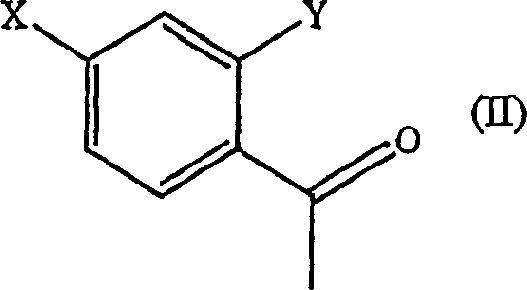 The process operates in aqueous media at moderate temperatures, which significantly reduces energy consumption and allows for the use of standard stainless-steel fermentation equipment. This shift from chemical to biological catalysis not only enhances the sustainability of the production route but also improves the overall cost-efficiency by reducing the number of synthetic steps and purification stages required to achieve pharmaceutical-grade purity.
The process operates in aqueous media at moderate temperatures, which significantly reduces energy consumption and allows for the use of standard stainless-steel fermentation equipment. This shift from chemical to biological catalysis not only enhances the sustainability of the production route but also improves the overall cost-efficiency by reducing the number of synthetic steps and purification stages required to achieve pharmaceutical-grade purity.
Mechanistic Insights into Microbial Oxidoreductase-Catalyzed Reduction
The core of this technology lies in the enzymatic activity of oxidoreductases, specifically ketoreductases, which facilitate the transfer of hydride equivalents from cofactors like NADH or NADPH to the carbonyl group of the substrate. The microorganisms act as factories for these enzymes, often regenerating the necessary cofactors in situ through the metabolism of added carbon sources such as glucose. This cofactor regeneration is critical for economic viability, as it avoids the need for stoichiometric amounts of expensive cofactors. The enzyme's active site provides a chiral environment that dictates the stereochemical outcome of the reduction, ensuring that the hydride attack occurs from a specific face of the planar ketone group. 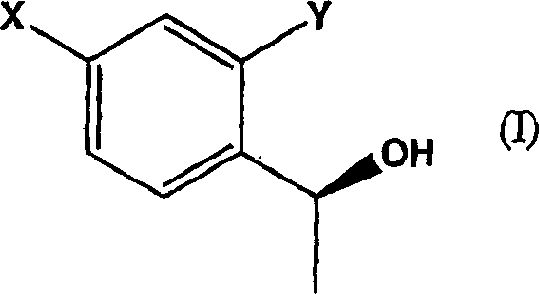 This precise spatial arrangement results in the formation of the (S)-1-arylethanol structure with high fidelity. The patent highlights that the reducing capacity of the cells is dependent on the growth stage and the specific strain used, indicating that process optimization involves careful control of fermentation parameters to maximize enzyme expression and activity.
This precise spatial arrangement results in the formation of the (S)-1-arylethanol structure with high fidelity. The patent highlights that the reducing capacity of the cells is dependent on the growth stage and the specific strain used, indicating that process optimization involves careful control of fermentation parameters to maximize enzyme expression and activity.
Impurity control is inherently superior in this biocatalytic system due to the high substrate specificity of the enzymes involved. Unlike chemical reducers that might react with other reducible groups on the molecule, the oxidoreductases are highly selective for the ketone functionality, leaving other sensitive substituents intact. This selectivity minimizes the formation of side products such as over-reduced species or by-products from competing chemical reactions. The patent data demonstrates that by selecting the appropriate microorganism, manufacturers can achieve conversion rates exceeding 90% with enantiomeric excess values often surpassing 99%. This level of purity significantly reduces the burden on downstream processing, as fewer chromatographic or crystallization steps are needed to remove impurities, thereby increasing the overall yield and reducing the cost of goods sold for the final active pharmaceutical ingredient.
How to Synthesize (S)-1-arylethanols Efficiently
Implementing this synthesis route requires a structured approach to fermentation and biotransformation to ensure consistent quality and yield. The process generally involves cultivating the selected microorganism in a nutrient-rich medium to achieve sufficient biomass and enzyme activity before introducing the ketone substrate. The reaction conditions, including pH, temperature, and aeration, must be tightly controlled to maintain enzyme stability and cofactor regeneration efficiency. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the patented results.
- Prepare microbial cell suspension using selected strains like Pichia methanolica or Candida species in a buffered medium.
- Add the substituted acetophenone substrate (Formula II) to the suspension with a carbon source like glucose for cofactor regeneration.
- Maintain aerobic conditions at 25°C to 40°C with stirring until complete conversion to the chiral alcohol (Formula I) is achieved.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this microbial reduction technology offers tangible benefits that extend beyond mere technical feasibility. The shift to a biocatalytic process fundamentally alters the cost structure and risk profile of manufacturing chiral intermediates. By eliminating the reliance on precious metal catalysts and hazardous reagents, the process reduces exposure to volatile raw material markets and regulatory compliance costs associated with heavy metal disposal. The use of readily available fermentation infrastructure allows for flexible production scaling, enabling manufacturers to respond quickly to changes in demand without significant capital investment in specialized chemical reactors. This flexibility is crucial for maintaining supply continuity in the face of market fluctuations or unexpected disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the reduction in purification steps lead to substantial cost savings in pharmaceutical intermediates manufacturing. The in situ regeneration of cofactors using inexpensive carbon sources like glucose further drives down operational expenses. Additionally, the higher reaction yields and reduced waste generation contribute to a more efficient use of raw materials, lowering the overall cost per kilogram of the produced intermediate. These efficiencies translate into a more competitive pricing structure for the final drug product, enhancing market accessibility.
- Enhanced Supply Chain Reliability: Utilizing robust microbial strains that can be stored and propagated easily ensures a stable and reliable supply of the biocatalyst. Unlike chemical catalysts that may have long lead times or supply constraints, microorganisms can be maintained in-house or sourced from multiple certified suppliers. This redundancy mitigates the risk of supply chain interruptions and provides greater control over the production schedule. The ability to produce the intermediate on demand reduces the need for large inventory holdings, freeing up working capital and reducing storage costs.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory shake flasks to industrial fermenters, facilitating the commercial scale-up of complex pharmaceutical intermediates. The aqueous nature of the reaction and the absence of toxic solvents simplify waste treatment and ensure compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces environmental liability but also aligns with the sustainability goals of major pharmaceutical companies, making it a preferred choice for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective reduction technology. These answers are derived from the specific data and embodiments disclosed in the patent documentation, providing clarity on performance metrics and operational requirements. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What is the enantiomeric excess achievable with this microbial reduction process?
A: The patent data indicates that optimized conditions using specific microorganisms can achieve enantiomeric excess values greater than 99%, ensuring high optical purity for pharmaceutical applications.
Q: Which microorganisms are most effective for this stereoselective reduction?
A: Strains such as Pichia methanolica, Candida sonorensis, and Rhodotorula glutinis have demonstrated high conversion rates and stereoselectivity for reducing substituted acetophenones to (S)-1-arylethanols.
Q: How does this biocatalytic method compare to chemical reduction in terms of impurity profile?
A: The enzymatic approach offers superior specificity, significantly reducing the formation of unwanted by-products and simplifying downstream purification compared to traditional chemical reducing agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-arylethanols Supplier
The technical potential of this microbial reduction pathway is immense, offering a route to high-value chiral building blocks with unmatched purity and efficiency. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with state-of-the-art fermentation and downstream processing capabilities, ensuring stringent purity specifications and rigorous QC labs are in place to meet global regulatory standards. We understand the critical nature of chiral intermediates in drug development and are committed to delivering consistent quality.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective supply of high-purity chiral alcohols for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
