Advanced Synthesis of Dicyclic Hexane Liquid Crystal Monomers for High-Performance Displays
The landscape of liquid crystal display (LCD) technology continues to evolve, driven by the relentless demand for materials that offer superior electro-optical performance, including low viscosity and high resistivity. Patent CN101580714B introduces a transformative preparation method for dicyclic hexane liquid crystal monomers, addressing critical bottlenecks in traditional manufacturing processes. This intellectual property outlines a robust four-step synthetic pathway that utilizes stable, commercially available raw materials to achieve a total yield exceeding 55 percent and a remarkable final purity of 99.96 percent. For R&D directors and procurement strategists in the electronic chemicals sector, this methodology represents a significant leap forward, offering a reliable electronic chemical supplier pathway that mitigates the risks associated with unstable intermediates and complex purification protocols often seen in legacy technologies.
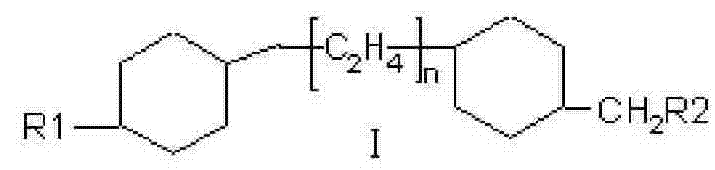
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bicyclic ethane derivatives has been plagued by inefficient routes that compromise both yield and operational safety. Early methodologies, such as those disclosed in patent DE332173, relied on extended reaction sequences starting from 4-(4'-alkyl-cyclohexyl)-cyclohexane carboxylic acid. As illustrated in the reaction scheme below, these traditional pathways involve multiple transformation steps that cumulatively degrade the overall process efficiency, leading to substantial material loss and increased production costs. Furthermore, alternative approaches utilizing alkyl acyl chlorides as starting materials, while shorter in step count, introduce severe handling challenges due to the inherent instability and high reactivity of the acyl chloride functional group.
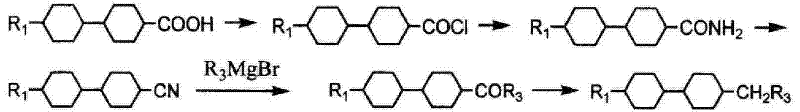
The susceptibility of acyl chlorides to hydrolysis creates a volatile production environment where moisture control becomes a critical and costly constraint. Even minor exposure to atmospheric humidity can lead to the degradation of the starting material before the reaction commences, resulting in unpredictable yields and the generation of difficult-to-remove acidic impurities. This instability not only jeopardizes the consistency of the final product quality but also necessitates specialized storage and handling infrastructure, thereby inflating the capital expenditure required for manufacturing facilities. Consequently, the industry has long sought a more robust synthetic strategy that balances step economy with chemical stability to ensure scalable and cost-effective production.
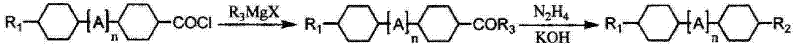
The Novel Approach
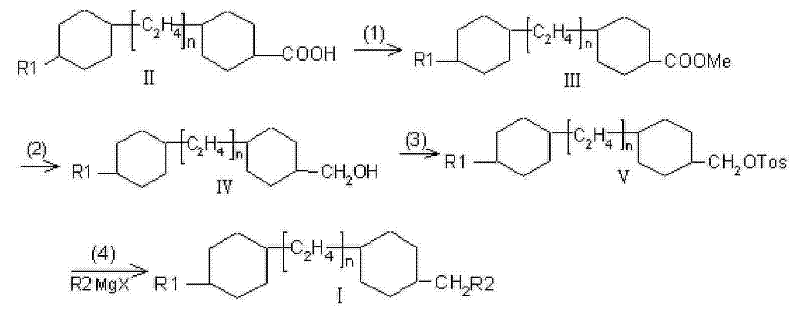
In stark contrast to these legacy methods, the novel approach detailed in CN101580714B employs a meticulously designed four-step sequence that prioritizes intermediate stability and reaction controllability. The process initiates with the esterification of a trans-4-(trans-4-alkyl-cyclohexyl)cyclohexanecarboxylic acid, followed by a selective reduction to the corresponding alcohol. This alcohol is then converted into a tosylate derivative, which serves as a superior leaving group for the final Grignard coupling step. This strategic selection of intermediates ensures that each stage of the synthesis proceeds under mild conditions, avoiding the harsh reagents and extreme temperatures that often characterize older protocols. The result is a streamlined workflow that maintains high stereochemical integrity while delivering exceptional purity levels suitable for high-end TFT-LCD applications.
Mechanistic Insights into LiCuCl2-Catalyzed Grignard Coupling
The cornerstone of this synthetic innovation lies in the final coupling step, where a lithium cuprate catalyst plays a pivotal role in facilitating the nucleophilic substitution. In traditional Grignard reactions involving alkyl halides or sulfonates, the reaction kinetics can be sluggish, often requiring elevated temperatures that promote undesirable elimination side reactions. However, the introduction of a lithium cupric chloride catalyst system dramatically enhances the electrophilicity of the tosylate intermediate, allowing the alkyl magnesium halide to attack efficiently at temperatures as low as 10-50°C. This catalytic acceleration not only shortens the reaction time but also suppresses the formation of alkene by-products, which are common impurities in uncatalyzed substitutions. The mechanism likely involves the transmetallation of the Grignard reagent to form a more reactive organocuprate species in situ, which possesses superior nucleophilic character towards the sterically hindered cyclohexyl ring system.
Furthermore, the choice of the tosylate leaving group is mechanistically superior to halides in this specific context due to its non-nucleophilic nature and excellent leaving group ability. Unlike chloride or bromide ions, which can sometimes participate in reverse reactions or coordinate with the catalyst to inhibit turnover, the tosylate anion is stable and inert under the reaction conditions. This ensures that the equilibrium is driven decisively towards the formation of the desired carbon-carbon bond. Additionally, the mild alkaline conditions used in the preceding tosylation step ensure that the sensitive trans-trans stereochemistry of the bicyclic core is preserved throughout the sequence. By avoiding strong bases or acidic conditions that could induce epimerization, the process guarantees that the final liquid crystal monomer possesses the precise geometric configuration required for optimal mesogenic properties and low viscosity in the final display mixture.
How to Synthesize Dicyclic Hexane Liquid Crystal Monomer Efficiently
The implementation of this synthesis requires careful attention to reagent quality and temperature control, particularly during the reduction and coupling phases. The process begins with the conversion of the carboxylic acid precursor into a methyl ester using methanol and an acid catalyst, a straightforward equilibrium reaction that sets the stage for subsequent reduction. Following isolation, the ester is subjected to hydride reduction using a mixed system such as lithium chloride and potassium borohydride, which offers a safer and more controllable alternative to pure lithium aluminum hydride. The resulting alcohol is then activated via tosylation, creating the requisite substrate for the final organometallic coupling. Detailed standardized operating procedures for each of these critical transformations are provided below to ensure reproducibility and safety in a pilot or commercial plant setting.
- Perform acid-catalyzed esterification of trans-4-(trans-4-alkyl-cyclohexyl)cyclohexanecarboxylic acid with methanol to form the methyl ester intermediate.
- Reduce the methyl ester to the corresponding benzyl alcohol derivative using a borohydride or aluminide reducing agent system in an organic solvent.
- Convert the alcohol intermediate into a tosylate leaving group by reacting with p-toluenesulfonyl chloride under alkaline conditions.
- Execute the final carbon-carbon bond formation via a lithium cuprate-catalyzed Grignard reaction between the tosylate and an alkyl magnesium halide.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented methodology offers profound advantages by decoupling production efficiency from the volatility of specialized reagent markets. The reliance on stable carboxylic acid starting materials, rather than moisture-sensitive acyl chlorides, drastically simplifies logistics and warehousing requirements. This shift eliminates the need for expensive inert atmosphere storage for raw materials and reduces the risk of batch rejection due to precursor degradation during transit. Moreover, the use of commodity chemicals such as methanol, tosyl chloride, and standard Grignard reagents ensures that the supply chain remains resilient against market fluctuations, providing procurement managers with a predictable and secure sourcing strategy for critical display materials.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the elimination of yield losses associated with unstable intermediates. By utilizing a tosylate intermediate instead of an acyl chloride, the process avoids the costly hydrolysis side reactions that typically plague traditional syntheses, leading to a direct improvement in mass balance. Additionally, the high selectivity of the cuprate-catalyzed coupling step minimizes the formation of by-products, which significantly reduces the burden on downstream purification processes such as chromatography or recrystallization. This streamlined purification requirement translates into lower solvent consumption and reduced waste disposal costs, contributing to a substantially lower cost of goods sold (COGS) without compromising on the stringent purity specifications demanded by the LCD industry.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining long-term contracts with panel manufacturers. Since the reaction conditions are mild and the intermediates are chemically stable, the risk of unexpected production stoppages due to runaway reactions or equipment corrosion is significantly mitigated. This operational stability allows for more accurate production planning and inventory management, enabling suppliers to meet tight delivery windows with greater confidence. Furthermore, the accessibility of the raw materials means that supply disruptions are less likely to occur compared to routes relying on exotic or highly regulated precursors, thereby securing the continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as esterification, reflux, and liquid-liquid extraction that are well-understood in chemical engineering. The absence of heavy metal catalysts, which often require complex and expensive removal steps to meet electronic grade specifications, simplifies the regulatory compliance landscape. The mild reaction temperatures and the use of common organic solvents like THF and dichloromethane facilitate efficient solvent recovery and recycling, aligning with modern green chemistry principles. This environmental compatibility not only reduces the ecological footprint of the manufacturing process but also lowers the operational costs associated with waste treatment and environmental permitting, making it an ideal candidate for large-scale industrialized production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: Why is the tosylate intermediate preferred over acyl chlorides in this synthesis?
A: Unlike acyl chlorides which are highly reactive and prone to hydrolysis leading to yield loss, the tosylate intermediate offers superior chemical stability during storage and handling, ensuring consistent reaction outcomes and higher final purity.
Q: What is the role of the lithium cuprate catalyst in the final step?
A: The lithium cuprate catalyst significantly accelerates the nucleophilic substitution rate of the Grignard reagent on the tosylate group while minimizing side reactions such as elimination, thereby improving overall reaction efficiency and product selectivity.
Q: How does this method ensure high stereochemical purity?
A: The mild reaction conditions employed throughout the four-step sequence, particularly the retention of configuration during the reduction and substitution steps, preserve the critical trans-trans stereochemistry required for optimal liquid crystal performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclic Hexane Liquid Crystal Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101580714B can be fully realized in a manufacturing environment. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of dicyclic hexane monomer meets the exacting standards required for next-generation TFT-LCD panels. Our capability to manage complex organometallic reactions safely and efficiently positions us as a leader in the supply of high-performance electronic chemicals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth in the competitive display materials market.
