Scalable Enzymatic Production of Optically Active D-Pantolactone for Pharmaceutical Intermediates
Scalable Enzymatic Production of Optically Active D-Pantolactone for Pharmaceutical Intermediates
The global demand for high-purity chiral intermediates, particularly for the synthesis of essential vitamins like Pantothenic Acid (Vitamin B5), necessitates robust and economically viable manufacturing technologies. Patent CN1761742A introduces a groundbreaking biocatalytic approach that leverages recombinant DNA technology to produce lactonase enzymes with superior stability and activity. This innovation addresses critical bottlenecks in the traditional production of optically active γ-lactone derivatives, specifically D-pantolactone, which serves as a pivotal precursor for D-pantothenic acid, D-panthenol, and coenzyme A. By utilizing genetically engineered filamentous fungi capable of secreting glycosylated lactonase, this method offers a sustainable alternative to classical chemical resolution, promising significant improvements in yield, optical purity, and process safety for the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial manufacture of D-pantolactone relied heavily on the optical resolution of chemically synthesized racemic mixtures. This traditional paradigm required the use of stoichiometric amounts of expensive and often toxic chiral resolving agents, such as quinic acid or strychnine, to separate the desired D-enantiomer from the L-enantiomer. These processes were inherently inefficient, typically capping theoretical yields at 50% unless complex racemization steps were employed, which added further cost and environmental burden. Furthermore, the recovery of the resolving agents was often difficult and incomplete, leading to substantial waste generation and increased disposal costs. The reliance on harsh chemical conditions also posed risks regarding product degradation and the formation of difficult-to-remove impurities, complicating the downstream purification required for pharmaceutical-grade intermediates.
The Novel Approach
In stark contrast, the technology disclosed in CN1761742A utilizes a highly specific enzymatic kinetic resolution driven by recombinant lactonase. By cloning the lactonase gene from Fusarium oxysporum and expressing it in optimized fungal hosts like Aspergillus oryzae or Acremonium chrysogenum, the process achieves selective hydrolysis of the racemic substrate without the need for hazardous chemical reagents. The key innovation lies in the co-expression of the enzyme with its native signal peptide, ensuring proper secretion and glycosylation. This biological precision allows for the direct conversion of the racemate into the desired chiral acid or the enrichment of the unreacted lactone with high enantiomeric excess. The elimination of heavy metal catalysts and organic resolving agents drastically simplifies the workflow, reducing the environmental footprint while enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Recombinant Lactonase Expression and Glycosylation
The core scientific advancement of this patent lies in the sophisticated genetic engineering strategy employed to overcome the stability issues plaguing earlier recombinant enzymes. Previous attempts to express lactonase in bacterial hosts like E. coli resulted in proteins lacking post-translational modifications, specifically glycosylation, which rendered them unstable and less active under industrial conditions. The present invention solves this by introducing the full-length lactonase gene, including the DNA sequence encoding the NH2-terminal signal peptide region, into eukaryotic filamentous fungal hosts. This signal peptide directs the nascent polypeptide into the secretory pathway, where it undergoes essential glycosylation in the endoplasmic reticulum. The resulting recombinant enzyme possesses a molecular weight and structural conformation identical to the wild-type natural enzyme, conferring exceptional thermal and pH stability that is crucial for large-scale bioprocessing.
Furthermore, the mechanism of asymmetric hydrolysis is tightly controlled by the enzyme's stereoselectivity. The recombinant lactonase selectively recognizes and hydrolyzes the D-configuration of the pantolactone ring, opening it to form D-pantothenic acid while leaving the L-pantolactone untouched, or vice versa depending on the specific enzyme variant used. This kinetic resolution is highly efficient, with experimental data indicating that immobilized transformants can achieve hydrolysis rates approaching 50% with optical purity exceeding 95% e.e. The ability to immobilize these recombinant cells on carriers like alginate gel or synthetic resins further enhances the operational stability, allowing the biocatalyst to be reused across multiple batches. This mechanistic robustness ensures consistent product quality and minimizes the risk of batch-to-batch variability, a critical factor for regulatory compliance in pharmaceutical manufacturing.
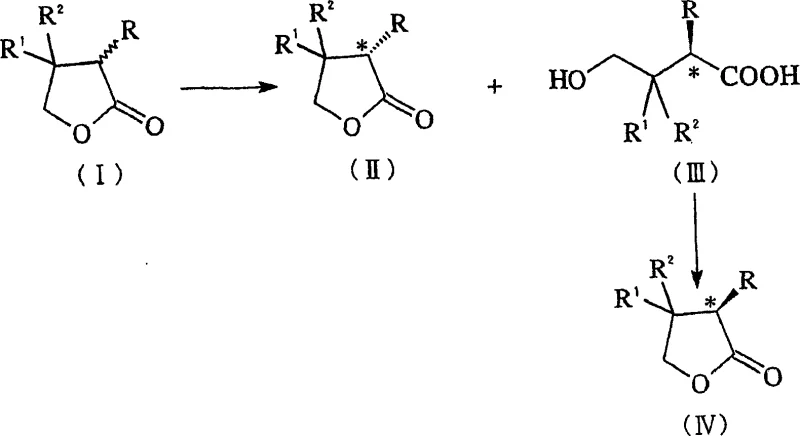
How to Synthesize D-Pantolactone Efficiently
The implementation of this enzymatic route requires a systematic approach to strain construction, fermentation, and biocatalysis. The process begins with the construction of an expression vector containing the lactonase gene fused with a signal peptide sequence, which is then transformed into a suitable fungal host. Following the selection of high-producing transformants, the organisms are cultivated in optimized media to maximize enzyme secretion. The harvested biomass or supernatant is then utilized as the biocatalyst in a buffered aqueous system containing the racemic substrate. Detailed standard operating procedures for strain maintenance, fermentation parameters, and downstream isolation are critical for reproducibility. For a comprehensive guide on the specific laboratory protocols and scaling parameters derived from the patent examples, please refer to the standardized synthesis steps outlined below.
- Transform filamentous fungal hosts such as Aspergillus oryzae or Acremonium chrysogenum with DNA encoding lactonase and a signal peptide region.
- Cultivate the transformants under aerobic conditions to secrete the glycosylated recombinant lactonase into the medium or accumulate it within the cells.
- Contact the enzyme system with racemic pantolactone to selectively hydrolyze the D-isomer, followed by separation and acidification to recover the optically active product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this recombinant enzymatic technology represents a strategic opportunity to optimize cost structures and mitigate supply risks. The primary economic driver is the drastic reduction in raw material costs associated with the elimination of chiral resolving agents. In traditional chemical processes, these agents can account for a significant portion of the total variable cost, and their price volatility can disrupt budget forecasting. By replacing them with a biocatalytic system that uses water as the primary solvent and renewable carbon sources for fermentation, manufacturers can achieve substantial cost savings. Additionally, the high specificity of the enzyme reduces the formation of by-products, which lowers the burden on purification units and decreases the consumption of solvents and energy during the isolation phase.
From a supply chain reliability perspective, this technology offers enhanced scalability and continuity. Fermentation-based production is inherently more scalable than extraction from natural sources or complex multi-step chemical synthesis. The use of robust fungal hosts ensures that production capacity can be ramped up quickly to meet surges in demand for Vitamin B5 derivatives without the long lead times associated with building new chemical synthesis lines. Moreover, the ability to immobilize the enzyme allows for continuous or semi-continuous processing, which improves equipment utilization rates and reduces the overall manufacturing cycle time. This operational flexibility ensures a steady flow of high-purity intermediates, safeguarding against market shortages and enabling just-in-time delivery models for downstream pharmaceutical customers.
Environmental compliance and sustainability are also major commercial advantages that translate into long-term viability. The enzymatic process operates under mild conditions of temperature and pressure, significantly reducing energy consumption compared to high-temperature chemical reactions. Furthermore, the aqueous nature of the reaction minimizes the generation of hazardous organic waste, simplifying wastewater treatment and reducing disposal fees. As global regulations on industrial emissions become stricter, adopting this green chemistry approach future-proofs the supply chain against regulatory shocks. The combination of lower operational expenditures, reduced environmental liability, and improved production efficiency makes this technology a compelling choice for maintaining a competitive edge in the global market for fine chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant lactonase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the process. Understanding these details is essential for technical teams evaluating the integration of this biocatalytic route into existing manufacturing frameworks. For more specific technical data or custom feasibility studies, our team is available to provide detailed assessments tailored to your production requirements.
Q: How does the recombinant lactonase improve stability compared to wild-type enzymes?
A: By introducing the signal peptide region during recombinant expression in filamentous fungi, the enzyme undergoes natural glycosylation. This post-translational modification significantly enhances thermal and operational stability compared to non-glycosylated versions produced in bacterial systems like E. coli.
Q: What are the primary cost advantages of this enzymatic route over chemical resolution?
A: This method eliminates the need for expensive chiral resolving agents such as quinic acid or strychnine. Additionally, the ability to recycle the unreacted isomer and the high specificity of the enzyme reduce raw material waste and downstream purification costs.
Q: Can this process be scaled for industrial production of Vitamin B5 intermediates?
A: Yes, the patent demonstrates successful immobilization of the recombinant cells, which allows for repeated batch operations. The use of robust fungal hosts like Aspergillus oryzae ensures high cell density fermentation suitable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Pantolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis routes described in CN1761742A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory concepts into commercial reality. Our facilities are equipped with state-of-the-art fermentation suites and downstream processing units capable of handling complex biocatalytic pathways. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of D-pantolactone meets the highest international standards for pharmaceutical applications.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can optimize your manufacturing processes, reduce costs, and secure a sustainable supply of critical chiral intermediates for the global market.
