Revolutionizing Carbocyclic Nucleoside Production via Efficient Metal Hydride Reduction Technology
Revolutionizing Carbocyclic Nucleoside Production via Efficient Metal Hydride Reduction Technology
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex antiviral intermediates, particularly those required for carbocyclic nucleosides like Abacavir. Patent CN1277807C introduces a transformative methodology for preparing (1S,4R)- or (1R,4S)-1-amino-4-(methylol)-2-cyclopentene, a critical scaffold in modern antiretroviral therapy. This technology departs from laborious chiral pool syntheses, utilizing a direct metal hydride reduction of 2-azabicyclo[2.2.1]hept-5-en-3-one, commonly known as Vince lactam. By leveraging specific catalytic conditions involving lithium salts and controlled solvent systems, this process achieves high stereoselectivity and yield. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more cost-effective and scalable manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active aminoalcohols for nucleoside analogs relied heavily on natural chiral pools such as D-glucose or D-serine. These traditional routes are inherently inefficient, often necessitating upwards of fifteen distinct synthetic steps to construct the cyclopentene ring and install the necessary functional groups. Each step introduces potential yield losses, requires expensive protecting group chemistry, and generates significant chemical waste. Furthermore, methods utilizing lithium aluminium hydride reductions on amino acid precursors have shown poor control over the double bond integrity, often leading to over-reduction and saturated byproducts that are difficult to separate. The reliance on costly resolving agents like (-)-dibenzoyl tartaric acid in older protocols further exacerbates the economic burden, making these processes unsuitable for large-scale commercial production where margin compression is a constant pressure.
The Novel Approach
The innovative process described in the patent data circumvents these bottlenecks by employing a direct reduction strategy on the bicyclic ketone scaffold. Instead of building the ring from scratch, the method starts with the readily available Vince lactam and reduces the carbonyl functionality directly to the aminoalcohol. This approach drastically simplifies the synthetic tree, eliminating multiple protection and deprotection stages. The use of metal hydrides, specifically tailored with lithium salt additives and protic co-solvents, allows for precise control over the reaction trajectory. This ensures that the olefinic double bond remains intact while the ketone is reduced, preserving the structural integrity required for subsequent nucleoside coupling. This streamlined workflow not only accelerates the timeline from raw material to finished intermediate but also aligns perfectly with the needs of a reliable pharmaceutical intermediate supplier seeking to optimize throughput.
![Reduction of 2-azabicyclo[2.2.1]hept-5-en-3-one to aminoalcohol using metal hydride](/insights/img/aminoalcohol-synthesis-vince-lactam-pharma-supplier-20260308201030-02.png)
Mechanistic Insights into Metal Hydride-Catalyzed Reduction
The core of this technological advancement lies in the nuanced application of metal hydride chemistry. While simple reductions with reagents like lithium aluminium hydride are known to lack selectivity, the patented method introduces a sophisticated modulation of reactivity. By utilizing alkali metal or alkaline earth metal hydrides, such as sodium borohydride or lithium borohydride, in conjunction with lithium salts like LiCl or LiBr, the electronic environment of the reducing agent is altered. This modification enhances the nucleophilicity towards the carbonyl carbon while minimizing attack on the conjugated double bond. The reaction is typically conducted in non-protic or mixed protic-aprotic solvent systems, such as tetrahydrofuran or dioxane mixed with lower alcohols. The presence of water or alcohol additives acts as a proton source that facilitates the breakdown of the borate intermediate, driving the reaction to completion without requiring harsh acidic workups that could degrade the sensitive aminoalcohol product.
Impurity control is another critical aspect addressed by this mechanistic design. In conventional reductions, the formation of saturated cyclopentane derivatives is a common side reaction that compromises the purity profile essential for API manufacturing. The specific stoichiometry and temperature controls defined in the patent—often maintaining temperatures between 60°C and 150°C depending on the hydride source—suppress these side pathways. Furthermore, the subsequent resolution steps, whether enzymatic using lipases like Novozym 435 or chemical using tartrate salts, provide a secondary purification layer. The enzymatic resolution exploits the steric differences between enantiomers, acylating one while leaving the other untouched, which allows for easy physical separation. This dual-layer control of reaction selectivity and downstream purification ensures that the final high-purity pharmaceutical intermediate meets the stringent specifications required by global regulatory bodies.
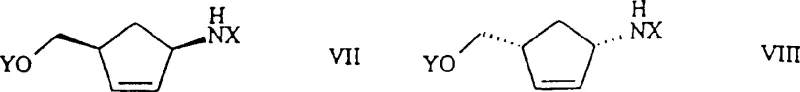
How to Synthesize 1-amino-4-(methylol)-2-cyclopentene Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and enantiomeric excess. The process begins with the preparation of the reducing agent, often generated in situ or purchased as a stable suspension, followed by the controlled addition of the bicyclic ketone substrate. Solvent choice is paramount; mixtures of ethers and alcohols are preferred to balance solubility and reactivity. Following the reduction, the crude aminoalcohol is subjected to resolution. For those prioritizing green chemistry, the enzymatic route offers a mild alternative, whereas the tartrate resolution provides a robust, scalable option for bulk manufacturing. The detailed standardized synthesis steps see the guide below.
- Reduce racemic or optically active 2-azabicyclo[2.2.1]hept-5-en-3-one using metal hydrides like LiBH4 or NaBH4 in the presence of lithium salts and alcohol additives.
- Perform kinetic resolution on the resulting racemic aminoalcohol using lipases (e.g., Novozym 435) or chemical resolution using D-(-)-tartaric acid.
- Convert the resolved aminoalcohol into the final carbocyclic nucleoside precursor via coupling with dichloropyrimidine derivatives and subsequent cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits beyond mere technical feasibility. The primary advantage is the substantial cost savings derived from the drastic reduction in synthetic steps. By eliminating the need for long linear sequences starting from sugars or amino acids, the consumption of raw materials, solvents, and energy is significantly lowered. This efficiency directly impacts the cost of goods sold (COGS), allowing for more competitive pricing in the global market. Additionally, the reliance on commodity chemicals like borohydrides and tartaric acid, rather than exotic chiral catalysts or scarce natural products, mitigates supply risk. This ensures a stable supply chain capable of withstanding market fluctuations and raw material shortages, which is critical for maintaining continuous API production schedules.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences removes the need for expensive reagents and extensive purification operations between each stage. This consolidation of the synthetic route leads to a drastic simplification of the manufacturing process, reducing labor hours and equipment occupancy time. Consequently, the overall production cost is optimized, providing a significant economic advantage over legacy methods that suffer from low atom economy and high waste generation.
- Enhanced Supply Chain Reliability: The starting material, Vince lactam, is a well-established industrial chemical with multiple global sources, ensuring that the supply chain is not dependent on a single vendor or a volatile agricultural harvest. The robustness of the reduction chemistry means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply output. This reliability is essential for long-term contracts with major pharmaceutical companies that demand guaranteed delivery timelines.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial stainless steel reactors. The solvent systems employed are standard organic solvents that can be efficiently recovered and recycled, minimizing environmental impact and waste disposal costs. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory compliance regarding waste management and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational realities of the process. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using metal hydride reduction over traditional chiral pool synthesis for this intermediate?
A: Traditional methods starting from D-glucose or D-serine require up to 15 synthetic steps with complex protection and deprotection sequences. The patented metal hydride reduction route significantly shortens the synthetic pathway, directly converting the bicyclic ketone to the aminoalcohol scaffold, thereby reducing material costs and processing time.
Q: How is optical purity achieved in this manufacturing process?
A: Optical purity is secured through highly efficient resolution techniques. The process utilizes either biocatalytic kinetic resolution with immobilized lipases like Candida antarctica lipase B or classical chemical resolution using D-(-)-tartaric acid, both of which yield enantiomerically pure tartrate salts or free bases suitable for downstream nucleoside synthesis.
Q: Is this process scalable for commercial API production?
A: Yes, the methodology is designed for industrial applicability. The reduction step employs robust reagents like sodium borohydride or lithium borohydride which are commercially available in bulk, and the reaction conditions (temperature and solvent systems) are optimized for large-scale reactors, ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-amino-4-(methylol)-2-cyclopentene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of antiviral drug development. Our technical team has extensively analyzed the pathways described in CN1277807C and possesses the expertise to execute this complex chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot scale to full commercialization. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-amino-4-(methylol)-2-cyclopentene meets the highest industry standards for chirality and chemical purity.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall production costs.
