Advanced Radical Synthesis of Tetrahydropyrrole Betaines for Commercial Detergent Applications
Introduction to Novel Tetrahydropyrrole Betaine Technology
The landscape of specialty surfactant manufacturing is undergoing a significant transformation driven by the need for safer, more efficient synthetic routes. Patent CN85105497A introduces a groundbreaking methodology for the preparation of new 3-sulfinate methyl- or 3-sulfonate methyl-4-sulfomethyltetrahydropyrrole betaines and their salts. This technology represents a pivotal shift away from traditional alkylation methods that rely on hazardous reagents, offering a robust alternative for producing high-performance ampholytic surfactants. The core innovation lies in the utilization of a free radical cyclization mechanism initiated by persulfates in the presence of bisulfites, enabling the direct construction of the pyrrolidine ring system with integrated sulfonic acid functionality. This approach not only mitigates severe safety risks associated with carcinogenic sultones but also delivers exceptional reaction kinetics, often achieving quantitative conversion in mere minutes. For industrial partners seeking a reliable specialty surfactant supplier, understanding the mechanistic advantages of this radical pathway is essential for evaluating its potential in modern detergent and intermediate manufacturing pipelines.
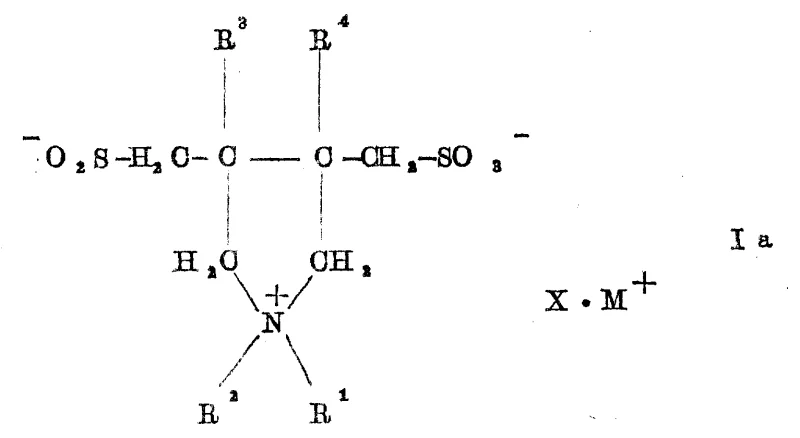
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur-containing betaines has been dominated by the quaternization of tertiary amines using hydroxyalkyl sulfonic acid derivatives or, more commonly, cyclic sultones such as propane sultone. While effective in forming the zwitterionic structure, these conventional pathways suffer from critical drawbacks that hinder modern sustainable manufacturing. The primary concern is the toxicological profile of propane sultone, which is classified as a dangerous carcinogen, necessitating elaborate containment systems, specialized personal protective equipment, and rigorous waste treatment protocols to ensure worker safety and environmental compliance. Furthermore, reactions involving sultones often require extended reaction times and elevated temperatures to drive the alkylation to completion, leading to lower space-time yields and increased energy consumption. The formation of complex by-product mixtures is also a frequent issue, complicating downstream purification and reducing the overall purity of the final active ingredient, which is detrimental for applications requiring high-purity tetrahydropyrrole betaines in sensitive formulations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a radical-mediated cyclization of diallyl or triallyl ammonium salts with bisulfites, catalyzed by persulfates or other oxidizing agents. This method fundamentally bypasses the need for hazardous alkylating agents, replacing them with inexpensive and readily available inorganic salts like sodium bisulfite and ammonium persulfate. The reaction proceeds through a unique sulfur-cyclization-sulfinic acid mechanism that is remarkably fast and selective. By simply adjusting the pH of the reaction medium between 1.5 and 6.0, manufacturers can precisely dictate whether the final product contains a sulfinic acid group or a sulfonic acid group, providing unparalleled flexibility in molecular design. This process operates efficiently at moderate temperatures, often relying on the exothermic nature of the radical initiation to sustain the reaction, thereby reducing external heating requirements. The result is a streamlined synthesis that offers superior safety profiles, reduced operational complexity, and the ability to generate diverse betaine structures suitable for various cost reduction in detergent manufacturing strategies without compromising on performance or quality standards.
Mechanistic Insights into Persulfate-Initiated Radical Cyclization
The chemical elegance of this synthesis lies in its radical mechanism, which facilitates the simultaneous formation of the heterocyclic ring and the introduction of sulfur-containing functional groups. The process begins with the generation of sulfate radical anions from the thermal decomposition of the persulfate initiator. These highly reactive species abstract hydrogen or add to the double bonds of the diallyl ammonium precursor, triggering a cascade of radical additions involving the bisulfite ion. Unlike traditional ionic substitutions, this radical pathway allows for the closure of the five-membered pyrrolidine ring while incorporating the sulfomethyl side chain in a single concerted sequence. The presence of the persulfate is critical, as it not only initiates the chain reaction but also influences the oxidation state of the sulfur atoms incorporated into the molecule. At lower pH values, typically between 2 and 4, the reaction environment favors the stabilization of the sulfinic acid moiety, leading to the formation of Formula Ia compounds. The rapid kinetics observed, with reactions completing in seconds to minutes upon initiator addition, suggest a highly efficient propagation cycle that minimizes the lifetime of reactive intermediates, thereby suppressing side reactions such as polymerization or over-oxidation that often plague free radical chemistries in aqueous media.
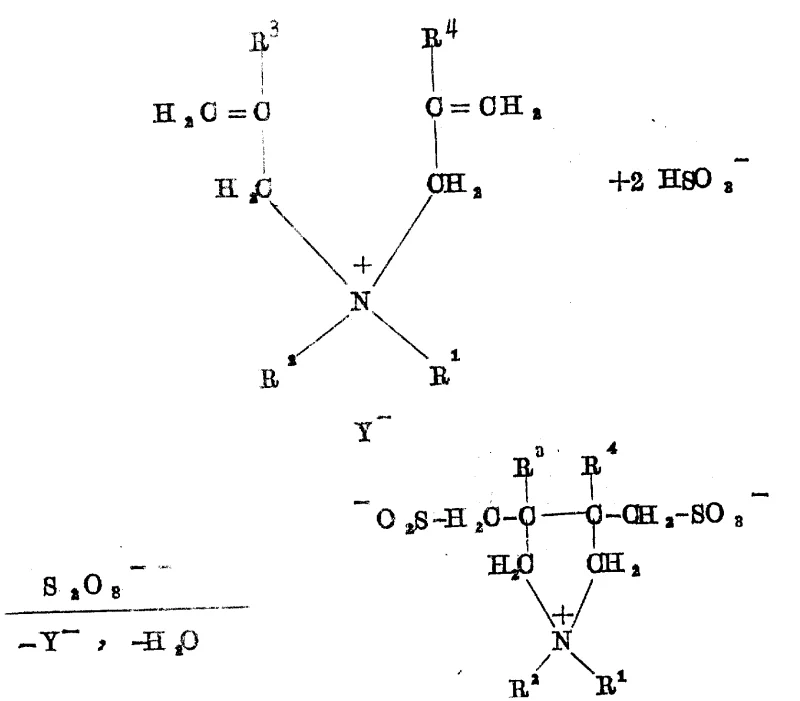
Furthermore, the selectivity of the reaction is intricately linked to the proton concentration in the solution, which modulates the reactivity of the bisulfite-sulfite buffer system. When the pH is adjusted to a slightly higher range, specifically between 2.5 and 6.0, and the molar ratio of oxidant is increased, the mechanism shifts to favor the formation of the fully oxidized sulfonate group, yielding Formula Ib structures. This pH-dependent switch is a powerful tool for process chemists, allowing for the targeted synthesis of specific betaine variants without changing the core raw materials. The mechanism also accounts for the formation of spirocyclic by-products when tetraallyl ammonium salts are used, demonstrating the versatility of the radical cyclization to accommodate different degrees of allylic substitution. Understanding these mechanistic nuances is vital for scaling up the process, as it informs the precise control strategies needed for initiator dosing and temperature management to ensure consistent product quality and maximize the yield of the desired commercial scale-up of complex surfactant molecules.
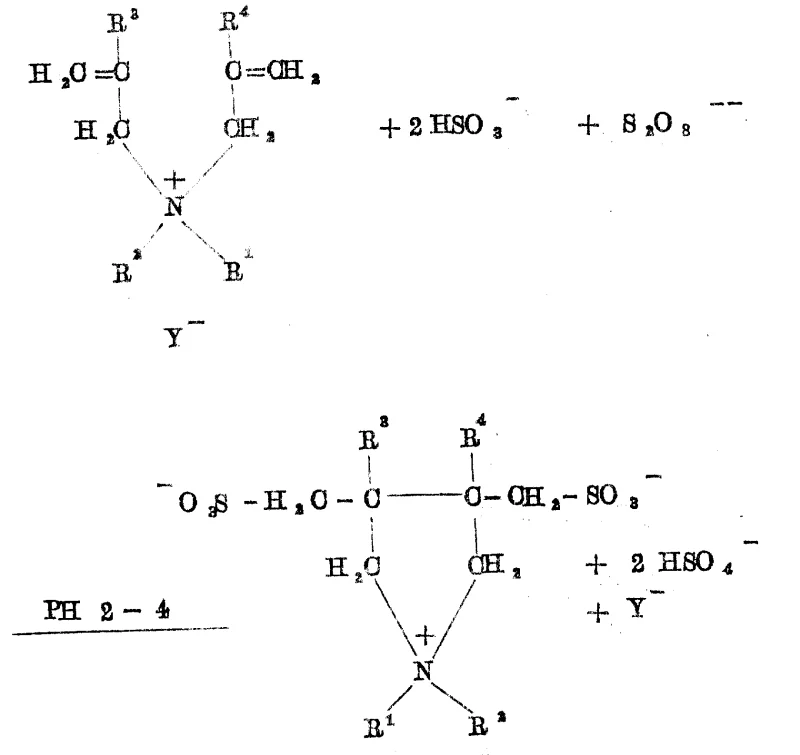
How to Synthesize Tetrahydropyrrole Betaine Efficiently
Implementing this synthesis route requires careful attention to the order of addition and the maintenance of specific reaction parameters to harness the full potential of the radical cyclization. The general procedure involves preparing a homogeneous aqueous solution of the diallyl ammonium salt and the bisulfite source, followed by a critical pH adjustment step using mineral acids or bases to set the trajectory for either sulfinic or sulfonate formation. Once the reaction mixture is stabilized at the target pH and temperature, the persulfate initiator is introduced, often resulting in an immediate and vigorous exotherm that signals the onset of rapid cyclization. Due to the speed of the reaction, which can be quantitative within minutes, the process is well-suited for semi-continuous or continuous flow operations where reagents are mixed just prior to entering a heated zone. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding initiator concentrations and workup procedures.
- Prepare an aqueous solution of diallyl ammonium salt and mix with alkali metal bisulfite, adjusting the pH to between 1.5 and 6.0 depending on the desired sulfinate or sulfonate product.
- Initiate the exothermic radical cyclization reaction by adding a catalytic amount of persulfate oxidant, ensuring temperature control as the reaction completes within minutes.
- Isolate the final betaine product by neutralizing the reaction mixture, removing inorganic salts via filtration or alcohol extraction, and drying the crystalline residue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent technology offers tangible strategic benefits that extend beyond simple chemical novelty. The elimination of carcinogenic sultones from the supply chain removes a significant regulatory burden and liability risk, simplifying the logistics of raw material handling and storage. Since the key reagents—diallyl ammonium salts, bisulfites, and persulfates—are commodity chemicals produced on a massive global scale, the supply security for this process is inherently robust, reducing the risk of disruptions caused by the scarcity of specialized fine chemical intermediates. This reliance on bulk commodities also translates into significant cost reduction in surfactant manufacturing, as the raw material costs are generally lower and more stable compared to proprietary alkylating agents. Additionally, the extremely short reaction times dramatically increase reactor throughput, allowing existing production assets to generate significantly more product per annum without the need for capital-intensive expansion of reactor farm capacity.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the replacement of expensive and hazardous sultones with low-cost inorganic oxidants and salts. By avoiding the need for specialized containment infrastructure required for carcinogens, facilities can reduce capital expenditure on safety systems and lower ongoing operational costs related to waste disposal and environmental monitoring. The high atom economy of the radical addition further ensures that raw materials are efficiently converted into the final product, minimizing waste generation and maximizing the value derived from every kilogram of input material, which directly improves the gross margin profile of the manufactured surfactant.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for long-term production planning, and this technology leverages a supply chain based on widely available industrial chemicals rather than niche intermediates. The diallyl ammonium precursors can be synthesized from abundant allyl chloride and amines, creating a resilient upstream supply network that is less susceptible to market volatility. Furthermore, the simplicity of the reaction conditions means that production can be easily transferred between different manufacturing sites globally without requiring highly specialized equipment or unique catalysts, ensuring continuity of supply even in the face of regional disruptions or geopolitical tensions affecting specific chemical corridors.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates primarily inorganic salts like sodium sulfate as by-products, which are easier to manage and treat compared to the complex organic waste streams associated with sultone alkylation. The rapid nature of the reaction reduces the energy footprint per unit of product, aligning with corporate sustainability goals and carbon reduction targets. Scaling up is facilitated by the ability to run the process continuously, which provides better control over the exothermic heat release compared to large batch reactors, thereby enhancing operational safety and allowing for the commercial scale-up of complex betaine derivatives to meet growing global demand for mild, high-performance cleansing agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical cyclization technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details helps in assessing the compatibility of this method with existing manufacturing infrastructure and quality standards.
Q: Why is this radical synthesis method preferred over traditional sultone alkylation?
A: Traditional methods often utilize propane sultone, which is a known carcinogen requiring stringent safety measures. This patent describes a safer radical addition process using bisulfite and persulfate that avoids hazardous alkylating agents entirely while achieving quantitative conversion in minutes.
Q: How does pH control influence the selectivity between sulfinate and sulfonate groups?
A: The reaction selectivity is highly dependent on initial pH. Operating at pH 2 to 4 favors the formation of the sulfinic acid derivative (Formula Ia), whereas higher pH ranges around 2.5 to 6.0 with specific oxidant ratios facilitate the formation of the sulfonate derivative (Formula Ib), allowing precise tuning of the hydrophilic head group.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the reaction is extremely rapid, often completing quantitatively within seconds to minutes upon initiator addition. This high space-time yield allows for continuous processing in smaller reactor volumes compared to batch processes that require hours, significantly enhancing production throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyrrole Betaine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced radical synthesis technology for the next generation of mild and effective surfactants. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications capable of handling the precise pH control and rapid reaction kinetics required by this process, guaranteeing a final product that meets the highest international standards for personal care and industrial cleaning applications. We are committed to delivering high-purity tetrahydropyrrole betaines that leverage the safety and efficiency benefits of this patent-protected methodology.
We invite you to collaborate with our technical team to explore how this innovative chemistry can optimize your product portfolio and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your supply chain goals and accelerate your time to market with safer, superior surfactant solutions.
