Revolutionizing Phenoxyprimidine Production: Advanced Synthesis for Commercial Scale-up
The landscape of agrochemical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for safer, more efficient, and scalable synthetic routes. Patent CN1040749C introduces a groundbreaking process for the preparation of phenoxypyrimidine compounds, which serve as critical building blocks in the production of advanced fungicides and mycocides. This technology addresses long-standing challenges in heterocyclic chemistry by replacing hazardous reagents with stable, commercially available alternatives, thereby enhancing both operational safety and economic viability. For R&D Directors and Procurement Managers seeking to optimize their supply chains, understanding the nuances of this patent is essential for securing a competitive edge in the global market. The invention specifically targets the synthesis of complex structures where W represents specific methoxy and ester configurations, offering a versatile platform for derivative development. By leveraging this intellectual property, manufacturers can achieve substantial improvements in yield consistency and impurity profiles, which are paramount for regulatory compliance in the agrochemical sector.
The transition from legacy synthesis methods to this novel approach represents a paradigm shift in how high-value intermediates are produced. Conventional methods often relied on the use of diazomethane or vitriolic methanol solutions for methylation, processes that are fraught with safety risks including explosion hazards and toxicity concerns. Furthermore, traditional formylation attempts on coumarin-2(3H) ketones frequently resulted in low success rates and difficult purification workflows. In stark contrast, the novel approach detailed in the patent utilizes a robust reaction between benzofuranone derivatives and trimethyl orthoformate in the presence of acid anhydrides. This method not only mitigates safety risks but also streamlines the reaction pathway, allowing for the direct formation of 3-(α-methoxyl group) methylene radical benzo furans-2(3H)-ketone with high efficiency. The ability to control the ratio of acetal to acrylate forms through solvent selection provides an additional layer of process control that was previously unavailable.
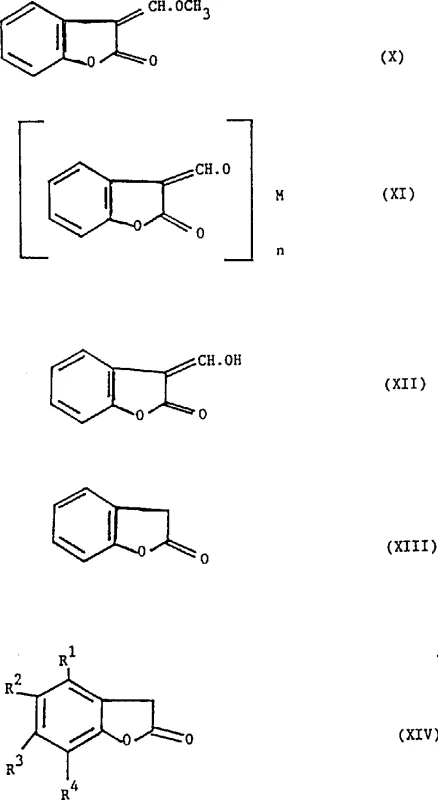
This structural versatility is visually represented in the core intermediate scaffolds, where the precise arrangement of substituents determines the reactivity and final application of the molecule. The novel pathway enables the generation of these critical structures under much milder conditions, typically ranging from 90°C to 130°C, which reduces energy consumption and equipment stress. For supply chain leaders, this translates to a more reliable production schedule with fewer interruptions caused by safety incidents or complex waste treatment requirements associated with hazardous reagents. The elimination of diazomethane also removes the need for specialized containment infrastructure, lowering the barrier to entry for contract manufacturing organizations looking to adopt this technology. Consequently, the overall cost of goods sold is optimized not just through raw material savings, but through reduced overhead in safety management and environmental compliance.
Mechanistic Insights into Trimethyl Orthoformate Condensation
A deep dive into the reaction mechanism reveals the elegance of using trimethyl orthoformate as a carbon source for the exocyclic double bond formation. The process initiates with the activation of the benzofuranone ring, where the acid anhydride acts as a dehydrating agent to drive the equilibrium towards the desired methoxymethylene product. This step is crucial for preventing the formation of unwanted by-products that often plague traditional methylation reactions. The subsequent nucleophilic attack by the methoxide ion, generated in situ from sodium methylate, facilitates the coupling with the halogenated pyrimidine ring. This condensation reaction is highly sensitive to stoichiometry and temperature, with the patent specifying optimal molar ratios between the benzofuranone derivative and the pyrimidine compound to maximize yield. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters for specific substrate variations, ensuring consistent quality across different batches.
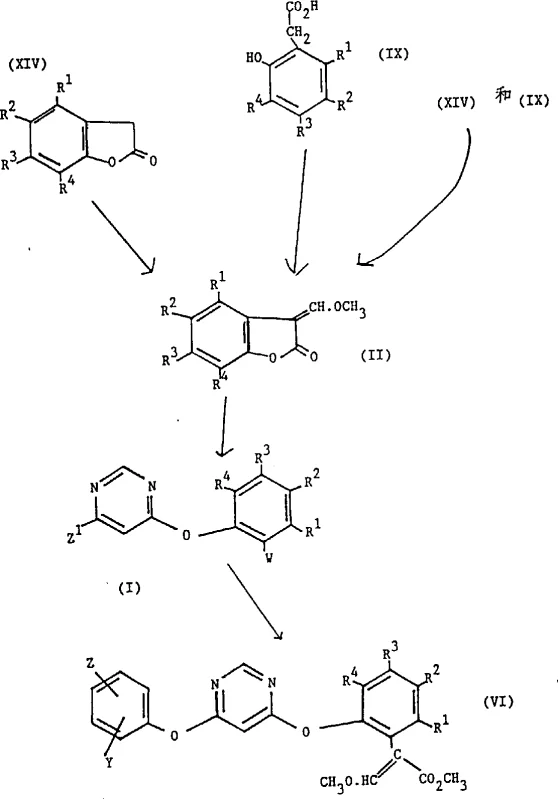
The comprehensive reaction scheme illustrates the complete transformation from simple precursors to the complex phenoxypyrimidine target, highlighting the key intermediates and transition states involved. Impurity control is inherently built into this mechanism, as the specific reaction conditions favor the formation of the (E)-isomer, which is often the biologically active configuration required for fungicidal activity. The ability to isolate the intermediate in a substantially pure state, defined as greater than 85% purity, simplifies downstream processing and reduces the load on purification columns or crystallization steps. For R&D teams, this means faster method development cycles and more robust analytical protocols. The use of common solvents like toluene, methyl acetate, and tetrahydrofuran further enhances the practicality of the method, as these materials are readily available in bulk quantities globally. This mechanistic robustness ensures that the process remains stable even when scaled up to multi-ton reactors, providing confidence to stakeholders investing in long-term production capacity.
How to Synthesize 3-(α-methoxymethylene)benzofuran-2(3H)-one Efficiently
Implementing this synthesis route requires a clear understanding of the sequential steps involved in transforming the starting materials into the high-value intermediate. The process begins with the cyclization or functionalization of the benzofuranone core, followed by the critical condensation with trimethyl orthoformate. Detailed standard operating procedures are essential to maintain the precise temperature controls and reagent addition rates specified in the patent documentation. Operators must be trained to handle the exothermic nature of the methoxide addition carefully to prevent thermal runaways. The following guide outlines the standardized workflow that ensures reproducibility and safety across all production scales. Adhering to these steps is vital for achieving the theoretical yields and purity profiles promised by the intellectual property.
- React benzofuran-2(3H)-one derivatives with trimethyl orthoformate and acetic anhydride at elevated temperatures to form the methoxymethylene intermediate.
- Treat the resulting intermediate with sodium methylate and a halogenated pyrimidine compound in a suitable solvent system.
- Isolate the final phenoxyprimidine product through crystallization or chromatographic purification to ensure high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this patented process offers profound commercial advantages that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of chemical enterprises. By shifting away from hazardous and expensive reagents like diazomethane, manufacturers can realize significant cost reductions in raw material procurement and waste disposal. The stability of the intermediates produced allows for longer storage times and more flexible logistics planning, reducing the risk of supply disruptions due to material degradation. Furthermore, the use of commodity chemicals such as acetic anhydride and trimethyl orthoformate ensures that the supply chain is not dependent on niche suppliers who might face capacity constraints. This diversification of the supply base enhances negotiation leverage and protects against price volatility in the global chemical market. For procurement managers, this translates to a more predictable budget and a lower total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and hazardous methylation agents drastically simplifies the downstream purification process, removing the need for expensive heavy metal scavenging resins or complex extraction protocols. This streamlining of the workflow reduces labor hours and utility consumption, leading to substantial operational savings. Additionally, the higher selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which increases the overall yield of the final active ingredient. These efficiency gains compound over large production volumes, resulting in a highly competitive cost structure that can be passed on to customers or retained as margin. The qualitative improvement in process economics makes this technology an attractive option for companies looking to optimize their manufacturing footprint without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are globally available and produced by multiple vendors significantly de-risks the supply chain against geopolitical or logistical shocks. The reagents required for this synthesis, such as sodium methylate and various organic solvents, are staple chemicals in the fine chemical industry with robust production networks. This abundance ensures that lead times for raw material delivery remain short and consistent, allowing for just-in-time inventory strategies that reduce working capital requirements. Moreover, the stability of the synthesized intermediates means that safety stock levels can be managed more effectively, providing a buffer against unexpected demand spikes. For supply chain heads, this reliability is crucial for maintaining service levels to downstream pharmaceutical and agrochemical clients who depend on uninterrupted material flow.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, operating at atmospheric or slightly elevated pressures that do not require specialized high-pressure vessels. This compatibility with standard glass-lined or stainless steel reactors facilitates a smooth transition from pilot scale to commercial production without the need for major capital expenditure on new equipment. From an environmental perspective, the process generates less hazardous waste compared to traditional methods, aligning with increasingly stringent global regulations on chemical emissions and disposal. The reduced environmental footprint not only lowers compliance costs but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions. Companies adopting this green chemistry approach position themselves as responsible partners in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and industry best practices. These insights are designed to clarify the operational benefits and technical feasibility for decision-makers evaluating this route for their own production lines. Understanding these details helps in assessing the fit of this technology within existing manufacturing frameworks and regulatory strategies. The answers provided reflect the consensus on the advantages of orthoformate chemistry over legacy methylation techniques.
Q: How does this process improve safety compared to conventional diazomethane methods?
A: This patented methodology eliminates the need for hazardous diazomethane reagents, replacing them with stable trimethyl orthoformate and acetic anhydride, significantly reducing operational risks and explosion hazards in the manufacturing facility.
Q: What are the purity capabilities for the 3-(α-methoxymethylene)benzofuran-2(3H)-one intermediate?
A: The process allows for the isolation of the key intermediate in substantially pure states, often exceeding 85% purity directly from the reaction mixture, with further recrystallization steps available to meet stringent pharmaceutical or agrochemical grade specifications.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize common industrial solvents like toluene and methyl acetate and operate at manageable temperatures and pressures, making the technology highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(α-methoxymethylene)benzofuran-2(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the evolving demands of the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity 3-(α-methoxymethylene)benzofuran-2(3H)-one derivatives that meet stringent purity specifications required for next-generation fungicides. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch, guaranteeing consistency and reliability for our partners. By integrating the principles of Patent CN1040749C into our manufacturing repertoire, we offer a supply solution that balances cost-efficiency with uncompromising quality standards.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio and streamline your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of partnering with a leader in fine chemical synthesis. Let us help you secure a sustainable and competitive advantage in the production of high-value agrochemical intermediates.
