Revolutionizing Active Vitamin D Production: High-Yield Microbial Conversion for Global Supply Chains
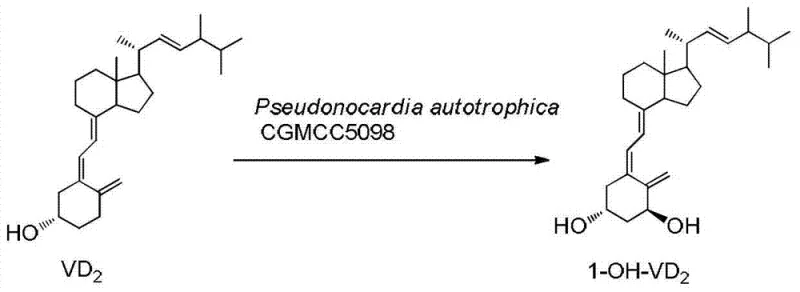
The pharmaceutical landscape for treating osteoporosis and chronic renal failure is undergoing a significant transformation driven by the innovations detailed in patent CN103451242A. This pivotal intellectual property introduces a groundbreaking industrial preparation method for 1-alpha-hydroxyvitamin D2 and D3, the critical precursors for active drugs like paricalcitol and doxercalciferol. Unlike traditional multi-step organic syntheses that struggle with low efficiency and hazardous reagents, this technology leverages the unique metabolic capabilities of the microbial strain Pseudonocardia autotrophica CGMCC No.5098. By enabling a direct, one-step microbial conversion of vitamin D substrates, this process achieves a remarkable biotransformation yield of 40-50%, effectively bypassing the complex protection and deprotection sequences that have long plagued chemical manufacturers. For global supply chain leaders and R&D directors, this represents not just a laboratory curiosity, but a viable, high-efficiency pathway to secure the supply of high-purity vitamin D pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1-alpha-hydroxyvitamin D compounds has been fraught with significant technical and economic challenges that hinder cost-effective manufacturing. Conventional chemical routes typically require a tedious sequence of approximately six distinct reaction steps, involving esterification protection, ring closure, oxidation, and subsequent ring opening. A major bottleneck in these traditional pathways is the necessity of using toxic selenium dioxide ($SeO_2$) for the critical oxidation step, which introduces severe environmental hazards and necessitates expensive waste disposal protocols. Furthermore, the chemical direct hydroxylation at the 1-alpha position is notoriously difficult to control, often resulting in poor regioselectivity and a dismal overall yield of only about 10%. These inefficiencies compound the final cost of goods sold (COGS) and create substantial supply chain vulnerabilities due to the reliance on hazardous reagents and complex purification processes.
The Novel Approach
In stark contrast, the novel biocatalytic approach disclosed in the patent data offers a streamlined, environmentally benign alternative that fundamentally reshapes the production economics. By utilizing the specific strain Pseudonocardia autotrophica CGMCC No.5098, manufacturers can achieve direct hydroxylation at the 1-position of vitamin D in a single fermentation step. This biological catalyst exhibits exceptional regioselectivity, eliminating the need for cumbersome group protection strategies that characterize chemical synthesis. The process operates under mild conditions—typically between 22-30°C and a neutral pH of 6.5-7.5—which significantly reduces energy consumption compared to harsh chemical reactions. Most critically for procurement teams, this method boosts the total yield to an impressive 40-50%, representing a five-fold increase in material efficiency over conventional methods, thereby offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cytochrome P450-Mediated Hydroxylation
The core scientific breakthrough of this technology lies in the enzymatic machinery of the actinomycete strain, specifically its ability to produce highly active cytochrome P450 enzymes. These heme-thiolate proteins function as mono-oxygenases, capable of activating molecular oxygen to insert a hydroxyl group directly onto the sterol backbone of the vitamin D substrate. In the context of this patent, the microbial cells act as microscopic factories where the cytochrome P450 system recognizes the specific stereochemistry of vitamin D2 or D3. The enzyme facilitates the abstraction of a hydrogen atom from the C-1 alpha position, followed by the rebound of a hydroxyl radical, resulting in the precise formation of the 1-alpha-hydroxy derivative. This enzymatic precision ensures that the product is formed with high stereoselectivity, minimizing the generation of unwanted isomers that would otherwise complicate downstream purification and reduce the effective purity of the final API intermediate.
From an impurity control perspective, this biocatalytic mechanism offers inherent advantages over chemical oxidation. Chemical methods often generate a broad spectrum of byproducts due to non-selective radical reactions or over-oxidation, requiring rigorous and yield-loss-inducing chromatographic separations. In the microbial system, the enzyme's active site acts as a molecular sieve, physically restricting access to only the target carbon atom. While side reactions can still occur in fermentation, the profile is generally cleaner and more predictable. The patent specifies that the addition of solubilizing agents like cyclodextrins (beta, gamma, or methylated derivatives) further enhances the bioavailability of the hydrophobic vitamin D substrate to the enzyme. This optimization ensures that the reaction kinetics favor the desired transformation, allowing for substrate concentrations of 1-15g/L while maintaining high conversion rates, ultimately delivering a crude product that is far easier to purify to stringent pharmaceutical standards.
How to Synthesize 1-alpha-Hydroxyvitamin D Efficiently
Implementing this biocatalytic route requires precise control over fermentation parameters to maximize the expression of the hydroxylase enzymes and the stability of the substrate. The process begins with the cultivation of the Pseudonocardia strain in a optimized medium containing carbon sources like glucose and nitrogen sources such as soybean cake powder. Once the biomass reaches the appropriate growth phase, typically after 3 days, the vitamin D substrate is introduced in a solubilized form using surfactants like Triton X-100 and cyclodextrins to overcome solubility limits in the aqueous media. The detailed standardized synthesis steps, including specific media formulations, sterilization protocols, and extraction procedures, are outlined in the guide below.
- Cultivate Pseudonocardia autotrophica CGMCC No.5098 in a nutrient-rich medium containing glucose, soybean cake powder, and mineral salts at 27°C for 3-4 days.
- Prepare the substrate solution by dissolving Vitamin D2 or D3 with cyclodextrin solubilizers and surfactants in ethanol, then add to the fermentation broth.
- Continue incubation for 1-10 days to allow regioselective hydroxylation, followed by extraction, concentration, and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical synthesis to this microbial conversion process offers transformative benefits that extend far beyond simple yield improvements. The elimination of toxic selenium dioxide not only aligns with increasingly strict global environmental regulations but also removes the logistical and financial burdens associated with handling and disposing of hazardous heavy metal waste. This shift significantly simplifies the manufacturing workflow, reducing the operational complexity and the risk of production stoppages due to regulatory compliance issues. Furthermore, the reduction from a six-step chemical sequence to a single biotransformation step drastically shortens the production cycle time, enhancing the agility of the supply chain to respond to market fluctuations in demand for osteoporosis treatments.
- Cost Reduction in Manufacturing: The economic implications of increasing the total yield from approximately 10% to 40-50% are profound, as it effectively quintuples the output from the same amount of starting raw material. By removing the need for multiple protection and deprotection reagents, as well as expensive oxidants, the direct material costs are substantially lowered. Additionally, the simplified downstream processing requires fewer unit operations, leading to significant savings in labor, energy, and solvent consumption. This comprehensive reduction in operational expenditure allows for a much more competitive pricing structure for high-purity vitamin D intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: Reliance on complex chemical syntheses often introduces multiple points of failure, where a delay in any single step or reagent can halt the entire production line. This biocatalytic method relies on robust, renewable biological catalysts and readily available commodity chemicals like glucose and vitamin D2/D3, ensuring a stable and continuous supply of raw materials. The mild reaction conditions reduce the wear and tear on equipment and minimize the risk of safety incidents, thereby ensuring consistent production uptime. This reliability is crucial for maintaining long-term contracts with pharmaceutical partners who require guaranteed delivery schedules for their active ingredient supply.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard fermentation technologies that can be easily expanded from laboratory shake flasks to industrial-scale bioreactors. The use of biodegradable nutrients and the absence of persistent toxic byproducts make the waste stream much easier to treat, facilitating compliance with modern green chemistry initiatives. This environmental friendliness not only reduces the carbon footprint of the manufacturing process but also future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational viability and sustainability for the production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is essential for making informed decisions about integrating this high-efficiency pathway into your existing production portfolio.
Q: What is the primary advantage of this biocatalytic method over traditional chemical synthesis?
A: The primary advantage is the drastic reduction in synthetic steps from approximately six chemical steps to a single biotransformation step, increasing total yield from roughly 10% to 40-50% while eliminating toxic selenium dioxide reagents.
Q: How does the process ensure regioselectivity at the 1-alpha position?
A: The process utilizes the specific enzymatic activity of cytochrome P450 enzymes produced by the Pseudonocardia autotrophica strain, which naturally targets the 1-alpha position of the vitamin D sterol backbone with high stereoselectivity.
Q: Is this manufacturing route suitable for large-scale industrial production?
A: Yes, the method employs standard fermentation techniques using accessible raw materials like Vitamin D2/D3 and common nutrients, making it highly scalable for commercial manufacturing of osteoporosis treatment intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-alpha-Hydroxyvitamin D Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced biocatalytic technologies to secure a competitive edge in the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-alpha-hydroxyvitamin D intermediate meets the highest international standards for safety and efficacy required by regulatory bodies worldwide.
We invite you to collaborate with us to leverage this innovative microbial conversion technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you build a more resilient and cost-effective supply chain for your vitamin D products.
