Scalable Production of High-Purity Optically Active 1-Amino-2-Vinylcyclopropane Carboxylic Acid Esters for Antiviral APIs
The pharmaceutical industry continuously demands robust and scalable routes for complex chiral intermediates, particularly those serving as building blocks for next-generation antiviral therapeutics. Patent CN103402972A introduces a significant advancement in the manufacturing of optically active 1-amino-2-vinylcyclopropane carboxylic acid esters, a critical scaffold in medicinal chemistry. This technology addresses the longstanding challenges associated with achieving high enantiomeric excess (e.e.) without resorting to cumbersome enzymatic processes or expensive chromatographic separations. By leveraging a combination of asymmetric phase-transfer catalysis and classical diastereomeric salt resolution, the method ensures a reliable supply of high-purity intermediates essential for the synthesis of potent bioactive compounds.
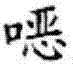
For R&D directors and process chemists, the ability to source high-purity pharmaceutical intermediates with defined stereochemistry is paramount for regulatory compliance and drug efficacy. The disclosed method utilizes readily available starting materials and avoids the substrate specificity limitations often encountered with biocatalysts. This approach not only simplifies the synthetic pathway but also enhances the reproducibility of the manufacturing process, making it an ideal candidate for commercial scale-up of complex cyclopropanes. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to offer superior quality control and consistency to our global partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active 1-amino-2-vinylcyclopropane carboxylic acid esters has relied heavily on enzymatic kinetic resolution. While effective in laboratory settings, these biological methods present significant drawbacks for industrial application. A primary limitation is the strict substrate specificity of the enzymes, which often necessitates additional synthetic steps, such as transesterification, to convert ethyl esters into methyl esters before the enzymatic reaction can proceed. This adds unnecessary complexity, increases raw material consumption, and extends the overall production timeline. Furthermore, enzymatic processes can be sensitive to reaction conditions, requiring precise control of pH and temperature, which complicates cost reduction in API manufacturing efforts at a large scale.
Another critical issue with conventional resolution methods is the difficulty in achieving consistently high optical purity in a single pass. Traditional chemical resolutions often suffer from low yields or require multiple recrystallization cycles that erode overall process efficiency. The reliance on specific enzyme preparations also introduces supply chain vulnerabilities, as biological reagents can vary between batches and have limited shelf lives. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for pharmaceutical companies aiming to accelerate their drug development pipelines while maintaining strict quality standards.
The Novel Approach
The innovative method described in the patent overcomes these hurdles by employing a chemically driven resolution strategy using optically active organic acids such as tartaric acid or camphorsulfonic acid. This approach eliminates the need for enzymatic steps and substrate modification, allowing for the direct resolution of the ethyl ester form. The process involves reacting the racemic or enriched amine ester with a chiral acid to form diastereomeric salts, which exhibit different solubility properties in specific solvent systems. By carefully selecting solvents like toluene mixed with alcohols, one diastereomer can be preferentially crystallized and isolated with high optical purity.
This chemical resolution is highly versatile and robust, capable of upgrading the optical purity from moderate levels (e.g., 70-80% e.e.) to exceptional levels exceeding 99% e.e. through simple recrystallization. The use of stable, non-biological resolving agents ensures consistent performance across batches and simplifies storage and handling requirements. Moreover, the method is compatible with a wide range of ester groups and can be seamlessly integrated into existing manufacturing infrastructure. This flexibility makes it a superior choice for reducing lead time for high-purity amino acid esters, providing a streamlined path from raw materials to GMP-grade intermediates ready for downstream coupling reactions.
Mechanistic Insights into Asymmetric Phase-Transfer Catalysis and Resolution
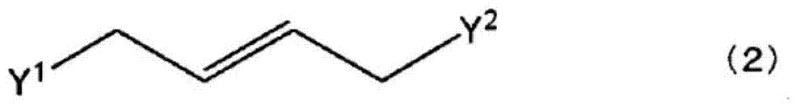
The synthesis begins with an asymmetric cyclopropanation reaction, where an N-arylmethylene glycine ester reacts with a 1,4-dihalo-2-butene derivative in the presence of a chiral quaternary ammonium salt catalyst. This phase-transfer catalysis (PTC) step is crucial for establishing the initial stereochemistry of the cyclopropane ring. The chiral catalyst, often a Maruoka-type spiro ammonium salt, creates a chiral environment at the interface of the organic and aqueous phases, directing the attack of the carbanion to favor one enantiomer over the other. This step typically yields the intermediate imine with moderate to good enantiomeric excess, setting the stage for the subsequent purification.
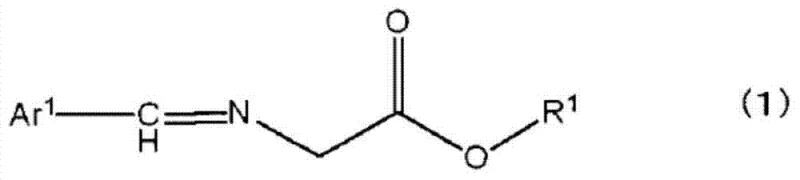
Following the cyclopropanation, the imine group is hydrolyzed under mild acidic conditions to reveal the free amine functionality, resulting in the 1-amino-2-vinylcyclopropane carboxylic acid ester. The true power of this technology lies in the subsequent resolution step. By introducing an optically active acid like L-tartaric acid or D-10-camphorsulfonic acid, the amine forms a diastereomeric salt. Due to the distinct three-dimensional arrangement of atoms, the (1R, 2S)-isomer salt and the (1S, 2R)-isomer salt possess different lattice energies and solubilities. In a optimized solvent mixture, the less soluble salt precipitates out of the solution, effectively separating the desired enantiomer from its mirror image. This thermodynamic control allows for the removal of trace impurities and the enrichment of the target isomer to pharmaceutical-grade specifications.
How to Synthesize Optically Active 1-Amino-2-Vinylcyclopropane Carboxylic Acid Ester Efficiently
Implementing this synthesis requires careful attention to solvent selection and temperature control to maximize yield and optical purity. The process generally involves dissolving the amine ester in a solvent like toluene, adding the chiral resolving agent, and inducing crystallization through cooling or anti-solvent addition. The isolation of the diastereomeric salt is followed by a basification step to liberate the free amine ester. This standardized approach ensures that the final product meets the rigorous demands of modern drug synthesis, providing a reliable foundation for the production of antiviral agents.
- Perform asymmetric cyclopropanation of N-arylmethylene glycine ester with 1,4-dihalo-2-butene using a chiral quaternary ammonium salt catalyst in a biphasic solvent system.
- Hydrolyze the resulting imine intermediate under acidic conditions to yield the racemic or enriched 1-amino-2-vinylcyclopropane carboxylic acid ester.
- Resolve the ester by forming a diastereomeric salt with optically active tartaric acid or camphorsulfonic acid, followed by crystallization and base treatment to isolate the pure enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this manufacturing method offers substantial strategic benefits that directly impact the bottom line and operational resilience. By shifting away from enzymatic processes, manufacturers can significantly reduce dependency on specialized biological reagents that often come with high price tags and volatile availability. The chemical resolution method utilizes commodity chemicals such as tartaric acid and common organic solvents, which are widely available in the global market. This transition simplifies the sourcing strategy, mitigates the risk of supply disruptions, and facilitates better long-term cost planning for large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of enzymatic steps removes the need for expensive enzyme procurement and the associated buffer systems required for biocatalysis. Furthermore, the ability to process the ethyl ester directly avoids the additional unit operations of transesterification, thereby reducing energy consumption, solvent usage, and labor hours. The high efficiency of the crystallization process also minimizes material loss, leading to improved overall yields and a more economical use of raw materials, which translates into significant cost savings for the final API.
- Enhanced Supply Chain Reliability: Chemical resolving agents are stable solids with long shelf lives, unlike enzymes which may require cold chain logistics and have limited stability. This stability simplifies inventory management and reduces the risk of batch-to-batch variability caused by reagent degradation. The robustness of the chemical process ensures consistent output quality, allowing supply chain managers to forecast production timelines with greater accuracy and reliability, ultimately supporting just-in-time manufacturing models for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard reactor equipment and straightforward workup procedures like filtration and distillation. The solvent systems employed, such as toluene and alcohols, are well-understood and can be efficiently recovered and recycled, minimizing waste generation. This aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, avoiding potential fines and enhancing the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these specialized intermediates. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industrial applications.
Q: Why is diastereomeric salt resolution preferred over enzymatic resolution for this intermediate?
A: Enzymatic resolution often requires specific substrate modifications, such as transesterification from ethyl to methyl esters, adding complexity and cost. Diastereomeric resolution using tartaric or camphorsulfonic acid allows direct processing of the ethyl ester with high optical purity (up to 99% e.e.) without additional enzymatic steps.
Q: What level of optical purity can be achieved with this method?
A: The patented method demonstrates the ability to achieve optical purities exceeding 90% e.e. initially, which can be further upgraded to 99% e.e. or higher through recrystallization of the diastereomeric salt, ensuring suitability for stringent pharmaceutical applications.
Q: Is the chiral catalyst reusable in this synthesis?
A: Yes, the optically active quaternary ammonium salt catalyst operates effectively in phase-transfer conditions. While the patent focuses on the resolution step for final purity, the catalytic asymmetric cyclopropanation step uses low catalyst loading (0.001 to 0.1 molar ratio), enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Amino-2-Vinylcyclopropane Carboxylic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-amino-2-vinylcyclopropane carboxylic acid ester meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges and deliver solutions that accelerate your time to market.
We invite you to collaborate with us to optimize your supply chain for antiviral API manufacturing. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. By partnering with us, you gain access to our deep expertise in process chemistry and our dedication to reliability. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth and innovation goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
