Advanced Ozone-Carbon Monoxide Deprotection Technology for Scalable Beta-Lactam Synthesis
Introduction to Green Deprotection Technologies
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for synthesizing complex heterocyclic intermediates, particularly beta-lactams which serve as the core scaffolds for numerous antibiotics. A critical bottleneck in these syntheses is the removal of protecting groups, specifically the p-methoxyphenyl (PMP) group, which is widely used to mask amide functionality during ring construction. Patent CN113549102B, filed in late 2021, introduces a groundbreaking methodology that addresses the environmental and economic inefficiencies of traditional deprotection routes. By replacing hazardous reducing agents with carbon monoxide following an ozonization step, this technology offers a cleaner, more scalable solution for producing high-purity intermediates like 4-acetoxy-azetidinone derivatives. This report analyzes the technical merits of this invention, demonstrating its value proposition for R&D teams aiming to optimize impurity profiles and supply chain leaders focused on reducing hazardous waste disposal costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of the p-methoxyphenyl protecting group has relied heavily on two primary strategies, both of which present significant drawbacks for large-scale manufacturing. The first approach utilizes Cerium Ammonium Nitrate (CAN) as a stoichiometric oxidant. While CAN is effective and rapid, it generates massive amounts of cerium-containing waste, creating a severe burden on wastewater treatment facilities and inflating the cost of goods due to the high price of the reagent. The second common method involves ozonization followed by reduction with thiourea. Although ozone is a greener oxidant than CAN, the subsequent requirement for thiourea introduces a new set of problems. Thiourea reduction produces substantial sulfur-containing wastewater, which is notoriously difficult and expensive to treat to meet modern environmental discharge standards. Furthermore, the use of thiourea can sometimes lead to sulfur-containing impurities that are challenging to purge from the final active pharmaceutical ingredient, posing a risk to product quality and regulatory approval.
The Novel Approach
The methodology disclosed in patent CN113549102B fundamentally reengineers the post-ozonization workflow to eliminate the need for thiourea entirely. In this novel sequence, the substrate undergoes controlled ozonization to form an oxidation intermediate, which is then subjected to a reduction reaction using carbon monoxide gas. This switch from a solid sulfur-based reductant to a gaseous carbon-based reductant is transformative. Carbon monoxide acts as a clean reducing agent that converts the oxidized intermediate directly to the desired free amine without introducing heteroatom contaminants like sulfur. The process operates under moderate pressures (0.1 to 2 MPa) and temperatures (10 to 90°C), conditions that are readily achievable in standard pharmaceutical autoclaves. By circumventing the generation of sulfur-laden effluent, this approach not only aligns with stricter global environmental regulations but also simplifies the downstream purification process, resulting in consistently high HPLC purities exceeding 99%.
Mechanistic Insights into Ozone-Carbon Monoxide Deprotection
The chemical mechanism underlying this invention involves a sophisticated interplay between oxidative cleavage and carbonyl-mediated reduction. Initially, the electron-rich p-methoxyphenyl ring of the protected amide is attacked by ozone at low temperatures, typically between -30°C and -10°C. This electrophilic attack disrupts the aromaticity of the anisole moiety, generating a reactive oxidation intermediate, likely a quinone-like species or a lactol derivative depending on the specific substitution pattern. The presence of alkaline additives, such as sodium acetate or potassium formate, during this stage is crucial; these buffers stabilize the reaction medium and accelerate the consumption of ozone, ensuring complete conversion of the starting material while minimizing over-oxidation side reactions. Following the formation of this intermediate, the system is quenched to destroy excess ozone, preparing the mixture for the reduction phase.
The subsequent reduction step leverages the unique reactivity of carbon monoxide under pressure. Unlike hydride donors or metal-based reductants, carbon monoxide facilitates a deoxygenation process that restores the amide nitrogen to its free state (or the desired deprotected form) while releasing carbon dioxide as the primary byproduct. This gas evolution drives the reaction forward and leaves no non-volatile residues in the reaction mixture. The absence of heavy metals or sulfur compounds means that the crude product contains significantly fewer inorganic impurities, drastically reducing the load on crystallization and filtration steps. This mechanistic elegance ensures that the final product, such as the critical beta-lactam intermediate 4AA, is obtained with exceptional chemical integrity, free from the trace metal or sulfur contaminants that often plague batches produced via CAN or thiourea routes.
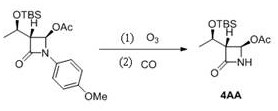
How to Synthesize 4AA Efficiently
The synthesis of (3R, 4R)-3-[(R)-1-tert-butyldimethylsilyloxyethyl]-4-acetoxy-2-azetidinone (4AA) via this patented route represents a benchmark for efficiency in beta-lactam manufacturing. The process begins with the dissolution of the PMP-protected precursor in a lower alcohol solvent, such as methanol, supplemented with a molar equivalent of sodium acetate to buffer the ozonization. The reaction is cooled to approximately -14°C, and ozone is introduced until the substrate is fully consumed. Crucially, the workup involves quenching with sodium thiosulfate to neutralize residual oxidant, followed immediately by the transfer of the mixture to a pressure vessel.
- Dissolve the PMP-protected substrate in an alcohol solvent with an alkaline additive and perform ozonization at -30 to -10°C.
- Quench the reaction mixture with a sulfite-based agent to neutralize excess ozone without using thiourea.
- Transfer the intermediate to an autoclave and react with carbon monoxide at 0.1-2 MPa and 10-90°C to effect deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ozone-carbon monoxide technology translates into tangible operational improvements and risk mitigation. The most immediate impact is the drastic simplification of waste management protocols. By eliminating the use of thiourea, the facility avoids the generation of sulfur-containing wastewater, which often requires specialized biological or chemical treatment processes that are both capital-intensive and operationally complex. This reduction in hazardous waste volume directly correlates to lower disposal fees and a smaller environmental footprint, enhancing the company's sustainability profile. Furthermore, carbon monoxide is a commodity chemical available at a fraction of the cost of specialized reagents like Cerium Ammonium Nitrate or high-purity thiourea, leading to significant raw material cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The replacement of expensive oxidants and reductants with ozone and carbon monoxide creates a leaner cost structure. Ozone is generated on-site from oxygen, eliminating the need to purchase and store large quantities of hazardous oxidizing salts. Similarly, carbon monoxide is inexpensive and widely available. The process also improves atom economy by avoiding the addition of heavy molecular weight reagents that end up as waste. These factors combine to lower the overall cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Reliance on thiourea or CAN exposes the supply chain to volatility in the markets for sulfur chemicals and rare earth metals. By shifting to a process dependent on gases (oxygen/ozone and CO), the manufacturing process becomes less susceptible to raw material shortages or price spikes associated with mined or synthesized solids. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without compromising yield, ensures consistent batch-to-batch performance. This reliability is critical for meeting the rigorous delivery schedules demanded by downstream API manufacturers, reducing the risk of production delays caused by reagent quality issues.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is streamlined because the unit operations involved—ozonization and gas-liquid hydrogenation-style reduction—are well-understood in the fine chemical industry. The equipment required, such as ozone generators and stirred autoclaves, is standard in modern multipurpose plants. Moreover, the process inherently supports green chemistry principles by minimizing waste generation at the source rather than treating it at the end of the pipe. This proactive approach to environmental compliance future-proofs the manufacturing site against tightening regulations, ensuring long-term operational continuity without the need for costly retrofits to wastewater treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this deprotection technology. These answers are derived directly from the experimental data and scope of patent CN113549102B, providing clarity on the feasibility and benefits of the method for potential licensees or manufacturing partners.
Q: Why is the Ozone-CO method superior to the traditional Thiourea reduction?
A: The traditional method requires stoichiometric amounts of thiourea to reduce the ozonized intermediate, generating significant sulfur-containing wastewater that is costly to treat. The patented Ozone-CO method replaces thiourea with carbon monoxide, eliminating this specific sulfur waste stream and reducing raw material costs while maintaining high yields above 80%.
Q: What are the critical safety parameters for the Carbon Monoxide reduction step?
A: The reaction operates under pressure (0.1 to 2 MPa) and elevated temperatures (10 to 90°C). Proper autoclave equipment with gas monitoring is essential. However, the process avoids the use of heavy metal oxidants like Cerium Ammonium Nitrate (CAN), simplifying the safety profile regarding toxic metal handling.
Q: Can this method be applied to substrates other than 4-acetoxy-azetidinones?
A: Yes, the patent claims applicability to a broad range of amides protected by p-methoxyphenyl groups, including those with various alkyl, aryl, and heterocyclic substituents, making it versatile for different pharmaceutical intermediate syntheses beyond just carbapenem precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4AA Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is essential for the future of the pharmaceutical industry. Our technical team has extensively evaluated the ozone-carbon monoxide deprotection strategy described in CN113549102B and confirmed its potential to deliver high-purity beta-lactam intermediates with superior consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of ozonization and high-pressure reduction is maintained safely and effectively at scale. Our stringent purity specifications and rigorous QC labs guarantee that every batch of 4AA or related intermediates meets the exacting standards required for GMP API synthesis.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. By working together, we can achieve significant process optimizations that benefit both the bottom line and the environment. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented method can secure your supply of critical pharmaceutical intermediates.
