Advanced Mild Synthesis of 4-Aminoisoxazole Hydrochloride for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high yield with operational safety, particularly for heterocyclic intermediates like 4-aminoisoxazole hydrochloride. A recent technological breakthrough documented in patent CN112457268A introduces a significantly milder and safer methodology for producing this critical building block, addressing long-standing challenges associated with traditional nitration processes. This innovation replaces the hazardous conventional mixed acid systems with a controlled ammonium nitrate and acetic anhydride protocol, fundamentally altering the risk profile of the manufacturing process. By shifting the reaction conditions to a moderate temperature range of 15-50°C, the new method mitigates the dangers of thermal runaway while simultaneously boosting the overall yield of the nitration step to approximately 76%. For R&D directors and process engineers, this represents a pivotal shift towards greener chemistry that does not compromise on efficiency or purity standards required for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-nitroisoxazole, the key precursor to 4-aminoisoxazole hydrochloride, has relied heavily on aggressive nitration agents such as a mixture of concentrated sulfuric acid and fuming nitric acid. This traditional approach is fraught with significant engineering and safety challenges, primarily due to the intensely exothermic nature of the reaction which makes temperature control difficult on a large scale. The high oxidative potential of fuming nitric acid not only increases the risk of accidental ignition or explosion but also often leads to the degradation of the sensitive isoxazole ring, resulting in substantial byproduct formation and lower yields. Literature data indicates that these older methods typically achieve yields around 53.5%, meaning nearly half of the valuable starting material is lost to side reactions or decomposition. Furthermore, the handling and disposal of large volumes of spent mixed acid create severe environmental burdens and require expensive corrosion-resistant equipment, driving up both capital expenditure and operational costs for manufacturers.
The Novel Approach
In stark contrast, the novel synthesis route described in the patent utilizes a much gentler nitrating system composed of ammonium nitrate dissolved in a mixture of acetic acid and acetic anhydride. This modification allows the nitration to proceed under remarkably mild conditions, with the reaction temperature strictly controlled between 15°C and 50°C, effectively eliminating the risk of thermal runaway. The use of ammonium nitrate as the nitrogen source, activated by the acetic anhydride, provides a steady and manageable release of the nitronium equivalent, ensuring a smoother reaction profile that is far more amenable to industrial scale-up. Experimental results demonstrate that this optimized protocol consistently delivers yields exceeding 76%, representing a dramatic improvement in atom economy and resource utilization compared to the legacy mixed acid process. The subsequent reduction step employs catalytic hydrogenation with palladium on carbon in the presence of hydrochloric acid, ensuring high selectivity for the amine product while forming the stable hydrochloride salt directly.
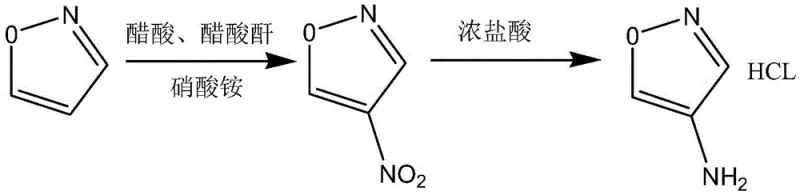
Mechanistic Insights into Mild Nitration and Catalytic Reduction
The core of this technological advancement lies in the generation of the active nitrating species within a non-aqueous, acidic medium that preserves the integrity of the heterocyclic ring. In the acetic anhydride-acetic acid system, ammonium nitrate reacts to form acetyl nitrate or a similar activated nitronium complex in situ, which acts as the electrophile attacking the electron-rich positions of the isoxazole ring. Unlike the chaotic and violent environment created by sulfuric/fuming nitric acid mixtures, this buffered system moderates the electrophilicity of the nitrating agent, favoring mono-nitration at the 4-position while minimizing poly-nitration or oxidative ring cleavage. The batch-wise addition of ammonium nitrate further refines this control, preventing local hotspots of high reagent concentration that could trigger uncontrolled exotherms. This precise mechanistic control is crucial for maintaining high purity profiles, as it significantly reduces the formation of tarry byproducts and isomeric impurities that are notoriously difficult to separate in downstream processing.
Following the nitration, the transformation of 4-nitroisoxazole to the final amine salt involves a heterogeneous catalytic hydrogenation mechanism that is equally critical for product quality. The use of 5% palladium on carbon (Pd/C) provides a high surface area for the adsorption of hydrogen and the nitro substrate, facilitating the six-electron reduction of the nitro group to the primary amine. Conducting this reaction in ethanol with concentrated hydrochloric acid serves a dual purpose: the acid protonates the emerging amine immediately to form the hydrochloride salt, which prevents over-reduction or side reactions of the free base, and it helps solubilize the intermediate. The reaction is conducted under elevated pressure (1.0-1.5 MPa) and moderate heat (40-50°C) to overcome kinetic barriers without compromising the stability of the isoxazole moiety. Finally, cooling the reaction mixture to 0-10°C induces crystallization of the product, allowing for efficient isolation via filtration and washing with glacial ethanol to remove residual catalyst and mother liquor impurities.
How to Synthesize 4-Aminoisoxazole Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and addition rates to maximize the benefits of the mild conditions. The process begins with dissolving isoxazole in the acetic acid/anhydride solvent matrix, followed by the controlled, batch-wise introduction of ammonium nitrate to manage the reaction exotherm effectively. Once the nitration is complete, the mixture is quenched into ice water to precipitate or allow extraction of the 4-nitroisoxazole intermediate, which is then subjected to the hydrogenation step. Detailed standard operating procedures regarding specific flow rates, agitation speeds, and filtration parameters are essential for reproducibility and safety.
- Dissolve isoxazole in a mixed solvent of acetic acid and acetic anhydride, then add ammonium nitrate in batches under controlled temperature (15-50°C) to synthesize 4-nitroisoxazole.
- Quench the nitration reaction into ice water, extract with ethyl acetate, and concentrate the organic phase to isolate the intermediate 4-nitroisoxazole.
- Reduce 4-nitroisoxazole using 5% palladium carbon catalyst in ethanol with concentrated hydrochloric acid under pressure (1.0-1.5 MPa) at 40-50°C, then cool and filter to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method offers profound strategic advantages that extend beyond simple yield improvements. By transitioning away from hazardous fuming nitric acid and concentrated sulfuric acid, facilities can significantly reduce their regulatory compliance burden and lower the costs associated with hazardous waste disposal and specialized storage infrastructure. The inherent safety of the mild nitration process minimizes the risk of production stoppages due to safety incidents, thereby ensuring a more reliable and continuous supply of this critical pharmaceutical intermediate. Furthermore, the substantial increase in yield directly translates to reduced raw material consumption per kilogram of finished product, driving down the variable cost of goods sold without requiring capital-intensive new equipment.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous mixed acids, combined with the higher yield of the nitration step, creates a compelling economic case for switching to this new route. The process avoids the need for complex neutralization steps and extensive wastewater treatment required for spent sulfuric acid, leading to significant operational expenditure savings. Additionally, the use of common solvents like ethyl acetate and ethanol simplifies solvent recovery and recycling loops, further enhancing the overall cost efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of the mild reaction conditions makes the process less susceptible to variations in ambient temperature or minor fluctuations in utility supply, ensuring consistent batch-to-batch quality. Since the reagents involved, such as ammonium nitrate and acetic anhydride, are widely available commodity chemicals, the supply chain is less vulnerable to the geopolitical or logistical constraints often associated with specialized corrosive acids. This stability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock.
- Scalability and Environmental Compliance: The温和 nature of the reaction facilitates easier scale-up from pilot plant to commercial production, as the heat removal requirements are far less demanding than those of traditional nitration. This scalability ensures that suppliers can rapidly ramp up production to meet surging market demand for downstream APIs. Moreover, the reduced generation of acidic waste and the avoidance of heavy metal contaminants (beyond the recoverable Pd/C catalyst) align perfectly with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-aminoisoxazole hydrochloride synthesized via this advanced mild method. These insights are derived directly from the patent specifications and practical engineering considerations to assist decision-makers in evaluating the feasibility of this route for their specific supply chains.
Q: Why is the ammonium nitrate/acetic anhydride system preferred over traditional mixed acid nitration?
A: Traditional mixed acid nitration using sulfuric and fuming nitric acid is highly exothermic and poses significant safety risks, including reaction runaway. The novel method utilizes a milder acetic anhydride-acetic acid system with ammonium nitrate, operating at lower temperatures (15-50°C) to drastically reduce hazard levels while improving yield from approximately 53% to over 76%.
Q: What are the critical parameters for the catalytic reduction step?
A: The reduction of 4-nitroisoxazole requires precise control of pressure and temperature to ensure complete conversion without degrading the isoxazole ring. The optimal conditions involve heating to 40-50°C under a hydrogen pressure of 1.0-1.5 MPa using 5% palladium on carbon as the catalyst in an ethanol solvent system containing concentrated hydrochloric acid.
Q: How does this synthesis route impact industrial scalability?
A: By eliminating the need for hazardous fuming nitric acid and managing the exotherm through batch addition of ammonium nitrate, the process becomes significantly safer and easier to control on a large scale. The simplified workup procedure involving ice water quenching and standard extraction further enhances its suitability for continuous or batch industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminoisoxazole Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for the long-term viability of pharmaceutical supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild nitration technology can be fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-aminoisoxazole hydrochloride meets the exacting standards required for GMP pharmaceutical manufacturing. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry with precision, delivering intermediates that facilitate smoother downstream synthesis for our clients.
We invite you to collaborate with us to optimize your sourcing strategy and leverage these technological advancements for your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and cost structure.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
