Advanced Synthesis of 8-Methyl-3-Trifluoromethyl Imidazopyrazine for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for complex heterocyclic intermediates, and patent CN113620957A presents a significant breakthrough in the preparation of 8-methyl-3-(trifluoromethyl)imidazo[1,5-a]pyrazine. This specific compound serves as a critical building block in the development of advanced therapeutic agents, necessitating a manufacturing process that balances high purity with operational safety and cost efficiency. The disclosed technology fundamentally shifts the paradigm from traditional, hazardous methods involving toxic cyanides and high-pressure hydrogenation to a more controlled radical chlorination and cyclization strategy. By utilizing 2,3-dimethylpyrazine as an inexpensive starting material and employing N-chlorosuccinimide (NCS) with precise batch feeding, the process achieves superior control over impurity profiles, specifically keeping disubstituted byproducts below critical thresholds. This innovation not only enhances the chemical quality of the intermediate but also aligns with modern green chemistry principles by eliminating heavy metal catalysts and corrosive reagents like phosphorus oxychloride. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable supply chain for high-purity pharmaceutical intermediates while mitigating regulatory and environmental risks associated with legacy synthesis methods.
The Limitations of Conventional Methods vs. The Novel Approach
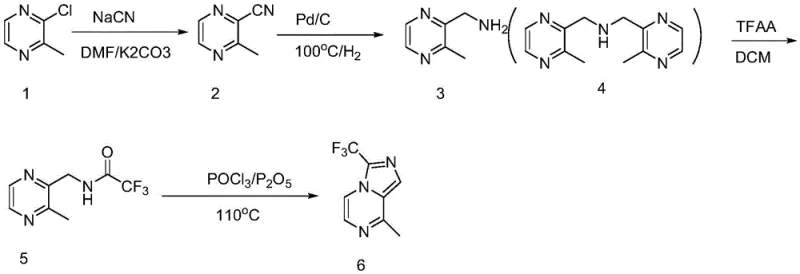
The Limitations of Conventional Methods
Historically, the synthesis of aminomethyl pyrazine derivatives has relied heavily on routes that pose significant safety and environmental challenges, often starting from expensive precursors like 2-chloro-3-methylpyrazine. Traditional methodologies frequently employ sodium cyanide for nucleophilic substitution, introducing extreme toxicity risks that require specialized containment and waste treatment infrastructure, thereby inflating operational costs. Furthermore, these legacy processes typically necessitate high-pressure hydrogenation steps using palladium on carbon catalysts, which not only increases the capital expenditure for equipment but also introduces the risk of disubstituted coupling impurities that are difficult to remove. The use of phosphorus oxychloride for subsequent ring-closing reactions generates substantial amounts of corrosive hydrogen chloride gas, leading to equipment degradation and complex post-reaction neutralization procedures. These factors collectively result in a manufacturing process that is fragile, expensive, and increasingly non-compliant with tightening global environmental regulations, making it unsuitable for sustainable commercial scale-up in modern facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a strategic radical chlorination of 2,3-dimethylpyrazine, which is a significantly more affordable and accessible raw material compared to chlorinated pyrazines. By initiating the reaction with AIBN and carefully controlling the addition of NCS in batches, the process effectively suppresses the formation of disubstituted impurities, ensuring a crude product yield that approaches theoretical maximums without compromising quality. This method bypasses the need for toxic cyanide reagents and high-pressure hydrogenation entirely, replacing them with a Gabriel-type substitution using phthalimide followed by hydrazine hydrate deprotection under atmospheric conditions. The final cyclization step employs polyphosphoric acid instead of phosphorus oxychloride, which drastically reduces the generation of hazardous waste gases and simplifies the workup procedure. This comprehensive re-engineering of the synthetic pathway results in a process that is not only safer and greener but also economically superior, offering a compelling value proposition for manufacturers seeking to optimize their production costs and supply chain resilience.
Mechanistic Insights into NCS-Initiated Radical Chlorination and Cyclization
The core chemical innovation lies in the precise control of the radical chlorination mechanism, where the interaction between N-chlorosuccinimide and azodiisobutyronitrile generates chlorine radicals that selectively target the methyl group of the pyrazine ring. The patent emphasizes the critical importance of batch feeding the chlorinating agent to maintain a low concentration of reactive radicals, which kinetically favors monochlorination over the thermodynamically possible disubstitution. This kinetic control is further enhanced by the selection of chloroform as the solvent, which provides an optimal medium for the radical propagation steps while facilitating the subsequent precipitation of impurities during the workup phase. The reaction temperature is meticulously managed between 50°C and 70°C to ensure sufficient energy for radical initiation without promoting thermal decomposition or side reactions that could compromise the integrity of the pyrazine core. By understanding these mechanistic nuances, process chemists can replicate the high selectivity observed in the patent examples, ensuring that the intermediate 2-chloromethyl-3-methylpyrazine is produced with minimal contamination from di-chlorinated species.
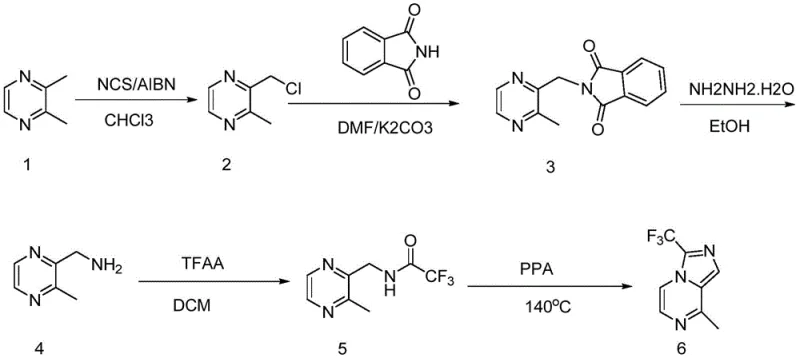
Following the chlorination, the transformation of the chloromethyl group into an aminomethyl functionality is achieved through a robust nucleophilic substitution and deprotection sequence that avoids the pitfalls of direct amination. The use of phthalimide as a nitrogen source allows for a clean substitution reaction in the presence of potassium carbonate, forming a stable phthalimide intermediate that protects the amine from over-alkylation or oxidation. Subsequent treatment with hydrazine hydrate efficiently cleaves the phthalimide protecting group under mild reflux conditions, releasing the free amine without affecting the sensitive pyrazine ring structure. The final ring closure to form the imidazo[1,5-a]pyrazine core is mediated by polyphosphoric acid, which acts as both a dehydrating agent and a catalyst to promote the intramolecular cyclization of the trifluoroacetylated intermediate. This mechanistic pathway ensures that the trifluoromethyl group is incorporated securely into the heterocyclic system, resulting in a final product with high structural fidelity and purity levels that exceed 97% as confirmed by HPLC analysis.
How to Synthesize 8-Methyl-3-(Trifluoromethyl)Imidazo[1,5-a]Pyrazine Efficiently
The synthesis of this high-value pharmaceutical intermediate requires strict adherence to the optimized reaction conditions outlined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the dissolution of 2,3-dimethylpyrazine in chloroform, followed by the controlled addition of NCS and AIBN under reflux, where temperature monitoring is critical to prevent runaway exotherms during the radical initiation phase. After the chlorination is complete, the crude chloromethyl intermediate is carried forward without extensive purification into the substitution step with phthalimide in DMF, where temperature control below 60°C is essential to manage gas evolution and prevent bumping. The subsequent deprotection and trifluoroacetylation steps must be performed with careful attention to moisture exclusion and acid gas scrubbing, particularly during the addition of trifluoroacetic anhydride which generates significant fumes. Finally, the cyclization in polyphosphoric acid requires gradient heating and cooling protocols to manage viscosity and ensure complete conversion, followed by a precise pH adjustment during the quench to isolate the product as a high-purity crystalline solid. Detailed standardized synthesis steps see the guide below.
- Perform radical monochlorination of 2,3-dimethylpyrazine using NCS and AIBN in chloroform with batch feeding to control impurities.
- Execute a Gabriel-type substitution with phthalimide followed by hydrazine hydrate deprotection to yield the aminomethyl intermediate.
- Complete the synthesis via trifluoroacetylation and polyphosphoric acid-mediated cyclization at controlled temperatures to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic advantages by fundamentally altering the cost structure and risk profile of the intermediate's production. The elimination of expensive starting materials like 2-chloro-3-methylpyrazine and the removal of precious metal catalysts such as palladium on carbon directly translate into a significantly reduced bill of materials, allowing for more competitive pricing in long-term supply agreements. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces the capital intensity required for manufacturing, enabling production in a wider range of facilities and thereby enhancing supply chain redundancy and continuity. The replacement of toxic cyanide and corrosive phosphorus oxychloride with safer reagents minimizes the regulatory burden and waste disposal costs, contributing to a more sustainable and environmentally compliant operation that aligns with corporate ESG goals. These qualitative improvements collectively create a more resilient supply chain capable of withstanding market fluctuations and regulatory changes while delivering consistent quality to downstream pharmaceutical customers.
- Cost Reduction in Manufacturing: The shift to cheaper raw materials and the removal of expensive catalysts drastically lower the direct production costs without the need for complex economic modeling to prove viability. By eliminating the requirement for high-pressure reactors and specialized cyanide handling infrastructure, the process reduces both capital expenditure and ongoing operational maintenance costs significantly. The simplified workup procedures resulting from the use of polyphosphoric acid also decrease the consumption of neutralizing agents and reduce the volume of hazardous waste requiring treatment. These factors combine to create a leaner manufacturing process that offers substantial cost savings potential, making the intermediate more affordable for generic drug developers and innovators alike.
- Enhanced Supply Chain Reliability: Sourcing 2,3-dimethylpyrazine is generally more stable and less prone to geopolitical supply disruptions compared to specialized chlorinated pyrazines, ensuring a more reliable flow of starting materials. The simplified reaction conditions reduce the likelihood of batch failures due to equipment malfunction or operator error, leading to higher overall equipment effectiveness and consistent output volumes. Additionally, the reduced hazard profile of the reagents facilitates easier transportation and storage, minimizing logistical bottlenecks that often plague the supply of dangerous chemicals. This enhanced reliability ensures that pharmaceutical manufacturers can maintain their production schedules without the fear of unexpected interruptions caused by raw material shortages or safety incidents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with batch feeding strategies and atmospheric pressure reactions that translate easily from pilot plant to multi-ton commercial production without significant re-engineering. The reduction in hazardous waste generation and the elimination of toxic reagents simplify the environmental permitting process, allowing for faster ramp-up of new production lines in regulated markets. The use of polyphosphoric acid also mitigates the risk of equipment corrosion, extending the lifespan of reactor vessels and reducing the frequency of costly replacements or repairs. This combination of scalability and compliance makes the technology an ideal candidate for long-term industrial adoption, supporting the growing demand for this critical pharmaceutical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and advantageous effects described in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing portfolios or for qualifying new suppliers for critical material needs. The information provided here serves as a foundational reference for further technical discussions and feasibility studies.
Q: How does this new method improve safety compared to conventional cyanide-based routes?
A: The novel process eliminates the use of highly toxic sodium cyanide and high-pressure hydrogenation, replacing them with safer radical chlorination and atmospheric pressure reactions, significantly reducing operational hazards and environmental waste.
Q: What is the expected purity level of the final intermediate using this protocol?
A: The optimized protocol utilizing polyphosphoric acid for ring closure and strict temperature control during chlorination consistently achieves HPLC purity levels exceeding 97%, meeting stringent pharmaceutical standards.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method uses cheap starting materials like 2,3-dimethylpyrazine and avoids expensive catalysts like palladium on carbon, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Methyl-3-(Trifluoromethyl)Imidazo[1,5-a]Pyrazine Supplier
As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses the technical capability to translate this patented laboratory methodology into a robust industrial process, ensuring that clients receive a consistent supply of high-quality intermediates. Our facilities are equipped to handle complex synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications. We understand the critical nature of pharmaceutical supply chains and have invested in rigorous QC labs to verify that every batch meets the required HPLC purity standards and impurity profiles. By leveraging our expertise in process optimization, we can further refine the synthesis to maximize yield and minimize waste, delivering a product that supports your drug development timelines and commercial manufacturing goals.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain for maximum efficiency. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your organization, and ask for specific COA data and route feasibility assessments to validate the quality and scalability of our production capabilities. Our team is ready to provide the technical support and commercial flexibility needed to secure your supply of this vital pharmaceutical intermediate, ensuring that your projects proceed without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
