Advanced Biocatalytic Synthesis of R-3-Hydroxybutyric Acid for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for producing chiral intermediates, and the technology disclosed in patent CN109852593B represents a significant leap forward in this domain. This patent introduces a novel recombinant ketoreductase, specifically engineered through site-directed mutagenesis to exhibit drastically improved catalytic activity for the synthesis of R-3-hydroxybutyric acid (R-3HB) and its various salts. R-3HB is a critical molecule, serving not only as a monomer for biodegradable polyhydroxyalkanoates (PHA) but also as a vital physiological ketone body with therapeutic potential for metabolic disorders and neurological conditions. The core innovation lies in the modification of the CmCR enzyme, where specific amino acid residues at positions 176, 191, and 195 are substituted to create mutants like CmCR-186, which demonstrate superior performance over wild-type variants. 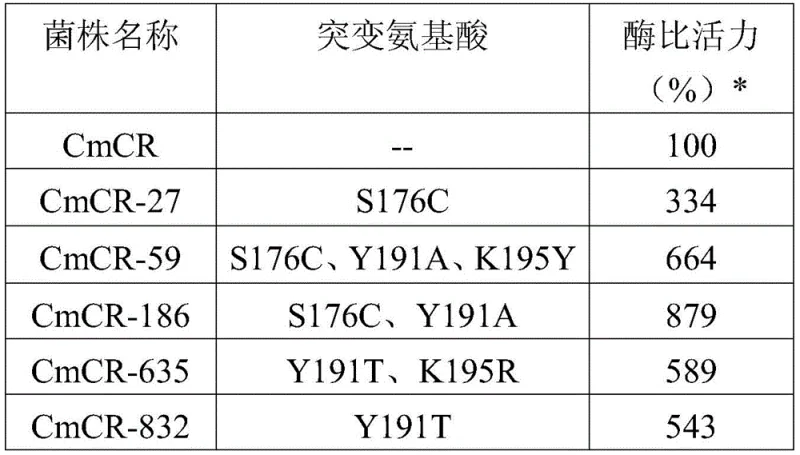 By leveraging this advanced biocatalyst, manufacturers can overcome the traditional limitations of chemical synthesis, such as harsh reaction conditions and poor stereo-selectivity, thereby establishing a more robust foundation for the reliable pharmaceutical intermediates supplier market.
By leveraging this advanced biocatalyst, manufacturers can overcome the traditional limitations of chemical synthesis, such as harsh reaction conditions and poor stereo-selectivity, thereby establishing a more robust foundation for the reliable pharmaceutical intermediates supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 3-hydroxybutyric acid has relied heavily on chemical synthesis routes that are fraught with significant technical and environmental challenges. Traditional methods often involve the Reformatsky reaction, which requires the use of zinc metal and α-haloesters, or the hydration of crotonic acid under acidic or basic conditions. These processes frequently necessitate the use of toxic reagents such as cyanides or heavy metal catalysts, creating substantial hazards for operators and generating difficult-to-treat wastewater streams. Furthermore, chemical synthesis typically results in racemic mixtures, meaning both R and S enantiomers are produced, which necessitates complex and expensive downstream chiral resolution steps to isolate the biologically active R-isomer. The reaction conditions are often extreme, involving high temperatures or strong acids, which can lead to side reactions, lower overall yields, and increased energy consumption. Consequently, the cost reduction in pharmaceutical intermediates manufacturing has been hindered by these inherent inefficiencies and the regulatory burden associated with heavy metal residues and hazardous waste disposal.
The Novel Approach
In stark contrast, the biocatalytic approach detailed in the patent utilizes a highly specific recombinant ketoreductase to achieve asymmetric reduction under mild, environmentally benign conditions. By employing engineered strains of Escherichia coli expressing the mutated CmCR enzyme, the process converts ethyl acetoacetate directly into the desired R-enantiomer with exceptional precision. This method operates at near-neutral pH levels (6.5-7.0) and moderate temperatures (28-30°C), significantly reducing energy requirements and eliminating the need for corrosive reagents. The use of a coupled enzyme system with formate dehydrogenase allows for the efficient regeneration of the necessary cofactor NADH, driving the reaction to completion with high atom economy. This shift from chemical catalysis to enzymatic biocatalysis not only simplifies the process flow but also inherently ensures high optical purity, effectively bypassing the need for chiral separation. For procurement teams, this translates to a streamlined supply chain for high-purity pharmaceutical intermediates that is both safer and more cost-effective in the long run.
Mechanistic Insights into CmCR-Catalyzed Asymmetric Reduction
The extraordinary performance of the novel biocatalyst stems from precise protein engineering targeting the enzyme's active site. Through homology modeling and structural analysis, researchers identified three critical amino acid residues—Serine 176, Tyrosine 191, and Lysine 195—as key determinants of substrate binding and catalytic efficiency. The patent highlights the CmCR-186 mutant, which incorporates substitutions at positions 176 (Serine to Cysteine) and 191 (Tyrosine to Alanine). These specific modifications alter the steric and electronic environment of the active pocket, facilitating a more favorable orientation for the ethyl acetoacetate substrate. The result is a dramatic enhancement in specific activity, with the CmCR-186 mutant exhibiting an 879% increase in catalytic vigor compared to the wild-type enzyme. This mechanistic optimization ensures that the hydride transfer from the NADH cofactor to the ketone substrate occurs with high fidelity, strictly favoring the formation of the R-configuration. Such deep understanding of the structure-activity relationship allows for the rational design of enzymes that are not only faster but also more robust under industrial process conditions.
Beyond mere speed, the engineered enzyme system provides a robust mechanism for impurity control, which is paramount for R&D directors focused on product quality. In chemical synthesis, side reactions such as over-reduction or polymerization can generate difficult-to-remove impurities that compromise the safety profile of the final API. However, the enzymatic route is highly selective, recognizing only the specific ketone functionality of the substrate while leaving other potential reactive groups untouched. The coupling with formate dehydrogenase ensures a continuous supply of reducing equivalents, preventing the accumulation of partially reduced intermediates that could lead to byproduct formation. Furthermore, the biological nature of the catalyst means that the reaction stops cleanly once the substrate is consumed, minimizing the generation of degradation products. This intrinsic selectivity results in a crude product with a much cleaner impurity profile, simplifying the subsequent purification steps such as nanofiltration and crystallization. Ultimately, this leads to a final product with an enantiomeric excess (ee) value exceeding 99.5%, meeting the stringent purity specifications required for clinical applications without extensive post-processing.
How to Synthesize R-3-Hydroxybutyric Acid Efficiently
The implementation of this biocatalytic process involves a series of well-defined steps that bridge laboratory innovation with industrial practicality. The workflow begins with the fermentation of the recombinant host strain to produce the enzyme, followed by the bioconversion reaction where the substrate is transformed into the chiral ester. Subsequent hydrolysis and purification steps yield the final salt forms suitable for commercial distribution. This standardized approach ensures reproducibility and scalability, making it an attractive option for contract development and manufacturing organizations looking to optimize their production pipelines. The detailed standard operating procedures for this synthesis are outlined below to guide technical teams in replicating these high-efficiency results.
- Ferment recombinant E. coli strains expressing CmCR mutants and Formate Dehydrogenase to obtain crude enzyme solutions.
- Perform enzymatic conversion of ethyl acetoacetate using ammonium formate as a hydrogen donor at pH 6.5-7.0 and 28-30°C.
- Hydrolyze the resulting ester, purify via nanofiltration and crystallization to obtain R-3-hydroxybutyrate salts with >99.5% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic technology offers compelling economic and operational benefits that extend far beyond simple yield improvements. The elimination of heavy metal catalysts and toxic reagents fundamentally alters the cost structure of production by removing the need for expensive metal scavenging resins and specialized hazardous waste treatment facilities. This simplification of the downstream processing train significantly reduces the overall manufacturing footprint and operational expenditure. Moreover, the high stability of the engineered bacterial strains ensures consistent batch-to-batch performance, mitigating the risk of production delays caused by catalyst failure or variability. By adopting this route, companies can secure a more resilient supply of critical intermediates, reducing lead time for high-purity pharmaceutical intermediates and enhancing overall supply chain reliability in the face of fluctuating raw material markets.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the purification workflow. Because the enzymatic reaction achieves such high chiral purity (>99.5% ee) directly, the costly and yield-loss-prone step of chiral resolution is entirely eliminated. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the absence of precious metal catalysts removes a significant variable cost component. The high conversion rates mean that less raw material is wasted, further driving down the cost of goods sold. These factors combine to create a substantially more economical manufacturing process compared to traditional chemical synthesis, allowing for better margin protection in competitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized chemical reagents and the regulatory hurdles associated with transporting hazardous materials. This biocatalytic route relies on renewable substrates and biologically derived catalysts that can be produced on-site via fermentation, reducing dependency on external chemical suppliers. The robustness of the CmCR-186 mutant ensures that production can be scaled up without losing efficiency, providing a stable source of material even during periods of high demand. This decentralization of catalyst production and the use of common fermentation infrastructure make the supply chain less vulnerable to geopolitical disruptions or logistics bottlenecks, ensuring a steady flow of materials to downstream customers.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling complex chemical syntheses involving dangerous reagents. The patent data demonstrates successful operation in 7-liter fermentors, indicating a clear path to commercial scale-up of complex pharmaceutical intermediates. From an environmental perspective, the process aligns perfectly with green chemistry principles by operating in aqueous media and generating biodegradable waste. This reduces the regulatory burden on manufacturers, who face increasing pressure to minimize their carbon footprint and eliminate toxic effluents. Compliance with strict environmental standards becomes easier and less costly, protecting the company's reputation and ensuring long-term operational licenses in regions with rigorous ecological regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant ketoreductase technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is crucial for stakeholders evaluating the potential integration of this biocatalytic route into their existing manufacturing portfolios.
Q: How does the CmCR-186 mutant improve production efficiency compared to wild-type enzymes?
A: The CmCR-186 mutant features specific amino acid substitutions (S176C, Y191A) that enhance substrate binding and catalytic turnover, resulting in an 879% increase in enzyme activity towards ethyl acetoacetate compared to the wild type.
Q: What are the chiral purity specifications achievable with this biocatalytic route?
A: This enzymatic process consistently achieves an enantiomeric excess (ee) value of greater than 99.5%, often reaching 99.9%, which eliminates the need for complex and costly chiral separation steps required in chemical synthesis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent demonstrates successful fermentation in 7L fermentors with high cell density and stable enzyme expression, indicating strong potential for scale-up to commercial production volumes while maintaining high yields above 85%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-3-Hydroxybutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route for R-3-hydroxybutyric acid and are fully equipped to bring this technology to commercial fruition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from pilot studies to full-scale manufacturing. Our state-of-the-art facilities include rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to optimize this biocatalytic process, ensuring maximum yield and minimal environmental impact for our global clientele.
We invite you to collaborate with us to unlock the full commercial value of this innovative synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this switch can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-quality R-3-hydroxybutyric acid for your next generation of therapeutic products.
