Revolutionizing Beta-Lactam Production: Safe Rhodium-Catalyzed Cyclization for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for synthesizing critical antibiotic cores, particularly within the beta-lactam class which serves as the backbone for carbapenems and penems. As detailed in patent CN1097424A, a groundbreaking approach has been established for the preparation of beta-lactam compounds through the cyclization of novel aza-cyclobutanone derivatives. This technology represents a paradigm shift from hazardous legacy processes to a safer, more controllable chemical pathway that leverages hypervalent iodine chemistry and transition metal catalysis. For global procurement and R&D teams, understanding this specific mechanistic advancement is crucial, as it directly impacts the safety profile, cost structure, and reliability of the supply chain for high-value antibiotic intermediates. The patent outlines a versatile synthetic route where specific substituents can be manipulated to yield various carbapenam derivatives, ensuring broad applicability across different therapeutic targets.
Furthermore, the strategic implementation of this synthesis route allows manufacturers to bypass the severe safety limitations inherent in older technologies. By replacing unstable diazonium precursors with stable iodo-intermediates, the process significantly mitigates operational risks during large-scale production. This transition not only enhances worker safety but also simplifies the regulatory compliance landscape for manufacturing facilities. Consequently, adopting this methodology positions supply chains to be more resilient against disruptions caused by safety incidents or strict environmental regulations regarding explosive materials. The ability to produce high-purity intermediates under mild conditions makes this patent a cornerstone for modern beta-lactam manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of carbapenam and carbapenem nuclei has relied heavily on methods involving the thermal decomposition of diazonium compounds derived from triazo-compound reagents. As noted in prior art such as EP0037080 A1, these conventional pathways necessitate heating diazonium compounds to temperatures between 50°C and 100°C, often in the presence of catalysts like copper sulfate or palladium acetate. The fundamental flaw in this approach lies in the intrinsic instability of the triazo-compound reagents and the resulting diazonium intermediates, which are prone to violent explosion upon heating. Handling large quantities of these energetic materials presents a danger close to unacceptable levels for modern industrial safety standards. Moreover, the requirement for high-temperature heating to induce cyclization increases energy consumption and complicates reactor design, necessitating specialized equipment capable of withstanding potential over-pressure events. These factors collectively render the conventional diazonium route suboptimal for commercial runs, creating bottlenecks in production capacity and inflating insurance and safety mitigation costs.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a sophisticated sequence involving the formation of an alpha-iodo enone intermediate followed by a mild rhodium-catalyzed cyclization. This method completely eliminates the need for explosive triazo-reagents and diazonium compounds, thereby removing the primary safety hazard associated with beta-lactam core synthesis. The process begins with the reaction of a beta-lactam precursor with a hypervalent iodine reagent, such as phenyliodine diacetate, in the presence of a base to generate a stable aza-cyclobutanone derivative. This intermediate is then subjected to cyclization using a rhodium catalyst at temperatures ranging from -20°C to 100°C, with a strong preference for mild conditions near room temperature. This dramatic reduction in thermal stress not only enhances safety but also improves the selectivity of the reaction, minimizing the formation of degradation byproducts. The result is a streamlined, safer, and more economically viable pathway that is ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
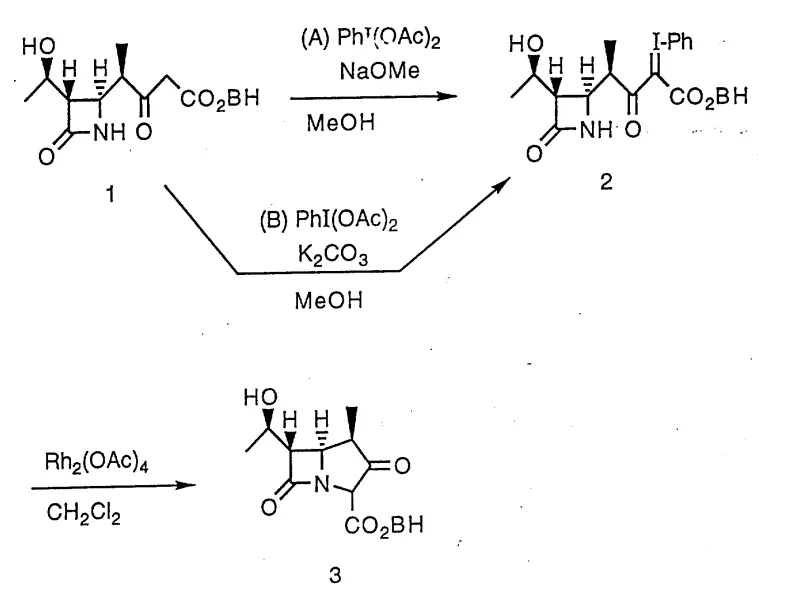
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise orchestration of hypervalent iodine oxidation followed by transition metal-catalyzed ring closure. The initial step involves the activation of the beta-lactam side chain using a reagent of the formula R3-I(Q)2, where Q is a substituent derived from an acid anion, such as acetate. In the presence of a base like potassium carbonate or sodium methoxide, this reagent facilitates the introduction of an aryl-iodonium group at the alpha-position relative to the carbonyl, creating a highly reactive yet isolable enone intermediate. This alpha-iodo functionality serves as a superior leaving group compared to the nitrogen-based leaving groups used in traditional methods, allowing for a much smoother intramolecular nucleophilic attack. The subsequent cyclization is driven by a rhodium catalyst, typically rhodium acetate (Rh2(OAc)4), which coordinates with the enone system to lower the activation energy for ring closure. This catalytic cycle proceeds efficiently in aprotic solvents such as dichloromethane or toluene, ensuring that the sensitive beta-lactam ring remains intact throughout the transformation.
From an impurity control perspective, this mechanism offers distinct advantages over thermal decomposition routes. The use of a specific rhodium catalyst allows for tight control over the stereochemistry of the newly formed ring junction, which is critical for the biological activity of the final antibiotic. Unlike the chaotic thermal breakdown of diazonium salts which can generate a wide array of radical-derived impurities, the metal-catalyzed pathway follows a defined coordination geometry that favors the desired stereoisomer. Furthermore, the mild reaction conditions prevent the thermal degradation of the sensitive four-membered beta-lactam ring, a common issue in high-temperature processes. The ability to purify the iodo-intermediate prior to cyclization adds an additional layer of quality control, allowing manufacturers to remove unreacted starting materials or side products before the final ring-closing step. This results in a final product with a significantly cleaner impurity profile, reducing the burden on downstream purification processes and increasing overall yield.
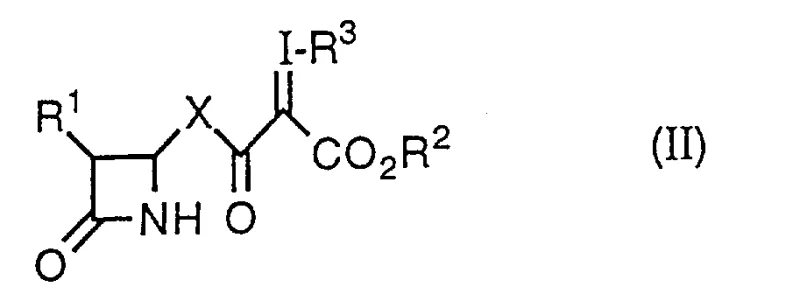
How to Synthesize Carbapenam Derivatives Efficiently
The synthesis of these critical antibiotic intermediates requires a disciplined approach to reagent selection and process control to maximize yield and safety. The patented route delineates a clear two-stage protocol that begins with the oxidative functionalization of the precursor and concludes with the catalytic ring closure. Operators must ensure strict stoichiometry during the iodination phase to prevent over-oxidation, while the cyclization step demands careful monitoring of catalyst loading to balance reaction rate against cost. The detailed standardized synthesis steps below outline the precise conditions required to replicate the high yields reported in the patent embodiments, serving as a foundational guide for process chemists looking to implement this technology.
- Oxidation of the beta-lactam precursor using phenyliodine diacetate (PhI(OAc)2) in the presence of a base like potassium carbonate or sodium methoxide to form the alpha-iodo enone intermediate.
- Purification of the iodo-intermediate via crystallization or filtration to remove inorganic salts and ensure high purity before the cyclization step.
- Intramolecular cyclization of the purified intermediate using a rhodium catalyst such as Rh2(OAc)4 in an aprotic solvent like dichloromethane at mild temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed synthesis route translates into tangible strategic benefits that extend far beyond simple chemical yield. The elimination of explosive precursors fundamentally alters the risk profile of the manufacturing facility, leading to substantial cost savings in terms of insurance premiums, safety infrastructure, and regulatory compliance overhead. By removing the need for specialized high-pressure reactors and blast-proof containment systems, capital expenditure for new production lines can be significantly reduced. Additionally, the use of stable intermediates allows for more flexible inventory management, as the iodo-compounds can potentially be stored or transported with greater ease than their unstable diazonium counterparts. This flexibility enhances supply chain resilience, ensuring that production schedules are not disrupted by safety hold-ups or stringent transport restrictions on hazardous materials.
- Cost Reduction in Manufacturing: The economic impact of switching to this safer methodology is profound, primarily driven by the removal of expensive safety mitigation measures and the optimization of reaction conditions. Since the process operates effectively at or near room temperature, energy consumption for heating and cooling is drastically simplified compared to the high-thermal demand of legacy diazonium routes. Furthermore, the avoidance of transition metals like copper or palladium in favor of highly efficient rhodium catalysts—used in minute quantities—can lead to a more streamlined metal clearance process, reducing the cost of scavengers and purification media. The higher selectivity of the reaction also means less raw material is wasted on byproduct formation, directly improving the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the logistical challenges associated with hazardous chemicals, and this new route effectively neutralizes those risks. By relying on commercially available and stable reagents like phenyliodine diacetate and rhodium acetate, manufacturers can secure long-term contracts with multiple suppliers without fear of sudden regulatory bans on explosive precursors. The robustness of the chemistry ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that could delay shipments to downstream API producers. This reliability is critical for maintaining just-in-time inventory levels and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates safety and environmental issues, but this methodology is inherently designed for expansion. The mild reaction conditions and the absence of nitrogen gas evolution (a byproduct of diazonium decomposition) simplify reactor engineering and waste gas treatment systems. From an environmental standpoint, the process generates less hazardous waste, aligning with increasingly strict global regulations on chemical manufacturing emissions. The ability to run the reaction in common solvents like dichloromethane or toluene, which have well-established recovery and recycling protocols, further supports sustainable manufacturing practices. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental laws but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-lactam synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production workflows.
Q: Why is the Rhodium-catalyzed route safer than traditional diazonium methods?
A: Traditional methods rely on heating diazonium compounds which pose severe explosion risks. The patented Rhodium-catalyzed route utilizes stable iodo-intermediates and mild cyclization conditions, eliminating the thermal instability hazards associated with triazo-compounds.
Q: What catalysts are suitable for this cyclization process?
A: The process specifically utilizes transition metal salts, with elemental rhodium salts being preferred. Rhodium acetate (Rh2(OAc)4) is highlighted as particularly effective, though trifluoroacetate and pivalate variants are also viable depending on solubility and reactivity requirements.
Q: Can this process be scaled for industrial antibiotic production?
A: Yes, the method is designed for industrial scalability. It operates at mild temperatures (-20°C to 100°C, preferably room temperature) and uses standard aprotic solvents like dichloromethane or toluene, avoiding the extreme conditions and safety protocols required for explosive precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenam Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value antibiotic intermediates. Our team of expert process chemists has extensively evaluated the rhodium-catalyzed cyclization technology described in CN1097424A and possesses the technical capability to translate this laboratory-scale innovation into robust commercial manufacturing processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale operation is seamless and compliant with international quality standards. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of carbapenam derivatives we produce.
We invite pharmaceutical partners to collaborate with us on optimizing their supply chains through the adoption of this advanced chemistry. By leveraging our expertise in hypervalent iodine chemistry and transition metal catalysis, we can help you achieve significant operational efficiencies and cost reductions in beta-lactam manufacturing. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for the iodo-intermediates or comprehensive route feasibility assessments for the final cyclization step, our team is ready to provide the detailed technical support necessary to secure your supply of high-purity beta-lactams.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
