Scalable Production of High-Purity 2-Amino Malonamide for Global API Manufacturing
Scalable Production of High-Purity 2-Amino Malonamide for Global API Manufacturing
The pharmaceutical industry's demand for robust antiviral intermediates has never been more critical, particularly for the synthesis of Favipiravir and related heterocyclic compounds. A significant technological breakthrough in this domain is detailed in patent CN113831255B, which outlines a highly efficient preparation method for 2-amino malonamide. This specific intermediate serves as a pivotal building block in the construction of complex imidazole cores found in modern antiviral therapeutics. The disclosed methodology represents a paradigm shift from traditional, cumbersome synthetic routes to a streamlined, aqueous-based process that prioritizes both operational simplicity and exceptional product quality. By leveraging a controlled low-temperature ammonolysis strategy, this approach achieves purity levels exceeding 99.5%, addressing the stringent regulatory requirements faced by API manufacturers globally. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply chain for high-value pharmaceutical intermediates.
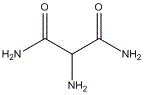
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-amino malonamide has been plagued by significant technical bottlenecks that hinder large-scale production efficiency. Conventional pathways often rely on organic solvents such as methanol or ethanol, which introduce substantial complications regarding solvent recovery, environmental waste disposal, and overall process safety. Furthermore, traditional methods frequently suffer from uncontrolled exothermic reactions during the amidation step, leading to thermal runaways that degrade product quality and pose serious safety risks to plant personnel. The reliance on harsh reaction conditions often necessitates the use of expensive, corrosion-resistant reactor materials, thereby inflating the capital expenditure required for setting up production lines. Additionally, older protocols typically involve multi-step workups and prolonged reaction times, resulting in lower overall yields and inconsistent purity profiles that fail to meet the rigorous specifications of modern GMP manufacturing environments. These inefficiencies create a fragile supply chain vulnerable to disruptions and cost volatility.
The Novel Approach
In stark contrast, the novel approach described in the patent data utilizes a refined aqueous system that fundamentally simplifies the production workflow while enhancing output metrics. By employing water as the primary solvent for the diethyl 2-aminomalonate hydrochloride solution, the process eliminates the need for volatile organic compounds, thereby reducing fire hazards and environmental compliance burdens. The core innovation lies in the precise control of the addition sequence: dripping the ester solution into cooled ammonia water rather than the reverse. This specific operational parameter effectively manages the heat of reaction, keeping the temperature safely below 10°C and preventing the formation of thermal degradation byproducts. The result is a single-vessel process that combines reaction and crystallization, removing the need for intermediate transfers and significantly shortening the production cycle. This streamlined methodology not only boosts throughput but also ensures a consistent, high-purity product suitable for direct use in sensitive downstream coupling reactions.
Mechanistic Insights into Aqueous Ammonolysis and Crystallization
The chemical transformation driving this synthesis is a nucleophilic acyl substitution, specifically the ammonolysis of the ester groups in diethyl 2-aminomalonate hydrochloride. In this mechanism, ammonia acts as a potent nucleophile, attacking the carbonyl carbon of the ester functionality to form a tetrahedral intermediate. The presence of water as a solvent plays a dual role: it stabilizes the ionic species involved, particularly the hydrochloride salt, and facilitates the proton transfer steps necessary for the collapse of the tetrahedral intermediate into the final amide product. Crucially, the low-temperature environment (maintained between -5°C and 10°C) kinetically favors the formation of the desired diamide over potential hydrolysis side products. If the temperature were to rise unchecked, the increased kinetic energy could promote the hydrolysis of the ester or the newly formed amide bonds, leading to the formation of malonic acid derivatives which are difficult to separate. Therefore, the thermal management strategy is not merely a safety precaution but a critical mechanistic requirement for preserving the structural integrity of the 2-amino malonamide backbone.
Following the reaction completion, the purification mechanism relies heavily on the differential solubility of the product in the aqueous matrix at varying temperatures. As the reaction mixture is cooled to below -5°C, the solubility of 2-amino malonamide decreases sharply, inducing supersaturation and subsequent nucleation. This low-temperature crystallization is vital for excluding impurities that remain soluble in the mother liquor, such as unreacted starting materials or mono-amide intermediates. The patent data highlights that crystallization times are flexible, but the temperature threshold is rigid; maintaining the slurry at sub-zero temperatures ensures the growth of well-defined crystals with high lattice purity. Subsequent filtration and drying at moderate temperatures (50-55°C) remove residual moisture without risking thermal decomposition, yielding a free-flowing solid with a purity profile that consistently exceeds 99.5%. This rigorous control over the solid-state chemistry ensures that the impurity spectrum is minimized, reducing the burden on downstream purification processes.
How to Synthesize 2-Amino Malonamide Efficiently
Implementing this synthesis route requires strict adherence to the thermodynamic parameters outlined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of a cooled aqueous ammonia solution, which serves as the reaction medium and nitrogen source. Operators must ensure that the cooling capacity of the reactor is sufficient to handle the exotherm generated during the addition phase. The diethyl 2-aminomalonate hydrochloride is dissolved in water to form a homogeneous feed solution, which is then introduced slowly into the ammonia reservoir. Throughout this addition, continuous monitoring of the internal temperature is mandatory to prevent excursions above 10°C. Once the addition is complete, the reaction mixture is held to allow for full conversion, typically monitored via HPLC to confirm the disappearance of the ester starting material. The detailed standardized synthesis steps, including specific molar ratios and agitation speeds, are provided in the technical guide below.
- Prepare a cooled aqueous ammonia solution and maintain the reaction temperature below 10°C to prevent exothermic runaway.
- Slowly add the diethyl 2-aminomalonate hydrochloride solution into the ammonia water while monitoring the temperature strictly.
- Upon completion, cool the mixture to below -5°C to induce crystallization, followed by filtration and drying at 50-55°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous synthesis route offers profound strategic advantages that extend beyond simple unit cost calculations. The elimination of organic solvents like methanol and ethanol removes a significant variable from the raw material sourcing equation, insulating the production process from the volatility of petrochemical markets. Furthermore, the simplified equipment requirements mean that production can be scaled rapidly using existing infrastructure without the need for specialized corrosion-resistant linings or explosion-proof solvent handling systems. This flexibility enhances supply chain resilience, allowing for quicker response times to fluctuating market demands for antiviral intermediates. The high yield and purity reported in the patent data translate directly into reduced waste generation and lower disposal costs, contributing to a more sustainable and economically viable manufacturing model. Ultimately, this process optimization aligns perfectly with the industry's drive towards greener chemistry and cost-effective production.
- Cost Reduction in Manufacturing: The transition to a water-based solvent system fundamentally alters the cost structure of 2-amino malonamide production by eliminating the expense associated with purchasing, recovering, and disposing of large volumes of organic solvents. Water is universally available and inexpensive, providing a drastic reduction in raw material overheads compared to alcohol-based methods. Additionally, the high conversion efficiency minimizes the loss of valuable starting materials, ensuring that the maximum amount of input is converted into sellable product. The simplified workflow, which combines reaction and crystallization in a single vessel, reduces labor hours and energy consumption associated with solvent swapping and multiple transfer operations. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), offering a competitive pricing advantage in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more stable and predictable supply chain for critical pharmaceutical intermediates. By utilizing readily available commodity chemicals like ammonia and water, the process reduces dependency on specialized reagents that may face supply shortages or logistical bottlenecks. The operational simplicity means that production can be easily transferred between different manufacturing sites or scaled up at existing facilities with minimal requalification effort. This adaptability ensures continuity of supply even in the face of regional disruptions or equipment maintenance schedules. Moreover, the high purity of the output reduces the risk of batch failures during downstream API synthesis, preventing costly delays in the final drug product manufacturing timeline.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a clear pathway to sustainable scale-up. The absence of volatile organic compounds (VOCs) simplifies compliance with increasingly stringent environmental protection regulations, reducing the need for complex abatement systems. The aqueous waste stream is easier to treat and manage compared to mixed organic-aqueous waste, lowering the environmental footprint of the facility. The mild reaction conditions and lack of corrosive byproducts extend the lifespan of production equipment, reducing capital replacement cycles. This combination of environmental stewardship and operational durability makes the technology highly attractive for long-term investment and large-scale commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-amino malonamide, derived directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational parameters and quality expectations for partners considering this technology for their supply chain. Understanding these details is crucial for assessing the feasibility of integrating this intermediate into your specific manufacturing workflow.
Q: Why is water preferred over alcohol solvents for this amidation reaction?
A: Water serves as a superior solvent compared to methanol or ethanol because it significantly enhances the purity of the final 2-amino malonamide while drastically reducing raw material costs. The patent data indicates that alcoholic solvents can lead to lower yields and purity profiles due to potential transesterification side reactions or solubility issues during crystallization.
Q: What is the critical temperature control range for ensuring high yield?
A: The reaction temperature must be strictly controlled below 10°C, ideally between -5°C and 10°C. Exceeding this range during the dropwise addition can cause rapid temperature spikes, leading to product degradation and reduced purity, whereas maintaining this low range ensures the stability of the amide bond formation.
Q: How does this process impact equipment corrosion requirements?
A: Unlike traditional methods that may require harsh acidic or basic conditions necessitating specialized corrosion-resistant alloys, this aqueous ammonolysis process operates under mild conditions. This reduces the capital expenditure on reactor materials, allowing for the use of standard stainless steel equipment without compromising safety or product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino Malonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapies depends on the availability of high-quality precursors like 2-amino malonamide. Our technical team has thoroughly analyzed the pathway described in CN113831255B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to life. We are equipped with rigorous QC labs and adhere to stringent purity specifications to ensure that every batch meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence allows us to navigate the complexities of low-temperature crystallization and aqueous processing with precision, delivering a product that supports your regulatory filings and production goals.
We invite you to collaborate with us to optimize your supply chain for Favipiravir and related intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can enhance your project's success. Let us be your partner in delivering reliable, high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
