Scalable Synthesis of Halogenated Benzoic Acid Derivatives for Commercial Production
Introduction to Advanced Halogenated Benzoic Acid Synthesis
The chemical landscape for producing complex fluorinated intermediates is constantly evolving, driven by the rigorous demands of modern pharmaceutical and agrochemical development. Patent CN102056883A introduces a robust and versatile methodology for the preparation of 3-chloro-2-fluoro-5-trifluoromethylbenzoic acid, designated as Formula I, which serves as a critical building block for numerous active ingredients. This invention addresses significant limitations found in earlier synthetic routes, particularly those disclosed in DE A1 3935934, which relied on hazardous reagents like tert-butyllithium at cryogenic temperatures of -78°C. By shifting towards more controllable lithiation conditions and efficient oxidation strategies, this technology offers a pathway that is not only chemically superior but also inherently safer for industrial implementation. The ability to synthesize such highly functionalized aromatic acids with precision is paramount for ensuring the quality and consistency of downstream drug substances.
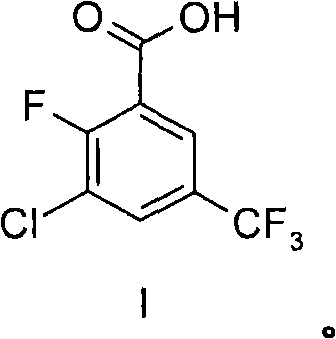
Furthermore, the strategic value of this intermediate cannot be overstated, as it finds application in the creation of potent bioactive molecules where specific halogen substitution patterns dictate biological activity. The patent outlines multiple embodiments, ranging from direct carboxylation via carbon dioxide to a two-step sequence involving formylation followed by oxidation. This flexibility allows process chemists to adapt the synthesis based on available infrastructure and raw material costs. For organizations seeking a reliable agrochemical intermediate supplier or partners in fine chemical manufacturing, understanding the nuances of this patented process provides a competitive edge in securing supply chains for high-value precursors. The transition from laboratory-scale curiosity to commercial viability is often hindered by safety and scalability issues, which this invention explicitly aims to resolve through optimized reaction parameters and workup procedures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-2-fluoro-5-trifluoromethylbenzoic acid has been plagued by operational difficulties that restrict its large-scale production. The prior art, specifically the method described in DE A1 3935934, necessitates the use of tert-butyllithium, a pyrophoric and highly corrosive reagent that poses severe safety risks in a manufacturing environment. Operating at temperatures as low as -78°C requires specialized cryogenic equipment and significant energy input, driving up both capital expenditure and operational costs. Moreover, the handling of such reactive organolithium species increases the likelihood of side reactions and thermal runaways if not meticulously controlled, leading to inconsistent yields and impurity profiles that are unacceptable for GMP-grade production. These factors collectively create a bottleneck for companies attempting to scale up the synthesis of this vital intermediate, often resulting in supply shortages or prohibitive pricing for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology presented in CN102056883A utilizes n-butyllithium or lithium diisopropylamide (LDA) under controlled conditions, which, while still requiring low temperatures, offers a more manageable risk profile compared to tert-butyllithium. The innovation extends beyond the initial lithiation step to include a highly efficient oxidation of the aldehyde intermediate using common oxidants like sodium hypochlorite in the presence of bromide salts. This approach eliminates the need for expensive or toxic heavy metal oxidants, aligning with green chemistry principles and simplifying waste treatment protocols. The patent further describes a telescoped process where the aldehyde is not isolated but directly converted to the acid, significantly reducing solvent consumption and processing time. This streamlined workflow represents a substantial advancement in cost reduction in pharma manufacturing, enabling producers to achieve higher throughput with reduced environmental impact. By mitigating the hazards associated with cryogenic operations and corrosive reagents, this novel approach facilitates the commercial scale-up of complex fluorinated intermediates.
Mechanistic Insights into Lithiation-Formylation and Oxidative Conversion
The core of this synthetic strategy relies on the precise regioselective deprotonation of 3-chloro-4-fluoro-benzotrifluoride. The presence of the trifluoromethyl group and the halogen substituents creates a specific electronic environment that directs the metalation to the desired position ortho to the fluorine atom. When treated with a strong base like n-butyllithium in tetrahydrofuran (THF) at temperatures between -78°C and -50°C, the proton at the 2-position is abstracted to form a stable aryl lithium species. This intermediate is then quenched with an electrophile, typically N,N-dimethylformamide (DMF), to introduce the formyl group, yielding 3-chloro-2-fluoro-5-trifluoromethylbenzaldehyde. The control of temperature during this addition is critical to prevent over-lithiation or decomposition of the sensitive organometallic intermediate. Following the formation of the aldehyde, the process employs an oxidative conversion to the carboxylic acid. The mechanism involves the generation of hypobromite in situ when sodium hypochlorite is combined with potassium bromide, which acts as a potent oxidant capable of converting the aldehyde functionality to the corresponding acid without affecting the sensitive chloro and fluoro substituents on the aromatic ring.
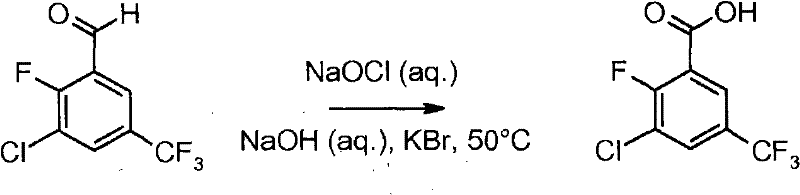
Impurity control is a central feature of this mechanistic design, particularly regarding the management of halogenated byproducts. The use of aqueous sodium sulfite as a quenching agent effectively neutralizes excess oxidizing species, preventing over-oxidation or degradation of the product. Furthermore, the purification strategy leverages the solubility differences of the acid and its salts; by adjusting the pH to greater than 11, organic impurities can be separated into the organic phase, while the desired product remains in the aqueous layer as a salt. Subsequent acidification to pH less than 2 precipitates the pure benzoic acid, which can then be recrystallized from solvents like cyclohexane or heptane to achieve high purity specifications. This rigorous control over the reaction environment and workup ensures that the final material meets the stringent quality standards required for high-purity OLED material or pharmaceutical intermediate applications. The ability to tune the oxidation conditions, whether using hypochlorite, Oxone, or permanganate, provides additional levers for optimizing the impurity profile based on specific customer requirements.
How to Synthesize 3-Chloro-2-fluoro-5-trifluoromethylbenzoic Acid Efficiently
Executing this synthesis requires careful attention to reaction conditions, particularly the maintenance of low temperatures during the lithiation step to ensure regioselectivity and safety. The process begins with the dissolution of the starting material, 3-chloro-4-fluoro-benzotrifluoride, in an anhydrous ether solvent such as THF, followed by the slow addition of the base. Once the aldehyde is formed, it can be isolated or carried forward directly. The oxidation step is exothermic and requires controlled addition of the oxidant to manage heat release. Detailed standard operating procedures for each stage, including specific molar ratios and quenching protocols, are essential for reproducibility. For a comprehensive guide on the exact experimental parameters and safety precautions, please refer to the standardized synthesis steps provided below.
- Deprotonate 3-chloro-4-fluoro-benzotrifluoride using n-BuLi or LDA in THF at low temperatures (-78°C).
- React the lithiated intermediate with DMF to form 3-chloro-2-fluoro-5-trifluoromethylbenzaldehyde.
- Oxidize the aldehyde intermediate using sodium hypochlorite (NaOCl) with a bromide catalyst or Oxone to yield the final benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented process offers tangible benefits that extend beyond simple yield improvements. The shift away from tert-butyllithium to n-butyllithium or Grignard reagents significantly reduces the cost of goods sold by utilizing more commercially available and less hazardous raw materials. This change also simplifies the regulatory compliance burden associated with storing and handling pyrophoric substances, thereby lowering insurance and facility maintenance costs. Additionally, the flexibility to use various oxidants allows manufacturing sites to leverage existing infrastructure, avoiding the need for costly new equipment installations. These factors collectively contribute to a more resilient supply chain, reducing the risk of production stoppages due to reagent shortages or safety incidents. For supply chain heads, the ability to source this intermediate from a reliable agrochemical intermediate supplier who utilizes such robust methods ensures continuity of supply for critical downstream products.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like tert-butyllithium directly lowers raw material costs and reduces the need for specialized cryogenic infrastructure. By enabling the use of aqueous oxidation systems with sodium hypochlorite, the process minimizes solvent waste and simplifies effluent treatment, leading to substantial cost savings in waste disposal. The telescoped nature of certain embodiments further reduces processing time and utility consumption, enhancing overall operational efficiency without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as n-butyllithium, DMF, and bleach ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic reagents. The robustness of the reaction conditions means that production can be scaled up with greater confidence, reducing lead times for high-purity intermediates. This stability is crucial for long-term planning and inventory management, allowing partners to secure consistent volumes of material needed for continuous manufacturing campaigns.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as liquid-liquid extraction and crystallization. The avoidance of heavy metal catalysts and the use of environmentally benign oxidants align with increasingly strict global environmental regulations. This compliance reduces the risk of regulatory shutdowns and enhances the sustainability profile of the final product, which is a growing priority for end-users in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-chloro-2-fluoro-5-trifluoromethylbenzoic acid. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is key to evaluating the feasibility of integrating this intermediate into your specific production workflows.
Q: What are the primary safety advantages of this new process compared to prior art?
A: The patented process avoids the use of highly corrosive tert-butyllithium at extreme low temperatures, replacing it with more manageable n-butyllithium or Grignard reagents, thereby significantly reducing operational hazards and scaling risks.
Q: Can the aldehyde intermediate be isolated before oxidation?
A: Yes, the process allows for the isolation of 3-chloro-2-fluoro-5-trifluoromethylbenzaldehyde, but it also supports a telescoped procedure where the crude aldehyde is directly oxidized, improving overall throughput and reducing solvent usage.
Q: What oxidizing agents are compatible with this synthesis route?
A: The patent demonstrates versatility by successfully utilizing sodium hypochlorite (with KBr or NaBr), potassium persulfate (Oxone), and potassium permanganate, allowing manufacturers to select based on cost and waste treatment capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-2-fluoro-5-trifluoromethylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-chloro-2-fluoro-5-trifluoromethylbenzoic acid meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that facilitate your research and manufacturing goals with reliability and precision.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can benefit your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project, ensuring a seamless partnership from development to commercial supply.
