Scalable Synthesis of 2-Acetyl-1,10-Phenanthroline for Advanced Photoelectric Applications
Introduction to Patent CN112939969A: A Paradigm Shift in Phenanthroline Synthesis
The landscape of synthesizing functionalized phenanthrolines, specifically 2-acetyl-1,10-phenanthroline, has historically been constrained by hazardous reagents and extreme operating conditions. Patent CN112939969A introduces a transformative methodology that replaces traditional cryogenic lithiation with a温和 yet robust sequence of condensation and cyclization reactions. This innovation is particularly critical for the production of photoelectric and catalytic materials, where supply chain stability and process safety are paramount. By leveraging a Schiff base formation followed by a Doebner-type condensation and subsequent decarboxylation, this route offers a viable pathway for industrial manufacturers seeking to mitigate the risks associated with organometallic reagents. The strategic shift from low-temperature batch processes to reflux-based continuous operations represents a significant advancement in process chemistry, aligning perfectly with modern green chemistry principles while maintaining high structural fidelity of the target molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-acetyl-1,10-phenanthroline relied heavily on the halogen-lithium exchange reaction using n-butyllithium at cryogenic temperatures of -78°C. This conventional approach presents severe logistical and safety challenges for large-scale manufacturing, primarily due to the requirement for specialized low-temperature reactors and the handling of pyrophoric reagents. Furthermore, alternative routes utilizing 3-acetyl acrolein suffer from raw material instability, as this aldehyde is prone to polymerization and requires stringent storage conditions that complicate inventory management. The use of trimethylsilyl cyanide in some variations introduces additional toxicity concerns, creating significant burdens for waste treatment and environmental compliance. These factors collectively result in high production costs, limited batch sizes, and inconsistent yields, making the conventional methods economically unviable for meeting the growing demand in the electronic materials sector.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a convergent synthesis strategy starting from readily available 8-aminoquinoline and methylglyoxal. This approach circumvents the need for cryogenic infrastructure by operating under mild thermal conditions, typically between 40°C and 70°C for the initial condensation. The core of this innovation lies in the construction of the phenanthroline skeleton via a Doebner reaction, which efficiently builds the heterocyclic ring system with high atom economy. By avoiding unstable intermediates like 3-acetyl acrolein and replacing them with stable ketones and amines, the process ensures a consistent supply of quality raw materials. This transition not only simplifies the operational workflow but also drastically reduces the generation of hazardous solid waste, positioning this synthesis route as a superior choice for sustainable industrial production of high-value electronic intermediates.
Mechanistic Insights into the Three-Step Cascade Synthesis
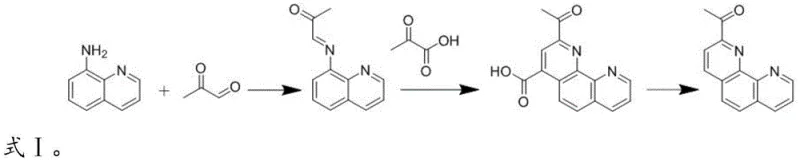
The chemical elegance of this synthesis lies in its sequential transformation of simple building blocks into a complex fused heterocycle. The process initiates with a Schiff base reaction where the primary amine of 8-aminoquinoline nucleophilically attacks the carbonyl carbon of methylglyoxal. This condensation eliminates water to form the imine intermediate, E-1-(quinoline-8-imino)propan-2-one, which serves as the crucial scaffold for ring closure. The reaction kinetics are carefully controlled by temperature and solvent polarity, ensuring the exclusive formation of the E-isomer which is geometrically predisposed for the subsequent cyclization. This step is fundamental as it installs the necessary carbon framework and nitrogen functionality required for the final phenanthroline structure without introducing extraneous protecting groups.
Following the imine formation, the system undergoes a Doebner reaction upon the addition of pyruvic acid and an acid catalyst such as polyphosphoric acid or vanadic acid. This step involves an electrophilic aromatic substitution where the electron-rich quinoline ring attacks the activated alpha,beta-unsaturated system generated in situ. The resulting cyclization forms the second pyridine ring, yielding 2-acetylphenanthroline-4-formic acid. Finally, the carboxylic acid moiety is removed via alkaline decarboxylation under reflux conditions. This thermal elimination of carbon dioxide restores the aromaticity of the newly formed ring and delivers the target 2-acetyl-1,10-phenanthroline. The entire cascade is designed to minimize side reactions, ensuring a clean impurity profile suitable for sensitive optoelectronic applications.
How to Synthesize 2-Acetyl-1,10-Phenanthroline Efficiently
Implementing this synthesis protocol requires precise control over stoichiometry and thermal parameters to maximize yield and purity. The process begins with the dissolution of 8-aminoquinoline in an alcoholic solvent, followed by the controlled addition of methylglyoxal to manage the exotherm of the Schiff base formation. Once the intermediate is isolated, it is subjected to the cyclization conditions in absolute ethanol with careful monitoring of water removal to drive the equilibrium forward. The final decarboxylation step demands strict pH control in the alkaline range to facilitate smooth gas evolution without degrading the acetyl group. For detailed operational parameters, stoichiometry ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Perform a Schiff base reaction by mixing 8-aminoquinoline and methylglyoxal in an alcohol solvent at 40-70°C to obtain E-1-(quinoline-8-imino)propan-2-one.
- Conduct a Doebner reaction by refluxing the intermediate with pyruvic acid and an acid catalyst (e.g., PPA or vanadic acid) in absolute ethanol to form 2-acetylphenanthroline-4-formic acid.
- Execute alkaline decarboxylation by adjusting the pH to 10-10.5 and refluxing the carboxylic acid intermediate to yield the final 2-acetyl-1,10-phenanthroline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this novel synthetic route offers substantial advantages by decoupling production from volatile supply chains associated with specialty organometallic reagents. The reliance on commodity chemicals like 8-aminoquinoline and pyruvic acid ensures a stable and predictable cost structure, shielding manufacturers from the price fluctuations often seen with lithium reagents or unstable aldehydes. Furthermore, the elimination of cryogenic processing removes the need for energy-intensive cooling systems, leading to significant reductions in utility costs and capital expenditure on specialized reactor infrastructure. This operational simplicity translates directly into improved margin potential for downstream users in the photoelectric industry, allowing for more competitive pricing of the final electronic materials.
- Cost Reduction in Manufacturing: The replacement of expensive n-butyllithium and cryogenic equipment with standard reflux setups and acid catalysts results in a drastic reduction in direct material and operational costs. By utilizing common solvents like ethanol and avoiding hazardous reagents that require special disposal protocols, the overall cost of goods sold is significantly optimized. This economic efficiency is further enhanced by the high atom economy of the Doebner reaction, which minimizes raw material waste and maximizes the output per batch, providing a clear financial advantage over traditional lithiation methods.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting materials are bulk chemicals with established global supply networks, unlike the niche and often constrained supply of 3-acetyl acrolein. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or strict environmental regulations regarding toxic waste. This reliability ensures consistent lead times for customers, enabling just-in-time manufacturing strategies for high-tech applications where material availability is critical for maintaining production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of dangerous exotherms associated with organolithium chemistry, allowing for safe expansion from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation, particularly the avoidance of heavy metal residues and toxic cyanide byproducts, simplifies regulatory compliance and lowers the burden on wastewater treatment facilities. This environmentally friendly profile aligns with increasingly stringent global sustainability standards, making the supply chain more resilient against future regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on reaction mechanisms and purification strategies. Understanding these details is essential for process engineers aiming to adapt this methodology for large-scale manufacturing environments.
Q: What are the critical reaction conditions for the Doebner cyclization step?
A: The Doebner reaction requires reflux conditions in absolute ethanol for 5-20 hours. Critical catalysts include polyphosphoric acid (PPA) or vanadic acid, with a mass ratio to the intermediate ranging from 0.01:1 to 8:1 to ensure efficient cyclization without excessive degradation.
Q: How is the final purity of 2-acetyl-1,10-phenanthroline ensured?
A: High purity (up to 98.9%) is achieved through a rigorous workup involving alkaline decarboxylation followed by cooling crystallization. The process includes activated carbon decolorization and recrystallization from anhydrous ethanol to remove trace organic impurities and colored byproducts.
Q: Why is this method superior to the traditional butyl lithium route?
A: This method eliminates the need for hazardous cryogenic conditions (-78°C) and expensive, pyrophoric reagents like n-butyllithium. It utilizes stable, commercially available starting materials like 8-aminoquinoline and methylglyoxal, significantly improving operational safety and industrial scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetyl-1,10-Phenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of advanced photoelectric devices. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-acetyl-1,10-phenanthroline meets the exacting standards required for electronic applications. Our commitment to quality assurance means that our clients can rely on consistent material properties, which is vital for maintaining the efficiency and longevity of their final products.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you reduce total acquisition costs while securing a stable supply of this critical intermediate. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can support your supply chain objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
