Advanced Selenium-Ionic Liquid Catalysis for High-Purity Agrochemical Intermediates
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic pathways, a shift clearly exemplified by the innovations disclosed in patent CN1761641A. This pivotal intellectual property introduces a robust method for producing oxygen-containing compounds through the reaction of olefin compounds with organic hydroperoxides, uniquely utilizing a selenium compound catalyst within an ionic liquid medium. For industry leaders seeking a reliable agrochemical intermediate supplier, this technology represents a critical advancement over legacy oxidation methods that have long relied on stoichiometric amounts of toxic reagents and volatile organic solvents. By leveraging the unique solvation properties of ionic liquids, such as alkyl-substituted imidazolium salts, the process achieves high selectivity and yield while fundamentally simplifying the downstream purification workflow. The ability to tune the oxidation state from alcohols to aldehydes or carboxylic acids simply by adjusting the molar ratio of the hydroperoxide offers unprecedented flexibility for commercial scale-up of complex organic oxidations. This report analyzes the technical merits and commercial implications of adopting this selenium-catalyzed ionic liquid system for the production of high-value fine chemicals and agrochemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the allylic oxidation of olefins to generate valuable oxygenated intermediates has been plagued by significant environmental and operational inefficiencies that hinder cost reduction in agrochemical manufacturing. Traditional protocols often necessitate the use of stoichiometric quantities of selenium dioxide or tin tetrachloride, which not only generates substantial amounts of heavy metal waste but also requires complex and energy-intensive regeneration steps to recover the selenium for reuse. Furthermore, these reactions were typically conducted in chlorinated solvents like methylene chloride, which pose severe occupational health risks and create substantial liabilities regarding solvent disposal and emissions compliance. The difficulty in separating the catalyst from the product mixture in homogeneous organic solutions often leads to product contamination, requiring additional purification stages that erode profit margins and extend production timelines. Consequently, the industry has faced a persistent bottleneck in scaling these oxidation reactions without incurring prohibitive costs or environmental penalties, creating a strong demand for alternative catalytic systems that can operate under greener conditions.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by employing a catalytic amount of selenium compound dispersed within a recyclable ionic liquid phase, effectively addressing the core deficiencies of prior art. This biphasic system allows the reaction to proceed with high efficiency while enabling the catalyst to remain sequestered in the ionic liquid layer post-reaction, facilitating straightforward separation from the organic product layer via simple decantation or extraction with non-polar solvents like n-hexane. The ionic liquid acts not merely as a solvent but as a stabilizing matrix that enhances the activity of the selenium species, allowing for lower reaction temperatures and reduced catalyst loading compared to conventional methods. This approach eliminates the need for hazardous chlorinated solvents, thereby aligning the manufacturing process with stringent global environmental regulations and improving workplace safety profiles. By enabling the direct reuse of the ionic liquid-catalyst mixture for multiple cycles without significant loss of activity, the process drastically reduces raw material consumption and waste generation, offering a compelling value proposition for reducing lead time for high-purity fine chemicals.
Mechanistic Insights into Selenium-Catalyzed Allylic Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the selenium catalyst and the ionic liquid medium, which modifies the electronic environment of the oxidation reaction to favor specific product outcomes. The selenium compound, which may include selenium dioxide, selenious acid, or organic selenides, activates the organic hydroperoxide to generate a reactive selenium-oxo species that selectively abstracts an allylic hydrogen from the olefin substrate. This abstraction initiates a radical or concerted pathway that installs the oxygen functionality at the allylic position, preserving the carbon-carbon double bond which is crucial for the biological activity of many agrochemical targets. The ionic liquid, typically composed of a cation such as 1-butyl-3-methylimidazolium and an anion like tetrafluoroborate, stabilizes the transition states and polar intermediates, thereby suppressing side reactions such as polymerization or over-oxidation that commonly degrade yield in traditional solvents. This precise control over the reaction trajectory ensures that the impurity profile remains minimal, a critical factor for high-purity pyrethroid intermediates where trace contaminants can affect the efficacy of the final pesticide formulation.
Furthermore, the mechanism allows for tunable selectivity based on the stoichiometry of the oxidant, providing manufacturers with the flexibility to target alcohols, aldehydes, ketones, or carboxylic acids from the same starting material. When a limited amount of organic hydroperoxide is employed, the reaction tends to halt at the alcohol or aldehyde stage, whereas an excess of the oxidant drives the transformation towards the carboxylic acid derivative. This versatility is underpinned by the stability of the selenium species within the ionic liquid, which prevents the formation of inactive selenium precipitates that would otherwise terminate the catalytic cycle. The ability to modulate the oxidation state without changing the catalyst system simplifies inventory management and reduces the need for multiple dedicated production lines. For R&D teams, understanding this mechanistic nuance is vital for optimizing process parameters to maximize the yield of the specific intermediate required for their synthesis tree, ensuring that the commercial scale-up of complex organic oxidations proceeds with predictable and reproducible results.
How to Synthesize Chrysanthemic Acid Derivatives Efficiently
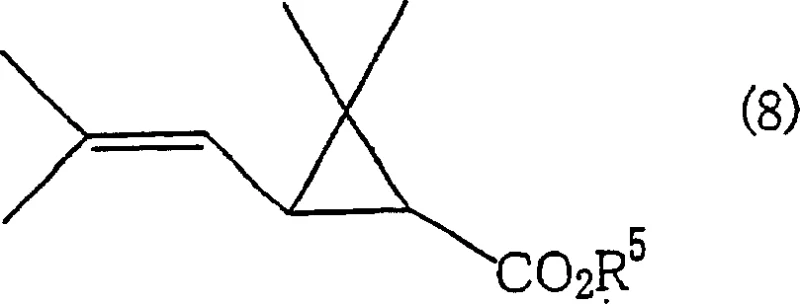
The practical application of this technology is best illustrated through the synthesis of chrysanthemic acid derivatives, which serve as the foundational acid moieties for a wide array of synthetic pyrethroid insecticides. The process begins with the preparation of the catalytic solution by dissolving a selenium compound, such as selenium dioxide, into a pre-dried ionic liquid like 1-butyl-3-methylimidazolium tetrafluoroborate, often with the addition of a co-catalyst like 3,4-dihydroxybenzoic acid to enhance activity. The olefin substrate, specifically a chrysanthemum monocarboxylate ester possessing the requisite allylic methyl group, is then introduced to the mixture along with a controlled dose of an organic hydroperoxide, such as tert-butyl hydroperoxide. The reaction is maintained at a moderate temperature range, typically between 60°C and 80°C, to ensure optimal kinetics while preventing thermal decomposition of the sensitive peroxide oxidant. Upon completion, the reaction mixture is subjected to a phase separation step where a non-polar solvent like n-hexane is added to extract the oxygenated product, leaving the selenium catalyst dissolved in the ionic liquid layer for immediate recycling.
- Prepare the catalytic system by mixing a selenium compound such as selenium dioxide with an alkyl-substituted imidazolium ionic liquid.
- Introduce the olefin substrate, specifically chrysanthemum monocarboxylate derivatives, and an organic hydroperoxide oxidant under controlled temperature conditions.
- Separate the product layer using a non-miscible solvent like n-hexane, allowing the ionic liquid catalyst layer to be recovered and reused for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this selenium-ionic liquid technology translates into tangible strategic advantages that extend beyond mere technical performance metrics. The primary economic driver is the drastic reduction in catalyst consumption and waste disposal costs, as the ability to recycle the ionic liquid phase multiple times diminishes the requirement for fresh selenium reagents and minimizes the volume of hazardous waste requiring treatment. This efficiency gain directly contributes to cost reduction in agrochemical manufacturing by lowering the variable cost per kilogram of the produced intermediate, thereby improving margin resilience in a competitive market. Additionally, the elimination of chlorinated solvents removes a significant regulatory burden and supply risk associated with the sourcing and handling of restricted substances, ensuring greater continuity of operations. The simplified workup procedure, which avoids complex distillation or chromatography for catalyst removal, shortens the overall batch cycle time, allowing facilities to increase throughput without capital expenditure on new reactor capacity. These factors combine to create a more robust and agile supply chain capable of responding swiftly to market demands for high-purity pyrethroid intermediates.
- Cost Reduction in Manufacturing: The implementation of a recyclable catalytic system fundamentally alters the cost structure by eliminating the need for stoichiometric reagents and reducing solvent purchase volumes. By recovering and reusing the ionic liquid-catalyst mixture, manufacturers avoid the recurring expense of selenium salts and the associated costs of neutralizing and disposing of heavy metal waste streams. This closed-loop approach ensures that the cost of goods sold remains stable even if raw material prices fluctuate, providing a hedge against market volatility. Furthermore, the energy savings derived from milder reaction conditions and simplified separation processes contribute to a lower overall carbon footprint, which is increasingly valued by downstream customers seeking sustainable supply partners.
- Enhanced Supply Chain Reliability: Relying on a process that utilizes stable and readily available ionic liquids reduces dependency on specialized solvents that may face supply constraints or regulatory bans. The robustness of the catalytic system ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification results that require reprocessing. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by global agrochemical formulators. By securing a manufacturing route that is less susceptible to regulatory changes regarding solvent usage, companies can guarantee long-term supply continuity for their key intermediates, strengthening their position as a reliable agrochemical intermediate supplier.
- Scalability and Environmental Compliance: The biphasic nature of the reaction system is inherently scalable, as the phase separation mechanics function effectively regardless of batch size, from pilot plant to full commercial production. This scalability is supported by the process alignment with green chemistry principles, which facilitates easier permitting and compliance with increasingly stringent environmental protection laws. The reduction in volatile organic compound (VOC) emissions and hazardous waste generation simplifies the environmental impact assessment process, accelerating the timeline for bringing new products to market. Consequently, manufacturers can expand capacity with greater confidence, knowing that the technology supports both economic growth and corporate sustainability goals without compromising on operational safety or regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oxidation technology, providing clarity for stakeholders evaluating its integration into their supply chains. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for industrial application. Understanding these details is essential for making informed decisions about process adoption and vendor selection.
Q: How does the ionic liquid system improve catalyst recovery compared to traditional solvents?
A: Unlike traditional volatile organic solvents where catalyst separation requires complex distillation or filtration, the ionic liquid forms a distinct phase that allows for simple decantation, enabling direct reuse of the selenium catalyst without significant loss of activity.
Q: What specific agrochemical intermediates can be produced using this oxidation method?
A: This method is particularly effective for synthesizing key pyrethroid intermediates, including various chrysanthemic acid derivatives, aldehydes, and carboxylic acids essential for next-generation insecticide formulations.
Q: Does this process eliminate the need for toxic chlorinated solvents?
A: Yes, the protocol replaces hazardous solvents like methylene chloride with stable ionic liquids and allows for the use of safer extraction solvents like n-hexane, significantly reducing environmental toxicity and regulatory compliance burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chrysanthemic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and a proven track record in process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the selenium-ionic liquid system are fully realized in a manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of oxygenated intermediate meets the exacting standards required for agrochemical synthesis. Our commitment to innovation allows us to offer customized solutions that optimize yield and cost-efficiency, positioning us as a strategic ally in your quest for superior supply chain performance.
We invite you to engage with our technical procurement team to discuss how this novel oxidation method can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to validate the quality and viability of our intermediates before committing to large-scale orders. Together, we can drive efficiency and sustainability in the production of essential agrochemical building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
