Industrial Scale Production of High-Purity Phyton Intermediates via Optimized Aldol Condensation
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to optimize the production of critical intermediates, particularly those serving as precursors for essential nutrients like Vitamin E. Patent CN1125805C introduces a transformative approach to synthesizing 6-methyl-3-hepten-2-one and its analogs, which are pivotal building blocks for phyton and isophytol. This technology addresses long-standing inefficiencies in cross-aldol condensation reactions by implementing a continuous addition strategy. Instead of traditional batch mixing which often leads to uncontrolled exotherms and poor selectivity, the patented process involves the sequential introduction of isovaleraldehyde and an aqueous base into a reactor containing acetone. This subtle yet powerful modification in process engineering ensures that the molar ratio of acetone to aldehyde remains favorable throughout the reaction timeline, drastically minimizing self-condensation byproducts. For R&D directors and process chemists, this represents a significant leap forward in achieving high-purity profiles without resorting to complex downstream purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C8 terpene ketones has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional pathways, such as the ethynylation of acetone followed by partial hydrogenation and Carroll rearrangement, involve multiple discrete unit operations that accumulate yield losses at every stage. Furthermore, alternative methods relying on the reaction of prenyl chloride with acetone necessitate the use of stoichiometric amounts of strong bases, resulting in the generation of massive quantities of salt waste that require costly disposal and environmental remediation. Other established techniques demand extreme reaction conditions, including temperatures exceeding 300°C and pressures up to 270 kg/cm², which mandate specialized high-pressure autoclaves and pose severe safety risks. These legacy processes not only inflate the capital expenditure required for plant infrastructure but also compromise the overall atom economy, making the final cost of goods sold prohibitively high for competitive markets.
The Novel Approach
In stark contrast, the methodology disclosed in CN1125805C offers a streamlined, industrially advantageous route that circumvents these traditional bottlenecks through precise kinetic control. By maintaining a large excess of acetone in the reaction mixture via continuous feeding, the process effectively suppresses the self-aldol condensation of isovaleraldehyde, which is a primary source of impurities in batch operations. This technique allows the reaction to proceed under mild conditions, typically between 40°C and 80°C, eliminating the need for energy-intensive heating or cryogenic cooling systems. The use of dilute aqueous alkali as a catalyst, rather than expensive organometallic reagents, further simplifies the workup procedure and reduces raw material costs significantly. This novel approach not only enhances the selectivity for the desired 6-methyl-3-hepten-2-one but also facilitates a direct one-pot conversion to saturated ketones when combined with hydrogenation, offering a versatile platform for producing various chain-extended analogs essential for the fragrance and vitamin sectors.
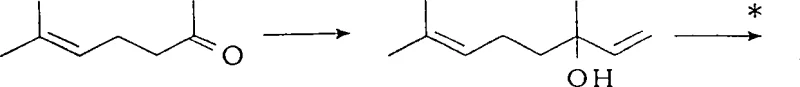
Mechanistic Insights into Cross-Aldol Condensation and Hydrogenation
The core chemical innovation lies in the manipulation of reaction kinetics during the cross-aldol condensation phase. In a standard batch scenario, the simultaneous presence of high concentrations of both carbonyl components and the base catalyst leads to a statistical distribution of products, including homo-aldol dimers of acetone and isovaleraldehyde. However, the continuous addition protocol ensures that at any given moment, the concentration of isovaleraldehyde is kept low relative to acetone. This kinetic bias favors the nucleophilic attack of the acetone enolate on the isovaleraldehyde carbonyl carbon, driving the equilibrium towards the cross-coupled product. Additionally, the gradual introduction of the base prevents localized high pH zones that could trigger rapid polymerization or degradation of the sensitive alpha,beta-unsaturated ketone product. Following the condensation, the subsequent hydrogenation step utilizes standard heterogeneous catalysts like palladium on carbon to selectively reduce the carbon-carbon double bond without affecting the carbonyl group, yielding the saturated 6-methyl-2-heptanone with exceptional purity.
Impurity control is inherently built into this mechanistic design, addressing a critical concern for regulatory compliance in API intermediate manufacturing. The suppression of self-condensation byproducts means that the crude reaction mixture contains significantly fewer heavy ends and oligomers, simplifying the distillation process required to isolate the target molecule. The patent data indicates that by optimizing the feed rates and maintaining the reaction temperature within the 70°C to 72°C range, the formation of the aldol adduct 6-methyl-4-hydroxyheptan-2-one is minimized to less than 5 mole percent. This high level of selectivity reduces the burden on downstream purification units, allowing for higher throughput and lower solvent consumption. Furthermore, the ability to tune the reaction by adjusting the molar ratio of acetone to aldehyde provides process engineers with a robust handle to manage exotherms and maintain consistent quality across different batch sizes, ensuring reproducibility from pilot plant to full commercial production.
How to Synthesize 6-Methyl-2-heptanone Efficiently
The synthesis of high-purity 6-methyl-2-heptanone begins with the preparation of the reaction vessel, typically a stainless steel autoclave equipped with precise metering pumps for reagent addition. Acetone is charged first and heated to the target operating temperature, creating a reservoir of nucleophile ready to react. The critical operational parameter is the synchronized continuous addition of isovaleraldehyde and the aqueous base solution, which must be controlled to match the consumption rate of the reactants. Detailed standardized synthetic steps see the guide below.
- Continuously add isovaleraldehyde and aqueous base into acetone to perform cross-aldol condensation, ensuring high selectivity for 6-methyl-3-hepten-2-one.
- Hydrogenate the resulting unsaturated ketone using a palladium catalyst under mild pressure to form 6-methyl-2-heptanone.
- Extend the carbon chain via vinylation or ethynylation followed by Carroll or Claisen rearrangement to synthesize Phyton or Isophytol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology translates into tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio; the process relies on commodity chemicals like acetone and isovaleraldehyde, which are globally available and subject to stable pricing dynamics, unlike specialized organometallic reagents that suffer from supply volatility. By eliminating the need for stoichiometric amounts of expensive bases or rare metal catalysts in the initial condensation step, the variable cost per kilogram of the intermediate is significantly reduced. This cost reduction in vitamin e precursor manufacturing is achieved without compromising quality, as the milder reaction conditions preserve the integrity of the molecular structure and reduce the formation of difficult-to-remove impurities that often plague cheaper synthetic routes.
- Cost Reduction in Manufacturing: The elimination of high-pressure and high-temperature requirements removes the necessity for specialized, capital-intensive reactor vessels, allowing production to occur in standard glass-lined or stainless steel equipment found in most multipurpose chemical plants. Furthermore, the avoidance of stoichiometric reagents like aluminum isopropoxide or Grignard reagents eliminates the generation of large volumes of inorganic salt waste, thereby reducing wastewater treatment costs and environmental compliance burdens. The streamlined workflow also decreases utility consumption, as the reaction proceeds efficiently at moderate temperatures without the need for extensive heating or cooling cycles, leading to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals mitigates the risk of supply disruptions that are common with niche specialty reagents. The robustness of the continuous addition process ensures consistent batch-to-batch quality, which is crucial for maintaining long-term contracts with downstream Vitamin E manufacturers. By reducing the complexity of the synthesis, the lead time for production campaigns can be shortened, allowing for more flexible inventory management and faster response to market demand fluctuations. This reliability makes the supplier a more attractive partner for global enterprises seeking to secure their supply chains against geopolitical or logistical uncertainties.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on heat management through feed rate control rather than complex pressure containment systems. This facilitates a smoother technology transfer from laboratory scale to multi-ton commercial production without significant re-engineering of the process parameters. From an environmental perspective, the use of aqueous base and the minimization of organic solvent usage align with green chemistry principles, reducing the facility's carbon footprint. The simplified workup procedure generates less hazardous waste, easing the regulatory burden and ensuring long-term sustainability of the manufacturing operation in increasingly strict environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous aldol condensation technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on feasibility and performance metrics. Understanding these details is essential for stakeholders evaluating the integration of this route into their existing manufacturing portfolios.
Q: How does the continuous addition method improve selectivity in aldol condensation?
A: By continuously adding isovaleraldehyde and base into excess acetone, the local concentration of acetone remains high, suppressing self-condensation side reactions and significantly improving the yield of the desired cross-aldol product.
Q: What are the equipment requirements for this synthesis compared to conventional methods?
A: Unlike conventional methods requiring high temperature and pressure or expensive stoichiometric reagents like aluminum isopropoxide, this process operates under mild conditions with standard stainless steel reactors, reducing capital expenditure.
Q: Can this process be scaled for commercial production of Vitamin E intermediates?
A: Yes, the process utilizes readily available raw materials like acetone and isovaleraldehyde and avoids complex purification steps, making it highly suitable for large-scale commercial manufacturing of phyton and isophytol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methyl-2-heptanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply chain for vitamins and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot studies to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement advanced synthetic strategies, such as the continuous addition aldol condensation described in CN1125805C, allows us to offer cost-effective solutions without sacrificing the quality required for pharmaceutical and nutraceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits associated with switching to our supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring that your project moves forward with the most reliable and efficient chemical foundation available in the market today.
