Advanced Synthesis of Unnatural Ceramide Intermediates for High-Purity Glycolipid Manufacturing
Advanced Synthesis of Unnatural Ceramide Intermediates for High-Purity Glycolipid Manufacturing
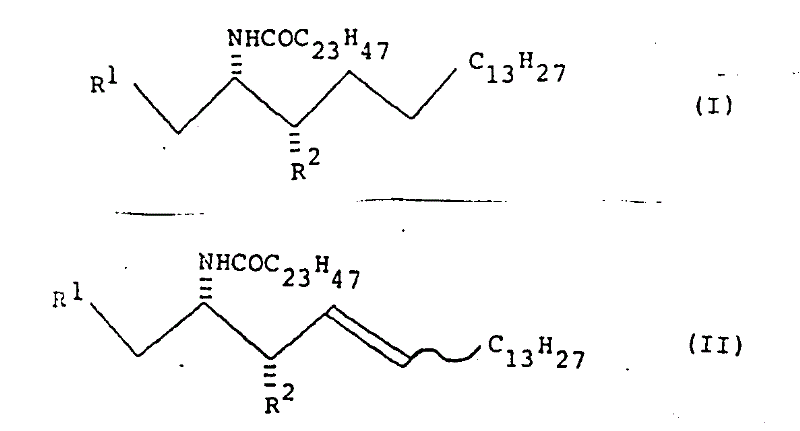
The pharmaceutical and biotechnology sectors are increasingly recognizing the critical role of glycolipids and gangliosides as potent tumor markers and molecular identifiers in oncology research. Patent CN1013440B introduces a groundbreaking methodology for synthesizing unnatural ceramide related compounds, specifically targeting the production of high-purity intermediates like (2S, 3S)-2-tetracosanamido-octadecane-1,3-diol. Unlike traditional approaches that struggle with isolation and purification from natural sources, this invention provides a robust chemical synthesis route that bypasses the need for complex optical resolution. By leveraging a chiral pool strategy starting from readily available D-glucose, the process ensures exceptional stereochemical fidelity, which is paramount for the biological activity of the resulting glycolipids. This technological leap addresses the longstanding challenge of producing enantiomerically pure sphingosine derivatives efficiently.
For procurement managers and supply chain directors, the implications of this patent extend far beyond the laboratory bench. The ability to synthesize these complex lipid structures without relying on scarce natural extracts or inefficient racemic separations translates directly into enhanced supply chain stability and potential cost optimization. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this synthesis allows for better forecasting of raw material requirements and production timelines. The patent outlines multiple pathways, including the use of ethyl vinyl ether protecting groups, offering flexibility in manufacturing strategies to mitigate risks associated with single-route dependencies. This adaptability is crucial for maintaining continuous supply lines for high-value oncology research materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of ceramide moieties has been fraught with significant technical hurdles that impact both yield and purity. Conventional methods, such as those proposed by Grob, often relied on non-chiral starting materials, resulting in racemic mixtures that required subsequent optical resolution. This resolution step is notoriously inefficient, theoretically discarding up to 50% of the synthesized material while incurring additional costs for chiral resolving agents and separation processes. Furthermore, isolating natural sphingosine glycolipids from mammalian cells is extremely difficult and yields are negligible, making it impossible to meet the demands of commercial-scale drug development. The presence of impurities and isomers in racemic syntheses can also complicate downstream biological testing, leading to ambiguous data in tumor marker identification studies.
The Novel Approach
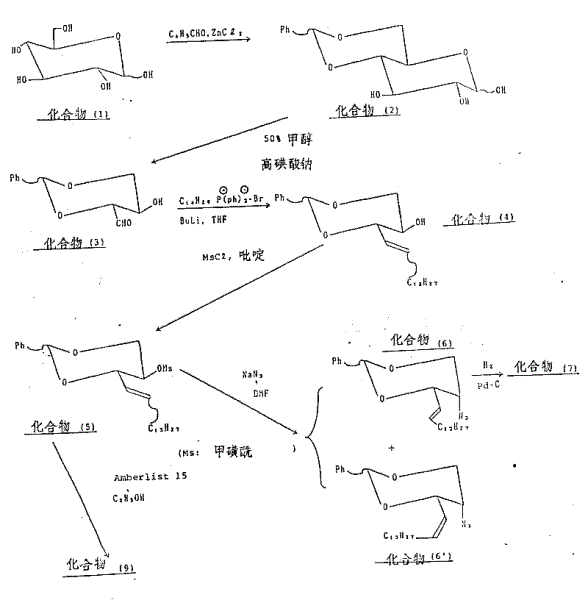
The novel approach detailed in CN1013440B fundamentally shifts the paradigm by utilizing a chiral pool synthesis starting from D-glucose. This strategy capitalizes on the inherent chirality of the sugar molecule to pre-establish the necessary stereocenters, effectively rendering the optical resolution step obsolete. The process involves a series of highly selective transformations, including zinc chloride-catalyzed acetal formation and sodium metaperiodate oxidative cleavage, which preserve the optical integrity of the intermediate. By avoiding the generation of unwanted enantiomers from the outset, the overall atom economy of the process is significantly improved. This methodological refinement not only enhances the final product's purity but also streamlines the manufacturing workflow, reducing the number of unit operations required to reach the target unnatural ceramide structure.
Mechanistic Insights into Stereoselective Azidation and Acylation
A critical component of this synthesis is the precise control of stereochemistry at the C2 and C3 positions of the sphingosine backbone. The mechanism relies heavily on an SN2 nucleophilic substitution reaction where a secondary mesylate group is displaced by an azide ion. Since SN2 reactions proceed with Walden inversion, the configuration at the C2 center is inverted from (2R) in the precursor to the desired (2S) in the product. This mechanistic certainty is vital for R&D directors who require batch-to-batch consistency in the stereochemical profile of their intermediates. Following the azidation, the azide group is reduced to an amine, which is then acylated with lignoceric acid (tetracosanoic acid) to form the ceramide linkage. The use of specific catalysts like Amberlist 15 for deprotection steps further ensures that the sensitive amide bond remains intact while removing acid-labile protecting groups.
Furthermore, the patent describes alternative pathways for controlling the geometry of the double bond in the long hydrocarbon chain, distinguishing between E (trans) and Z (cis) isomers. For instance, the use of Lindlar catalyst during hydrogenation allows for the selective formation of cis-alkenes, whereas other reduction conditions favor trans-alkenes. This level of control is essential because the biological function of glycolipids is often dependent on the specific geometry of their lipid tails. The ability to toggle between these isomers using defined catalytic conditions demonstrates a sophisticated understanding of reaction engineering. Such mechanistic depth ensures that the final unnatural ceramide compounds can be tailored to mimic specific natural gangliosides or to serve as stable analogs for structural biology studies.
How to Synthesize (2S, 3S)-2-Tetracosanamido-octadecane-1,3-diol Efficiently
The synthesis of this high-value intermediate requires a disciplined approach to reaction conditions and purification to maintain the stringent purity specifications demanded by the pharmaceutical industry. The process begins with the protection of D-glucose, followed by chain elongation and functional group manipulation. Each step, from the Wittig olefination to the final deacetylation, must be monitored closely to prevent side reactions that could compromise the chiral integrity of the molecule. The detailed standardized synthetic steps provided in the patent serve as a robust foundation for process chemists looking to implement this route. For a comprehensive guide on the specific reagents, temperatures, and workup procedures required to execute this synthesis successfully, please refer to the structured protocol below.
- Protect D-glucose with benzaldehyde using ZnCl2 catalyst, followed by oxidative cleavage with sodium metaperiodate to generate the chiral aldehyde intermediate.
- Perform a Wittig reaction with a long-chain phosphonium salt to establish the carbon backbone, followed by mesylation and stereospecific azidation to invert the C2 configuration.
- Reduce the azide to an amine, acylate with lignoceric acid, and finally remove the protecting groups using Amberlist 15 to yield the final diol product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers substantial strategic advantages for organizations focused on cost reduction in pharmaceutical intermediates manufacturing. By eliminating the need for optical resolution, the process inherently reduces raw material consumption and waste generation, leading to a more sustainable and economically viable production model. The reliance on commodity chemicals like D-glucose and lignoceric acid ensures that the supply chain is not vulnerable to the volatility often associated with specialized chiral reagents. Additionally, the robustness of the reaction conditions, many of which can be performed at ambient or moderate temperatures, lowers the energy footprint of the manufacturing process. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of global drug development pipelines.
- Cost Reduction in Manufacturing: The elimination of the optical resolution step represents a significant saving in both material costs and processing time. Traditional methods lose at least half of the product during resolution, whereas this chiral pool approach retains the theoretical yield throughout the synthesis. Furthermore, the use of recyclable solid acid catalysts like Amberlist 15 simplifies the workup procedure, reducing the consumption of solvents and neutralizing agents. This streamlined workflow translates directly into lower operating expenses and a reduced cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral starting materials can often be a bottleneck in fine chemical manufacturing. However, this process utilizes D-glucose, a globally abundant and inexpensive renewable resource, as the primary chiral source. This decouples the production of complex ceramide intermediates from the supply constraints of exotic chiral pools. Moreover, the synthetic route avoids the use of highly toxic or regulated reagents that might face shipping restrictions, thereby ensuring smoother logistics and uninterrupted delivery schedules for international clients.
- Scalability and Environmental Compliance: The reactions described, such as the Wittig reaction and acylation, are well-established in industrial organic synthesis and are known to scale effectively from kilogram to multi-ton quantities. The process generates fewer byproducts compared to racemic synthesis, simplifying waste treatment and disposal. The ability to produce these compounds on a commercial scale without generating massive amounts of unwanted enantiomeric waste aligns with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these unnatural ceramide compounds. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these intermediates into their research or production workflows. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the chiral pool method superior to racemic resolution for ceramide synthesis?
A: The chiral pool method utilizing D-glucose inherently establishes the correct stereochemistry at C3 and C4, eliminating the need for difficult and yield-limiting optical resolution steps required in traditional racemic syntheses.
Q: How is the (2S, 3S) stereochemistry strictly controlled in this process?
A: The process relies on an SN2 nucleophilic substitution where a mesylate group is displaced by an azide ion. This inversion mechanism ensures the precise conversion from the (2R, 3S) precursor to the desired (2S, 3S) configuration.
Q: Can this synthesis be scaled for commercial production of glycolipid markers?
A: Yes, the route utilizes robust reactions such as Wittig olefination and standard acylation which are well-suited for scale-up, avoiding exotic reagents that typically hinder large-scale pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Unnatural Ceramide Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our technical team is adept at optimizing complex synthetic routes like the one described in CN1013440B to achieve stringent purity specifications required for oncology research. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the stereochemical integrity and chemical purity of every batch of unnatural ceramide intermediates we produce. Our commitment to quality assurance guarantees that the materials you receive are fit for purpose, whether for preclinical studies or clinical trial manufacturing.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that can help streamline your own internal development processes. Let us leverage our expertise in chiral synthesis to support your mission of developing next-generation glycolipid-based therapeutics and diagnostic markers.
