Scalable Synthesis of 3-Alkyl-Substituted Indoles for Commercial Pharmaceutical Production
The pharmaceutical and fine chemical industries continuously demand robust synthetic routes for complex heterocyclic compounds, particularly indole derivatives which serve as critical scaffolds for numerous bioactive molecules. Patent CN103304466A discloses a highly efficient synthetic method for 3-alkyl-substituted indole compounds, addressing significant limitations found in traditional methodologies. This innovation is particularly relevant for the production of key intermediates such as 3-(4-chlorobutyl)-5-cyanoindole, a precursor for the antidepressant Vilazodone. The disclosed method employs a strategic four-step sequence involving nitrogen protection, Friedel-Crafts acylation, mild reduction, and deprotection. By shifting away from hazardous reducing agents like lithium aluminum hydride, this technology offers a safer, more stable, and economically viable pathway for manufacturing high-purity pharmaceutical intermediates. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for optimizing supply chain resilience and reducing overall production costs in the competitive landscape of API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-alkyl-substituted indoles has relied heavily on direct reduction methods using powerful hydride reagents such as lithium aluminum hydride or borane complexes. While these traditional bibliographic methods can achieve the desired transformation, they are fraught with significant operational hazards and technical inefficiencies that hinder commercial scalability. The use of lithium aluminum hydride, for instance, requires strictly anhydrous conditions and poses severe fire and explosion risks upon exposure to moisture, necessitating expensive specialized equipment and rigorous safety protocols. Furthermore, these aggressive reducing agents often lack chemoselectivity, leading to the reduction of other sensitive functional groups present on the indole ring or side chains, which results in complex impurity profiles. The purification of the final product becomes exceedingly difficult, often requiring column chromatography which is impractical for multi-kilogram or ton-scale production. Consequently, the overall yields are frequently low, sometimes dropping below thirty percent, and the process stability is poor, making it unsuitable for consistent commercial manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a refined synthetic strategy that prioritizes safety, selectivity, and scalability without compromising on yield. This method utilizes a sodium borohydride-trifluoroacetic acid system for the critical reduction step, which operates under much milder conditions compared to traditional hydride reagents. The reaction can be conducted at temperatures ranging from minus ten to ten degrees Celsius, significantly reducing the thermal load and energy consumption of the process. By incorporating a nitrogen protection step prior to acylation, the method ensures that the electrophilic substitution occurs exclusively at the 3-position, thereby eliminating regio-isomeric impurities. The subsequent deprotection under alkaline conditions is straightforward and utilizes common industrial bases, avoiding the need for exotic reagents. This holistic improvement in the reaction design translates directly to enhanced process robustness, allowing for reliable production of high-purity intermediates suitable for stringent pharmaceutical applications.
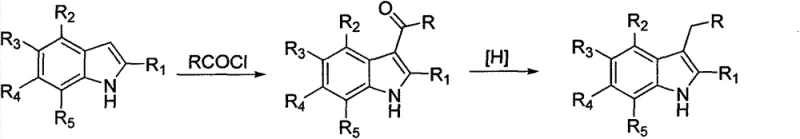
Mechanistic Insights into Protection-Assisted Acylation and Reduction
The core innovation of this synthetic route lies in the precise control of reactivity through a protection-deprotection strategy coupled with a chemoselective reduction mechanism. The process initiates with the protection of the indole nitrogen atom using reagents such as p-toluenesulfonyl chloride or benzenesulfonyl chloride. This step is crucial as it deactivates the nitrogen lone pair, preventing it from interfering with the subsequent Lewis acid-catalyzed Friedel-Crafts acylation. Without this protection, the nitrogen could coordinate with the Lewis acid or undergo N-acylation, leading to a mixture of products. Once protected, the indole ring undergoes acylation with an acyl chloride in the presence of a Lewis acid like aluminum chloride or zinc chloride. The electron-withdrawing nature of the protecting group actually enhances the regioselectivity for the 3-position, ensuring that the carbonyl group is introduced exactly where needed for the final alkyl chain. This level of control is fundamental for R&D teams aiming to minimize impurity formation and streamline the purification workflow.
Following acylation, the carbonyl group at the 3-position is reduced to a methylene group using a sodium borohydride and trifluoroacetic acid system. Mechanistically, the trifluoroacetic acid activates the sodium borohydride to generate a more potent reducing species in situ, capable of reducing the ketone without affecting other sensitive functionalities like halogens or nitriles that might be present on the aromatic ring. This is a distinct advantage over stronger reducing agents which might indiscriminately reduce multiple functional groups. The reaction proceeds smoothly at low temperatures, and the by-products are generally water-soluble salts that are easily removed during the aqueous work-up. Finally, the protecting group is cleaved under alkaline conditions, typically using sodium hydroxide in methanol or ethanol, regenerating the free indole nitrogen. This final step is high-yielding and clean, ensuring that the final 3-alkyl-substituted indole product meets the high-purity specifications required for pharmaceutical use.
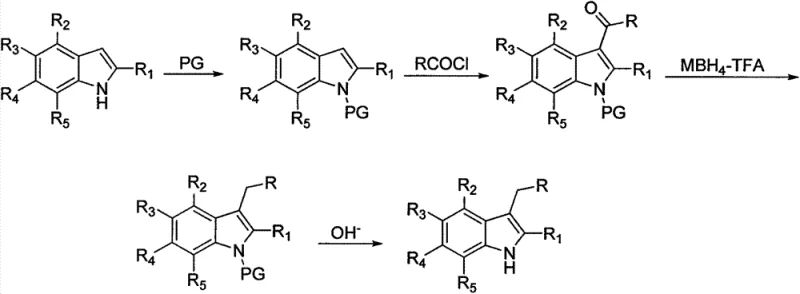
How to Synthesize 3-Alkyl-Substituted Indole Efficiently
The implementation of this synthetic route requires careful attention to reaction conditions and reagent stoichiometry to maximize efficiency and safety. The process begins with the dissolution of the starting indole in a chlorinated solvent such as dichloromethane, followed by the addition of a base to facilitate the protection step. Once the protected intermediate is isolated, it is subjected to Friedel-Crafts acylation under controlled low-temperature conditions to manage the exotherm. The subsequent reduction step is the most critical, requiring the batch-wise addition of sodium borohydride to a mixture of the ketone intermediate and trifluoroacetic acid, maintaining the temperature below zero degrees Celsius to prevent side reactions. Detailed standardized synthesis steps see the guide below.
- Protect the nitrogen atom on the indole ring using a protective reagent such as p-toluenesulfonyl chloride to form Product 1.
- Conduct a Friedel-Crafts acylation reaction between Product 1 and acyl chloride to introduce a carbonyl group at the 3-position.
- Reduce the 3-position carbonyl group to methylene using sodium borohydride and trifluoroacetic acid to obtain Product 2.
- Remove the protective group from Product 2 under alkaline conditions to yield the target 3-alkyl-substituted indole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthetic method offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like lithium aluminum hydride drastically simplifies the safety compliance requirements for the manufacturing facility, reducing the need for specialized explosion-proof infrastructure and lowering insurance and operational overheads. The use of common, commercially available reagents such as sodium borohydride and trifluoroacetic acid ensures a stable supply chain, as these materials are not subject to the same geopolitical or logistical constraints as exotic catalysts. Furthermore, the high selectivity of the reaction minimizes the generation of waste and by-products, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and treatment. This process is inherently designed for scalability, allowing manufacturers to ramp up production from pilot scale to multi-ton commercial volumes without encountering the bottlenecks typical of unstable chemistries.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reducing agents with cost-effective sodium borohydride significantly lowers the raw material costs per kilogram of the final product. Additionally, the simplified purification process, which avoids complex chromatography and relies on crystallization and extraction, reduces solvent consumption and labor hours. The high yield achieved through improved selectivity means less starting material is wasted, directly improving the overall material efficiency and reducing the cost of goods sold. These cumulative effects result in a more economically competitive product without sacrificing quality.
- Enhanced Supply Chain Reliability: By utilizing robust chemistry that tolerates minor variations in reaction conditions, the risk of batch failure is significantly minimized, ensuring consistent on-time delivery to customers. The reliance on widely available industrial chemicals rather than specialized custom reagents mitigates the risk of supply disruptions. This stability is crucial for long-term supply agreements with pharmaceutical companies that require guaranteed continuity of supply for their drug pipelines. The process reliability also allows for better inventory planning and reduced safety stock requirements.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous work-up procedures make this process highly amenable to scale-up in standard stainless steel reactors. The reduction in hazardous waste generation simplifies environmental permitting and compliance reporting. The ability to run the reaction at near-ambient or slightly cooled temperatures reduces the energy footprint of the manufacturing process. This alignment with green chemistry principles enhances the sustainability profile of the supply chain, a key metric for modern corporate procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 3-alkyl-substituted indoles. These answers are derived from the specific technical advantages and operational parameters detailed in the patent literature, providing clarity on the feasibility and benefits of this manufacturing route. Understanding these details helps stakeholders make informed decisions regarding process adoption and supplier selection.
Q: Why is the sodium borohydride-trifluoroacetic acid system preferred over lithium aluminum hydride?
A: The sodium borohydride-trifluoroacetic acid system offers superior operational safety and reaction stability compared to lithium aluminum hydride, which poses significant fire and explosion risks. This mild reduction method avoids the generation of hazardous by-products and simplifies the purification process, making it ideal for large-scale industrial applications.
Q: How does the nitrogen protection step improve the overall yield?
A: Protecting the nitrogen atom prevents unwanted side reactions at the indole nitrogen during the acylation and reduction steps. This ensures high regioselectivity for the 3-position substitution, significantly reducing impurity formation and facilitating easier downstream purification.
Q: Is this synthetic route suitable for commercial scale-up?
A: Yes, the process utilizes common industrial reagents and avoids extreme conditions or hazardous catalysts. The mild reaction temperatures and straightforward work-up procedures make this route highly scalable and compliant with standard environmental and safety regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkyl-Substituted Indole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting complex synthetic routes, such as the protection-assisted reduction method described, to meet the rigorous demands of the global pharmaceutical market. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 3-alkyl-substituted indole meets or exceeds the quality standards required for API intermediate usage. Our commitment to process safety and environmental responsibility ensures that your supply chain remains resilient and compliant with international regulations.
We invite you to collaborate with us to optimize your sourcing strategy for high-purity pharmaceutical intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a stable, cost-effective, and high-quality supply of critical chemical building blocks for your drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
