Optimizing Terpene-Based Cyclohexanol Production: A Technical Breakthrough in Catalytic Hydrogenation for Global Fragrance Supply Chains
The global demand for high-quality synthetic sandalwood alternatives has driven intense research into optimizing the production of terpene-based cyclohexanols, key intermediates that mimic the precious olfactory characteristics of natural sandalwood oil. Patent CN102421737A introduces a transformative methodology for preparing these valuable compounds through the hydrogenation of terpene-based phenols. This technical disclosure marks a significant departure from legacy industrial practices by utilizing a specialized Raney nickel-type catalyst doped with iron and chromium. The innovation lies not merely in the catalyst composition but in the profound impact this modification has on the thermodynamic and kinetic parameters of the reaction. By enabling the process to proceed under significantly milder conditions—specifically lower temperatures and drastically reduced hydrogen pressures—this technology addresses critical bottlenecks in scalability and safety that have long plagued the fine chemical manufacturing sector. For R&D directors and process engineers, this represents a viable pathway to enhance yield consistency while minimizing the degradation of sensitive terpene structures.
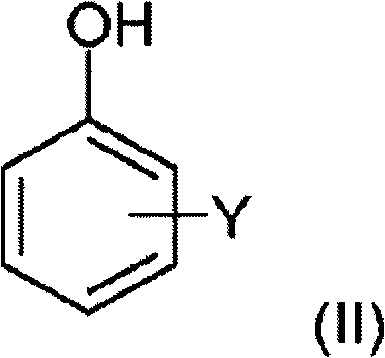
The structural versatility of the substrates applicable to this novel hydrogenation protocol is extensive, encompassing a wide range of terpene-substituted phenols. As illustrated in the general formula provided in the patent documentation, the process accommodates various substituents on the aromatic ring, including hydroxyl, alkoxy, or hydrogen groups, coupled with bulky bicyclic terpene moieties such as bornyl, isobornyl, or camphenyl groups. This broad substrate tolerance is crucial for manufacturers aiming to produce a diverse portfolio of fragrance ingredients from a single, robust platform technology. The ability to handle complex mixtures of positional and terpene isomers without compromising the integrity of the final olfactory profile underscores the robustness of the catalytic system. Furthermore, the retention of the terpene skeleton's stereochemistry during the rigorous conditions of hydrogenation ensures that the synthetic end-product closely matches the sensory attributes of its natural counterparts, a non-negotiable requirement for premium fragrance houses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of terpene-based cyclohexanols has been hindered by the severe operational constraints dictated by prior art technologies, most notably those described in US Patent 4,061,686. In these conventional processes, the hydrogenation of terpene phenols necessitates extreme reaction conditions that pose significant engineering challenges and safety risks. Specifically, the legacy methodology requires operating temperatures soaring between 200-300°C, with a preferred range of 225-250°C, coupled with exorbitantly high hydrogen pressures ranging from 200 to 250 bar. Such extreme parameters demand specialized, high-cost reactor vessels capable of withstanding immense stress, thereby inflating capital expenditure (CAPEX) for any facility attempting to adopt the technology. Moreover, the catalyst loading in these traditional methods is inefficiently high, often requiring 3-20 weight percent of standard Raney nickel relative to the substrate mass. This excessive catalyst usage not only drives up raw material costs but also complicates downstream processing, as the removal and disposal of large quantities of pyrophoric metal residues become a logistical and environmental burden.
The Novel Approach
In stark contrast to the brute-force approach of earlier patents, the methodology disclosed in CN102421737A achieves superior conversion efficiencies through intelligent catalyst design rather than sheer thermal and pressure intensity. The core innovation involves the use of a Raney nickel-type catalyst that retains residual aluminum and is metallurgically doped with specific quantities of iron and chromium. This modified catalyst system facilitates the hydrogenation reaction at markedly lower temperatures, typically extending up to 180-250°C, and preferably within the 190-220°C window. Even more impactful is the reduction in hydrogen pressure, which drops precipitously from the hundreds of bars required by conventional methods to a manageable 18-30 bar, with an optimal range of 20-25 bar. This drastic reduction in pressure fundamentally alters the safety profile and economic feasibility of the process, allowing it to be executed in standard high-pressure reactors rather than ultra-high-pressure vessels. Additionally, the catalyst loading is optimized to just 1-10 weight percent, significantly reducing the metallic waste stream and improving the overall atom economy of the synthesis.
Mechanistic Insights into Fe-Cr Doped Raney Nickel Hydrogenation
The enhanced performance of the doped Raney nickel catalyst can be attributed to subtle yet profound changes in the electronic and geometric properties of the active nickel sites. The introduction of iron and chromium dopants into the nickel-aluminum alloy precursor prior to alkaline leaching creates a heterogeneous surface with modified adsorption characteristics for both hydrogen and the aromatic substrate. The residual aluminum, maintained at levels between 5-10 weight percent, plays a critical role in stabilizing the porous microstructure of the catalyst, preventing sintering and loss of surface area under the elevated temperatures of the reaction. The iron and chromium components, present in ranges of 1-5 weight percent each, likely act as promoters that facilitate the dissociation of molecular hydrogen and its subsequent transfer to the aromatic ring. This synergistic effect lowers the activation energy barrier for the hydrogenation step, thereby permitting the reaction to proceed rapidly even at the reduced temperatures of 180-250°C. Such mechanistic efficiency ensures that the reaction kinetics are favorable without necessitating the thermal degradation pathways that often lead to unwanted by-products in high-temperature processes.
From an impurity control perspective, the specificity of this catalytic system is paramount for maintaining the quality of the final fragrance ingredient. One of the primary concerns in terpene chemistry is the potential for skeletal rearrangement or isomerization of the sensitive terpene moiety under harsh acidic or thermal conditions. The patent data explicitly confirms that during the hydrogenation process using this doped catalyst, the terpene-based units do not undergo isomerization. This selectivity is vital because the olfactory properties of sandalwood analogs are heavily dependent on the precise stereochemistry of the terpene group attached to the cyclohexanol ring. By avoiding the extreme conditions of 200-300°C and 200+ bar pressure, the process minimizes thermal stress on the molecule, thereby preserving the integrity of the bornyl, isobornyl, or camphenyl groups. Consequently, the resulting mixture of cyclohexanol isomers exhibits the desired sweet, woody aroma without the off-notes often associated with thermally degraded by-products, ensuring a high-purity profile suitable for direct application in fine fragrances.
How to Synthesize Terpene-Based Cyclohexanols Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of unit operations that are well-suited for batch processing in standard fine chemical facilities. The process begins with the preparation of the terpene phenol substrate, typically achieved via the Friedel-Crafts alkylation of phenol or guaiacol with camphene in the presence of a Lewis acid catalyst such as K10 clay or boron trifluoride complexes. Once the phenolic precursor is isolated and purified, it is charged into a stainless steel autoclave along with the proprietary Fe-Cr doped Raney nickel catalyst, usually supplied as an alkaline aqueous suspension to mitigate pyrophoric risks. The detailed standardized synthesis steps, including precise heating ramps, purging protocols to remove water and light alcohols, and specific workup procedures involving filtration and vacuum distillation, are outlined in the technical guide below.
- Preparation of Substrate: Condense phenol or guaiacol with camphene using a Lewis acid catalyst (e.g., K10 clay or BF3 complex) to generate the terpene-based phenolic precursor.
- Catalytic Hydrogenation: Load the terpene phenol substrate and the Fe-Cr doped Raney nickel catalyst (1-10 wt%) into a stainless steel autoclave. Pressurize with hydrogen to 18-30 bar and heat to 180-250°C.
- Workup and Purification: Upon completion, cool the reactor, filter off the catalyst, and purify the resulting terpene cyclohexanol mixture via vacuum distillation to isolate the desired olfactory active isomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented hydrogenation technology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift from ultra-high-pressure conditions to moderate pressure regimes significantly lowers the barrier to entry for manufacturing, as it eliminates the need for specialized, exorbitantly expensive reactors rated for 250 bar service. This compatibility with standard industrial equipment accelerates the timeline for capacity expansion and reduces the capital intensity of new production lines. Furthermore, the reduction in catalyst loading from nearly 20% to as low as 1-3% drastically cuts the consumption of nickel, a commodity metal subject to price volatility. This reduction in metal usage not only lowers direct material costs but also simplifies the waste management workflow, as there is significantly less spent catalyst to treat, filter, and dispose of in compliance with environmental regulations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in energy consumption and equipment maintenance costs associated with milder operating conditions. Operating at 18-30 bar instead of 200-250 bar means that compression costs for hydrogen are exponentially lower, and the mechanical stress on reactor seals and vessels is minimized, leading to longer asset lifecycles and reduced downtime for maintenance. Additionally, the lower temperature profile reduces the thermal load on heating systems, further contributing to utility savings. The elimination of extreme conditions also implies a safer working environment, potentially lowering insurance premiums and regulatory compliance costs associated with high-pressure hazardous operations. By removing the need for excessive catalyst quantities, the process also reduces the cost burden of catalyst procurement and the downstream costs associated with metal recovery or disposal services.
- Enhanced Supply Chain Reliability: The robustness of the Fe-Cr doped catalyst ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply to downstream fragrance blenders. Unlike conventional methods that may suffer from variability due to the sensitivity of extreme conditions, this optimized process offers a wider operating window, making it more forgiving to minor fluctuations in feedstock quality or utility availability. The use of commercially available starting materials like camphene and phenol, combined with a catalyst that can be prepared from standard nickel-aluminum alloys with simple doping, ensures that the raw material supply chain is resilient and not dependent on exotic or single-source reagents. This decentralization of supply risk is essential for long-term strategic planning and securing reliable contracts with major multinational clients who demand guaranteed continuity of supply.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the use of liquid-phase hydrogenation in standard autoclaves, a unit operation familiar to most contract development and manufacturing organizations (CDMOs). The milder conditions reduce the engineering complexity typically associated with scaling high-pressure exothermic reactions, thereby shortening the tech transfer timeline. From an environmental standpoint, the process aligns with green chemistry principles by improving atom economy through lower catalyst usage and reducing energy intensity. The ability to operate without solvent or with minimal alcohol solvents further simplifies the recovery of volatile organic compounds (VOCs), aiding in compliance with increasingly stringent global emissions standards. The reduced generation of heavy metal waste also eases the burden on wastewater treatment facilities, positioning the manufacturer as a sustainable partner in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this hydrogenation technology, derived directly from the experimental data and comparative examples provided in the patent literature. These insights are intended to clarify the operational boundaries and performance expectations for technical teams evaluating this process for potential adoption or licensing. Understanding these nuances is critical for accurately forecasting production yields and defining quality control specifications for the final terpene cyclohexanol products.
Q: How does the Fe-Cr doped Raney nickel catalyst improve upon conventional hydrogenation methods?
A: The incorporation of iron and chromium dopants into the Raney nickel lattice significantly enhances catalytic activity and selectivity. This modification allows the hydrogenation of terpene phenols to proceed at substantially lower temperatures (180-250°C vs. 200-300°C) and pressures (18-30 bar vs. 200-250 bar) compared to prior art, while maintaining high conversion rates and preserving the delicate olfactory properties of the final product.
Q: What are the specific operating conditions required for this hydrogenation process?
A: The process operates in the liquid phase using a catalyst loading of 1-10 weight percent relative to the substrate. The reaction is conducted under a hydrogen pressure of 18-30 bar, preferably 20-25 bar, and at temperatures ranging from 180-250°C. These milder conditions reduce energy consumption and equipment stress compared to traditional high-pressure hydrogenation protocols.
Q: Does this process affect the isomeric composition of the terpene moiety?
A: No, the process is designed to be highly selective for the aromatic ring hydrogenation without inducing isomerization of the terpene unit. Analytical data confirms that the terpene-based units (such as bornyl, isobornyl, or camphenyl groups) remain intact during the reaction, ensuring the final cyclohexanol mixture retains the specific scent profile required for high-end fragrance applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terpene-Based Cyclohexanols Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent examples can be reliably replicated on an industrial scale. We understand that the production of high-value fragrance intermediates demands stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting olfactory standards of the global perfume industry. Our state-of-the-art facilities are equipped to handle the specific safety requirements of hydrogenation processes, including the safe handling of pyrophoric catalysts and high-pressure gases, providing a secure environment for your intellectual property and product quality.
We invite you to collaborate with us to leverage this advanced hydrogenation technology for your supply chain needs. Whether you are looking to optimize an existing route or develop a new cost-effective source for sandalwood alternatives, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project milestones and quality targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
