Advanced One-Step Synthesis of Beta-Hydroxyethyl Cinnamaldehyde for Commercial Scale-Up
Introduction to Next-Generation Cinnamaldehyde Derivative Synthesis
The chemical landscape for functionalized cinnamaldehyde derivatives is undergoing a significant transformation driven by the urgent need for more sustainable and efficient synthetic pathways. As detailed in the recent patent CN111302914B, a groundbreaking methodology has been established for the preparation of beta-hydroxyethyl cinnamaldehyde, a critical intermediate widely utilized in the pharmaceutical and fragrance industries. This innovation addresses long-standing challenges in organic synthesis by leveraging the inherent reactivity of tetrahydrofuran (THF), a common and inexpensive solvent, transforming it from a passive medium into an active carbon source. The ability to construct complex molecular architectures in a single operational step represents a paradigm shift, offering substantial implications for process chemistry and industrial manufacturing scalability.
Traditionally, the synthesis of such hydroxy-functionalized aldehydes required cumbersome multi-step sequences involving hazardous reagents and strict temperature controls. The novel approach described in this patent utilizes a copper-catalyzed system that facilitates a direct coupling between benzaldehyde derivatives and THF. This not only simplifies the operational workflow but also drastically reduces the generation of chemical waste. For R&D directors and process engineers, this development signals a move towards atom-economical processes that align with green chemistry principles while maintaining high standards of product purity and structural integrity required for downstream applications in drug discovery and fine chemical production.
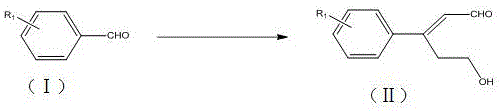
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the construction of the beta-hydroxyethyl side chain on cinnamaldehyde scaffolds was a laborious endeavor characterized by low overall efficiency and high operational costs. Conventional routes, as illustrated in earlier literature and prior art, typically necessitated a multi-step sequence starting with bromination reactions followed by reduction and protection/deprotection strategies. These traditional pathways often relied on stoichiometric amounts of hazardous halogenating agents and strong reducing metals, creating significant safety hazards and environmental burdens. Furthermore, the accumulation of impurities at each synthetic stage required rigorous purification protocols, leading to substantial material loss and extended production timelines that are incompatible with modern just-in-time manufacturing demands.
The reliance on pre-functionalized starting materials in older methodologies also imposed severe limitations on substrate scope and economic viability. Each additional synthetic step introduced potential points of failure, increasing the risk of batch-to-batch variability and complicating the regulatory approval process for pharmaceutical intermediates. The use of expensive reagents and the generation of large volumes of saline waste streams further exacerbated the cost structure, making the commercial production of high-purity beta-hydroxyethyl cinnamaldehyde economically challenging. Consequently, there was a critical industry need for a streamlined approach that could bypass these inefficiencies while delivering consistent quality.
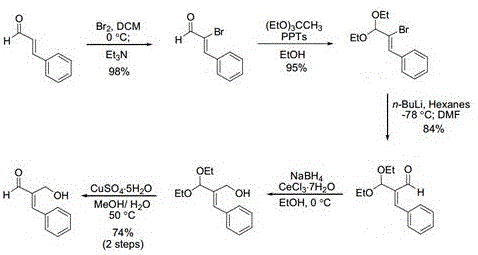
The Novel Approach
The methodology disclosed in patent CN111302914B revolutionizes this synthetic landscape by introducing a direct, one-pot coupling reaction that utilizes tetrahydrofuran as both the solvent and the reactant. This elegant strategy employs a copper catalyst system, specifically copper acetate, in conjunction with an amine ligand and a peroxide oxidant to activate the inert C-H bonds of the THF ring. This activation allows for the direct insertion of the hydroxyethyl fragment onto the aldehyde substrate without the need for pre-functionalization. The result is a dramatic reduction in synthetic steps, collapsing what was once a three or four-step sequence into a single thermal operation that proceeds with remarkable efficiency and selectivity.
Beyond the reduction in step count, this novel approach offers exceptional versatility regarding substrate tolerance. The reaction conditions are mild enough to accommodate a wide array of electronic environments on the aromatic ring, including electron-withdrawing groups like halogens and electron-donating groups like alkyl chains. Moreover, the system demonstrates robust compatibility with heterocyclic aldehydes, successfully processing substrates such as thiophene and furan derivatives which are often sensitive to harsh traditional conditions. This broad applicability makes the process highly attractive for the rapid generation of diverse compound libraries for medicinal chemistry programs, enabling faster lead optimization cycles.
Mechanistic Insights into Copper-Catalyzed C-H Functionalization
The core of this technological breakthrough lies in the sophisticated interplay between the copper catalyst and the peroxide oxidant, which together facilitate a radical-mediated C-H functionalization mechanism. Under the optimized thermal conditions of 70°C to 80°C, the copper species interacts with the peroxide to generate reactive radical intermediates capable of abstracting a hydrogen atom from the alpha-position of the tetrahydrofuran ring. This generates a stabilized carbon-centered radical on the THF molecule, which subsequently undergoes addition to the carbonyl group of the benzaldehyde substrate. This radical addition step is the key bond-forming event that constructs the carbon-carbon backbone of the target molecule, effectively merging the solvent molecule with the starting material in a highly atom-economical fashion.
Following the radical addition, the resulting intermediate undergoes a series of proton transfers and elimination steps, mediated by the amine ligand and formic acid present in the reaction mixture, to restore the conjugated double bond system characteristic of cinnamaldehyde derivatives. The presence of formic acid plays a dual role, acting both as a proton source to facilitate the final elimination and as a mild reducing agent to regenerate the active copper catalyst species, thereby sustaining the catalytic cycle. This intricate mechanistic balance ensures that the reaction proceeds with high chemoselectivity, minimizing side reactions such as over-oxidation or polymerization, which are common pitfalls in radical chemistry. The result is a clean reaction profile that yields the desired beta-hydroxyethyl cinnamaldehyde with minimal byproduct formation.
How to Synthesize Beta-Hydroxyethyl Cinnamaldehyde Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific stoichiometry and thermal parameters outlined in the patent to ensure reproducibility and safety. The process begins with the precise charging of the aldehyde substrate and the copper catalyst system into a reaction vessel equipped with temperature control. The use of tetrahydrofuran as the bulk solvent simplifies the setup, eliminating the need for additional solvent swaps. Operators must maintain the reaction temperature within the narrow window of 75°C to 80°C for a duration of approximately 3 hours to achieve optimal conversion rates. Deviations from this thermal profile can lead to incomplete reaction or degradation of the sensitive aldehyde functionality.
- Charge the reaction vessel with the aldehyde substrate (Formula I) and tetrahydrofuran solvent.
- Introduce the catalytic system comprising copper acetate, ethanolamine, formic acid, and tert-butyl peroxide.
- Heat the mixture to 75°C for 3 hours to facilitate ring-opening and coupling.
- Perform acidic workup followed by extraction and flash column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The most significant benefit stems from the utilization of tetrahydrofuran and benzaldehyde as primary feedstocks, both of which are commodity chemicals available in massive global volumes at very low cost. By eliminating the need for specialized, pre-functionalized building blocks that often carry high price tags and long lead times, manufacturers can significantly reduce their raw material expenditure. This shift to abundant, off-the-shelf reagents enhances supply chain security, mitigating the risks associated with sourcing niche intermediates from single-source suppliers who may face production disruptions.
Furthermore, the simplification of the manufacturing process from multiple steps to a single pot operation translates into drastic reductions in processing time and facility occupancy. In a commercial setting, this means higher throughput per reactor volume and lower utility consumption for heating, cooling, and agitation. The simplified workup procedure, which involves a straightforward acid wash and extraction, minimizes the consumption of auxiliary solvents and reduces the burden on waste treatment facilities. These operational efficiencies collectively contribute to a leaner manufacturing model, allowing companies to respond more agilely to market demand fluctuations while maintaining healthy profit margins in a competitive fine chemical marketplace.
- Cost Reduction in Manufacturing: The elimination of expensive halogenating agents and metal reducing agents, replaced by a catalytic amount of copper salt, fundamentally alters the cost structure of the synthesis. This removal of stoichiometric heavy metal reagents not only lowers direct material costs but also simplifies the downstream purification process, as there is no need for extensive heavy metal scavenging steps. The overall reduction in reagent complexity leads to substantial cost savings in both procurement and waste disposal, making the final product more price-competitive against natural extracts or legacy synthetic routes.
- Enhanced Supply Chain Reliability: By relying on universally available solvents and aldehydes, the supply chain becomes far more robust against geopolitical or logistical shocks. The ability to source THF and various substituted benzaldehydes from multiple global vendors ensures continuity of supply, preventing production bottlenecks. Additionally, the shorter synthesis timeline means that inventory turnover is accelerated, allowing procurement teams to operate with lower safety stock levels and reduced working capital tied up in WIP (Work In Progress) inventory, thereby improving overall cash flow efficiency.
- Scalability and Environmental Compliance: The one-pot nature of this reaction is inherently scalable, as it avoids the isolation of unstable intermediates that often pose safety risks during scale-up. The reduced generation of saline waste and heavy metal contaminants aligns perfectly with increasingly stringent environmental regulations, lowering the compliance burden and associated fees. This green chemistry profile not only future-proofs the manufacturing process against regulatory changes but also enhances the brand value of the end products by appealing to eco-conscious consumers and partners in the pharmaceutical and cosmetic sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is critical for successful technology transfer and process validation.
Q: What represents the primary advantage of this copper-catalyzed route over traditional methods?
A: The primary advantage is the direct utilization of tetrahydrofuran (THF) as a substrate in a single step, eliminating the need for multi-step halogenation and reduction sequences typically required to install the hydroxyethyl side chain.
Q: Is this synthesis method compatible with heterocyclic aldehydes?
A: Yes, the patent explicitly demonstrates high substrate universality, successfully synthesizing derivatives using thiophene-2-carbaldehyde and furfural, indicating robust tolerance for heterocyclic systems.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal conditions involve heating the reaction mixture to a temperature range of 70°C to 80°C for a duration of 3 to 6 hours, using copper acetate as the preferred catalyst in a THF solvent system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxyethyl Cinnamaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies like the one described in CN111302914B to drive innovation in the fine chemical sector. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this laboratory-scale breakthrough into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage supply is executed with precision and safety. We are committed to delivering high-purity intermediates that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite pharmaceutical and fragrance companies to collaborate with us to leverage this efficient synthesis route for their specific project needs. Whether you require custom synthesis of novel derivatives or reliable supply of the standard beta-hydroxyethyl cinnamaldehyde, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can optimize your supply chain and reduce your overall manufacturing costs.
