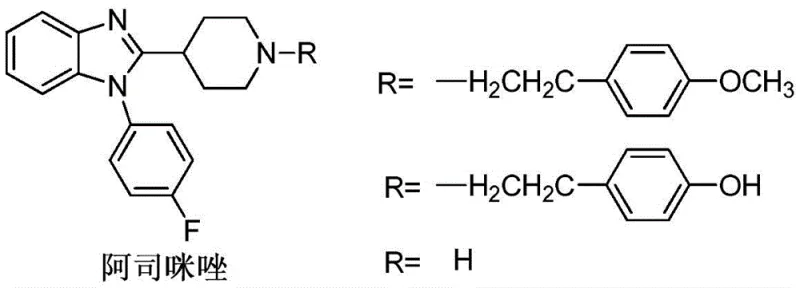
Revolutionizing Astemizole Intermediate Production with Metal-Free Organoboron Catalysis
Revolutionizing Astemizole Intermediate Production with Metal-Free Organoboron Catalysis
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the production of high-volume antihistamine agents like astemizole. As a potent and long-acting H1 receptor antagonist, astemizole relies heavily on the availability of high-purity benzimidazole derivatives as its critical building blocks. Traditional manufacturing routes have long been plagued by environmental concerns and purification bottlenecks, prompting a search for superior catalytic systems. A groundbreaking approach detailed in patent CN107188851B introduces a novel method utilizing fluorescently labeled non-metallic organic boron nitrogen Lewis acid-base bifunctional compounds. This technology represents a major leap forward, offering a pathway that effectively circumvents heavy metal pollution while ensuring exceptional selectivity and yield. By integrating advanced organocatalysis with real-time monitoring capabilities, this process addresses the dual demands of regulatory compliance and operational efficiency required by modern pharmaceutical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole derivatives, which serve as the core scaffold for astemizole, has relied heavily on harsh reaction conditions that pose significant operational and environmental risks. The traditional protocol typically involves the ring closure reaction between o-phenylenediamine and carboxylic acids or aldehydes under the catalysis of strong mineral acids such as concentrated hydrochloric acid or sulfuric acid. These aggressive reagents not only necessitate specialized corrosion-resistant equipment but also generate substantial amounts of acidic wastewater that require costly neutralization and treatment before disposal. Furthermore, alternative methods employing oxidants like iodine or hydrogen peroxide, or Lewis acids such as antimony trichloride, often suffer from poor atom economy and the inability to recover the catalyst. The presence of heavy metals like antimony or aluminum in the reaction mixture introduces a critical liability, as removing trace metal residues to meet stringent pharmacopeial standards requires additional, expensive purification steps such as chelation or extensive chromatography, thereby inflating the overall cost of goods.
The Novel Approach
In stark contrast to these legacy methods, the innovative strategy outlined in the referenced patent employs a sophisticated fluorescent-labeled organoboron nitrogen Lewis acid-base bifunctional catalyst to drive the condensation of o-phenylenediamine with various aldehydes. This metal-free system operates under remarkably mild conditions, often achieving completion at ambient temperatures around 25 °C in common solvents like toluene. The bifunctional nature of the catalyst allows it to simultaneously activate both the nucleophile and the electrophile, facilitating a highly efficient cyclization process that consistently delivers yields approaching 100% with near-perfect selectivity. Crucially, the absence of transition metals eliminates the risk of heavy metal contamination in the final API intermediate, drastically simplifying the downstream purification workflow. Moreover, the unique structural design of the catalyst enables it to be recovered and reused repeatedly; experimental data confirms that catalytic performance remains robust even after 10 consecutive cycles, offering a sustainable solution that aligns perfectly with the principles of green chemistry and circular manufacturing.
Mechanistic Insights into Organoboron Lewis Acid-Base Bifunctional Catalysis
The efficacy of this synthesis route is rooted in the unique electronic and structural properties of the organoboron catalyst, which functions through a cooperative Lewis acid-base mechanism. The central boron atom within the catalyst structure acts as a Lewis acid site, capable of coordinating with electron-rich species, while the adjacent nitrogen atoms provide Lewis basicity. This dual activation mode lowers the energy barrier for the nucleophilic attack of the diamine on the aldehyde carbonyl group, promoting the formation of the Schiff base intermediate and its subsequent oxidative dehydrogenation to the benzimidazole ring. Unlike simple Brønsted acids that protonate substrates indiscriminately, this bifunctional catalyst offers precise stereochemical and electronic control, which is essential for minimizing side reactions and byproduct formation. The rigidity of the catalyst framework ensures that the active sites are optimally positioned to stabilize the transition state, resulting in the high turnover frequencies observed in the preparation examples. This level of mechanistic sophistication allows the process to tolerate a wide range of functional groups on the aldehyde and amine substrates, making it a versatile platform for synthesizing diverse astemizole derivatives.
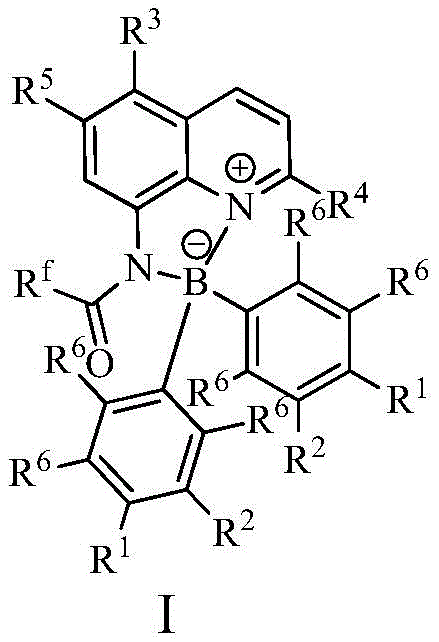
Beyond its catalytic prowess, the incorporation of a fluorescent label into the catalyst backbone provides an unprecedented advantage in quality assurance and process control. In conventional organocatalysis, determining the residual amount of catalyst in the final product often requires destructive testing or complex instrumental analysis like HPLC-MS. However, the fluorescent moiety in this specific catalyst system emits a detectable signal that correlates directly with its concentration. This feature enables manufacturers to rapidly quantify trace residues in the product stream using simple spectroscopic methods, ensuring that the final intermediate meets rigorous purity specifications without delay. From an impurity control perspective, this real-time detectability acts as a built-in safety net, allowing for immediate adjustments to the workup procedure if catalyst carryover is detected. Consequently, this mechanism not only enhances the chemical efficiency of the reaction but also fortifies the quality management system, reducing the risk of batch rejection due to unidentified organic impurities or catalyst remnants.
How to Synthesize Astemizole Key Intermediates Efficiently
Implementing this advanced synthetic route requires careful attention to reaction parameters to maximize the benefits of the organoboron catalyst. The process generally involves charging a reaction vessel with the selected aryl aldehyde and o-phenylenediamine substrates in a molar ratio that favors complete conversion, typically using a slight excess of the aldehyde. The fluorescent catalyst is then introduced at a loading as low as 0.01 to 0.12 mol%, demonstrating remarkable efficiency even at trace concentrations. The reaction mixture is stirred in a solvent such as toluene, acetonitrile, or THF at temperatures ranging from 0 to 120 °C, although many embodiments proceed effectively at room temperature. Monitoring the reaction progress via TLC ensures optimal stopping points to prevent over-reaction or degradation. Upon completion, the product is isolated through standard chromatographic separation techniques, while the catalyst, remaining in the filtrate or separable phase, is recovered for subsequent batches. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with aryl aldehyde, o-phenylenediamine, and the fluorescent organoboron catalyst in an organic solvent like toluene.
- Stir the mixture at temperatures ranging from 0 to 120 degrees Celsius for 0.5 to 12 hours until TLC indicates completion.
- Perform chromatographic separation to isolate the high-purity product and recover the catalyst for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free catalytic technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of heavy metal catalysts such as antimony or aluminum fundamentally alters the cost structure of the manufacturing process by removing the need for expensive metal scavenging resins and complex wastewater treatment protocols associated with toxic metal disposal. This simplification of the downstream processing train significantly reduces the consumption of auxiliary materials and shortens the overall production cycle time, leading to substantial cost savings in pharmaceutical intermediates manufacturing. Furthermore, the robustness of the catalyst, which can be recycled multiple times without loss of activity, decreases the dependency on continuous fresh catalyst procurement, thereby stabilizing raw material costs and insulating the supply chain from volatility in specialty chemical markets. These factors combine to create a more resilient and cost-effective production model that enhances competitiveness in the global API market.
- Cost Reduction in Manufacturing: The transition to a non-metallic organoboron catalyst system eliminates the substantial expenses associated with heavy metal removal and disposal. Traditional methods often require dedicated purification units to reduce metal content to ppm levels, involving costly reagents and extended processing times. By avoiding these steps entirely, manufacturers can achieve a leaner production process with lower utility consumption and reduced waste generation. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, further enhancing the overall material efficiency and reducing the loss of valuable starting materials during purification.
- Enhanced Supply Chain Reliability: The ability to recycle the catalyst for at least 10 cycles significantly reduces the volume of catalyst required per ton of product, mitigating supply risks associated with sourcing complex organic ligands. Since the reaction conditions are mild and do not rely on corrosive strong acids, the equipment maintenance intervals are extended, reducing unplanned downtime and ensuring consistent delivery schedules. The versatility of the catalyst in accepting various substituted aldehydes and diamines also allows for flexible production planning, enabling suppliers to respond rapidly to changes in demand for different astemizole analogues without retooling the entire synthesis line.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of common, non-hazardous solvents like toluene and the absence of exothermic hazards typical of strong acid catalysis. The green nature of the process aligns with increasingly stringent environmental regulations regarding volatile organic compounds and heavy metal discharge, future-proofing the manufacturing site against regulatory tightening. The fluorescent detection capability adds a layer of process analytical technology (PAT) that supports Quality by Design (QbD) initiatives, ensuring that scale-up does not compromise product quality and facilitating smoother regulatory filings for the final drug substance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent organoboron catalytic system for astemizole intermediate synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances is critical for R&D teams evaluating process transfer and for procurement officers assessing supplier capabilities. The technology offers a distinct advantage in terms of purity profile and operational safety, which are paramount in the highly regulated pharmaceutical sector.
Q: How does the fluorescent catalyst improve quality control?
A: The catalyst possesses intrinsic fluorescence, allowing for rapid spectral detection of trace residues in the final product without complex analytical methods.
Q: Can this catalyst be reused in industrial batches?
A: Yes, experimental data demonstrates the catalyst maintains high activity over at least 10 recycling cycles without significant loss in performance.
Q: What are the advantages over traditional acid catalysis?
A: Unlike strong acids or heavy metals, this organoboron system avoids equipment corrosion, eliminates heavy metal contamination, and simplifies wastewater treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Astemizole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free catalytic route in delivering high-value pharmaceutical intermediates with superior purity profiles. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in an industrial setting. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of astemizole intermediate meets the exacting standards required by global regulatory bodies. We are committed to leveraging this green chemistry innovation to provide our clients with a sustainable, cost-effective, and reliable supply of critical building blocks for antihistamine formulations.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of this process with your existing quality frameworks. Let us collaborate to engineer a more efficient and sustainable future for your pharmaceutical manufacturing needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
