Advanced Cyclization Technology for High-Purity Dibenzo[b,e]thioheptacyclo-11(6H)-ketone Intermediates
Introduction to Next-Generation Cyclization Technology
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable methods for synthesizing complex heterocyclic intermediates, particularly those serving as critical building blocks for next-generation antiviral therapies. Patent CN110698451A introduces a groundbreaking advancement in the preparation of substituted dibenzo[b,e]thio(oxy)heptacyclo-11(6H)-ketone compounds, which serve as essential precursors for pyrone and pyridone derivatives, including the prominent anti-influenza agent Baloxavir marboxil. This intellectual property details a novel synthetic route that replaces traditional, cumbersome cyclization agents with phosphorus pentoxide (P2O5), thereby addressing long-standing inefficiencies in waste generation and process safety. By leveraging this technology, manufacturers can achieve superior reproducibility and significantly higher purity profiles, ensuring that the final active pharmaceutical ingredients meet the stringent regulatory standards required for global market entry. The strategic shift towards this catalytic system represents a pivotal moment for supply chain stakeholders aiming to secure reliable sources of high-quality medicinal chemistry intermediates.
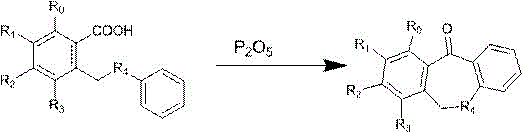
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dibenzo[b,e]thio(oxy)heptacyclo-11(6H)-ketone scaffolds has relied heavily on polyphosphoric acid (PPA) or Eaton’s reagent as cyclization promoters, methodologies that present substantial operational and environmental challenges for large-scale production facilities. Polyphosphoric acid is notoriously viscous, necessitating energy-intensive preheating protocols to improve fluidity before it can be effectively utilized in a reactor, which complicates process control and increases utility costs. Furthermore, the stoichiometric usage of PPA results in the generation of massive quantities of phosphoric acid waste liquid, documented to be more than 50 times the volume of waste generated by the newer phosphorus pentoxide method, creating a severe burden on wastewater treatment infrastructure. Similarly, while Eaton’s reagent offers cyclization capabilities, it is associated with prohibitively high raw material costs and generates waste acid volumes exceeding 20 times that of the P2O5 alternative, rendering it economically unviable for cost-sensitive commercial manufacturing campaigns.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent utilizes phosphorus pentoxide as a highly efficient cyclization catalyst, fundamentally transforming the reaction landscape into one that is safer, cleaner, and more economically attractive. This novel approach facilitates the intramolecular Friedel-Crafts acylation of substituted [(phenylthio/oxy)methyl]benzoic acids under mild to moderate thermal conditions, typically ranging from 100°C to 200°C, depending on the specific solvent system employed. The use of recyclable organic solvents such as toluene, xylene, or chlorobenzene allows for easy recovery and reuse, drastically reducing the overall solvent consumption and associated procurement costs. By eliminating the need for viscous polyacids and expensive reagents, this method not only simplifies the downstream processing steps but also ensures a dramatic reduction in the environmental footprint, aligning perfectly with modern green chemistry principles and the sustainability goals of leading pharmaceutical companies.
Mechanistic Insights into P2O5-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique ability of phosphorus pentoxide to act as a potent dehydrating agent and Lewis acid catalyst, promoting the formation of the seven-membered ring system with exceptional regioselectivity and efficiency. In this mechanism, P2O5 activates the carboxylic acid moiety of the substrate, facilitating the nucleophilic attack by the aromatic ring to close the cycle while simultaneously sequestering the water molecule produced during the condensation reaction. This dual functionality ensures that the equilibrium is driven strongly towards product formation, resulting in near-quantitative conversion rates as observed in the experimental data provided within the patent documentation. The reaction proceeds through a well-defined transition state that minimizes the formation of polymeric byproducts or isomeric impurities, which are common pitfalls in acid-catalyzed cyclizations involving sensitive fluorinated substrates.
Impurity control is further enhanced by the specific choice of reaction conditions and the inherent cleanliness of the P2O5 system, which avoids the charring and degradation often seen with harsher polyphosphoric acid treatments. For instance, in the synthesis of 7,8-difluorodibenzo[b,e]thiaheptacyclo-11(6H)-ketone, the process demonstrates an impressive ability to maintain structural integrity while achieving an HPLC purity of 99.2% and an isolated yield of 95%. 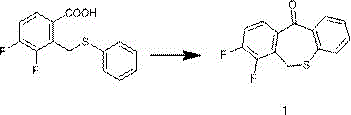 This high level of purity is critical for downstream coupling reactions in the synthesis of Baloxavir, where trace impurities can poison catalysts or complicate crystallization steps. The robustness of this mechanism across various substituents, including fluoro and hydrogen groups at different positions on the aromatic rings, underscores its versatility as a platform technology for generating diverse libraries of bioactive heterocycles.
This high level of purity is critical for downstream coupling reactions in the synthesis of Baloxavir, where trace impurities can poison catalysts or complicate crystallization steps. The robustness of this mechanism across various substituents, including fluoro and hydrogen groups at different positions on the aromatic rings, underscores its versatility as a platform technology for generating diverse libraries of bioactive heterocycles.
How to Synthesize Substituted Dibenzo Ketones Efficiently
Implementing this synthesis route in a pilot or production plant requires careful attention to the addition rate of the catalyst and the management of exothermic events during the reflux stage. The standardized protocol involves charging the substituted benzoic acid starting material and the chosen solvent into a reaction vessel, followed by the controlled, batch-wise addition of phosphorus pentoxide under vigorous stirring to ensure homogeneous mixing and heat dissipation. Once the catalyst is fully added, the mixture is heated to reflux, typically maintained for a duration of 3 to 5 hours, until liquid chromatography confirms the complete consumption of the starting material.
- Charge substituted [(phenylthio/oxy)methyl]benzoic acid and a solvent such as toluene or chlorobenzene into a reactor.
- Add phosphorus pentoxide (P2O5) in batches under stirring and heat the mixture to reflux temperature (100-200°C).
- Monitor reaction completion via HPLC, recover solvent, quench with ice water, extract with ethyl acetate, and purify to obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this P2O5-mediated cyclization process offers tangible benefits that extend far beyond simple chemical yield improvements, directly impacting the bottom line through operational efficiency and risk mitigation. The elimination of expensive and hazardous reagents like Eaton’s reagent translates into a significant reduction in raw material expenditure, while the ability to recycle solvents like toluene and xylene further drives down the variable costs associated with each production batch. Moreover, the drastic reduction in waste acid volume—reported to be over 50 times less than PPA methods—substantially lowers the costs related to waste disposal and environmental compliance, making the overall cost of goods sold (COGS) much more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The substitution of high-cost cyclization agents with economical phosphorus pentoxide creates a leaner manufacturing process that maximizes resource utilization. By avoiding the purchase of proprietary reagent mixtures and reducing the volume of waste requiring neutralization and disposal, manufacturers can achieve substantial cost savings without compromising on product quality. This economic efficiency is further amplified by the high yields obtained, which minimize the loss of valuable fluorinated starting materials and reduce the need for extensive purification steps that consume additional solvents and energy.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as phosphorus pentoxide and common industrial solvents ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized reagents. Since the raw materials are widely available from multiple global suppliers, the risk of production delays due to material shortages is significantly mitigated, guaranteeing consistent delivery schedules for downstream API manufacturers. This reliability is crucial for maintaining continuous production lines and meeting the rigorous Just-In-Time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which involves a straightforward aqueous quench and organic extraction, makes this process inherently scalable from kilogram to multi-ton quantities without the need for complex engineering modifications. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, reducing the regulatory burden and potential liability for manufacturing sites. This scalability ensures that the supply of these critical intermediates can be rapidly ramped up to meet surging demand for antiviral medications, providing a secure foundation for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the process parameters and performance metrics. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a transparent view of the method’s capabilities and limitations. Understanding these details is essential for R&D directors and process chemists who are tasked with validating new synthetic routes for regulatory filings and commercial production.
Q: Why is Phosphorus Pentoxide preferred over Polyphosphoric Acid (PPA) for this cyclization?
A: PPA is highly viscous and requires preheating, generating over 50 times more waste acid compared to the P2O5 method. P2O5 offers better fluidity, easier workup, and significantly reduced environmental impact.
Q: What represents the typical purity and yield for this synthesis route?
A: According to patent data, the process consistently achieves yields between 95% and 98.5%, with HPLC purity exceeding 99% for key intermediates like 7,8-difluorodibenzo[b,e]thiaheptacyclo-11(6H)-ketone.
Q: Is this process scalable for commercial API production?
A: Yes, the use of common solvents like toluene and xylene, which can be recycled, along with a simple aqueous quench workup, makes this method highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzo Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medicines, which is why we have invested heavily in mastering advanced synthetic technologies like the P2O5-catalyzed cyclization described in CN110698451A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of substituted dibenzo[b,e]thioheptacyclo-11(6H)-ketone meets or exceeds the required quality standards before it leaves our facility.
We invite you to collaborate with us to leverage these technical advantages for your upcoming projects, whether you are in the early stages of process development or looking to optimize an existing supply chain for cost and efficiency. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals in the competitive landscape of antiviral drug production.
