Scalable Solid-Phase Synthesis of Iseganan: Overcoming Disulfide Bond Challenges for Commercial Production
Introduction to Advanced Iseganan Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for producing complex antimicrobial peptides like Iseganan, a potent analog of porcine neutrophilic granulocyte peptide-1 with broad-spectrum activity against Gram-positive and Gram-negative bacteria. A pivotal advancement in this domain is detailed in Patent CN112500453A, which discloses a highly efficient solid-phase synthesis method specifically designed to overcome the historical challenges associated with forming multiple disulfide bonds. Unlike traditional approaches that rely on liquid-phase cyclization, this innovation utilizes a fragment condensation strategy where the critical 7-12 hexapeptide segment is synthesized and oxidized independently before being integrated into the full 17-mer sequence. This technical breakthrough not only enhances the regioselectivity of disulfide bridge formation but also establishes a reliable pathway for generating high-purity Iseganan suitable for clinical applications. For procurement leaders and R&D directors, understanding this shift from solution-phase to pure solid-phase processing is essential for evaluating potential suppliers capable of delivering consistent quality at scale.
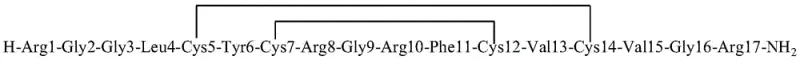
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iseganan has been plagued by significant technical hurdles, particularly regarding the precise formation of its two pairs of disulfide bonds. Prior art, such as the methods described in patent CN102336813A, typically involved synthesizing a linear peptide on a solid support followed by cleavage and subsequent cyclization in the liquid phase. This approach necessitates the selective removal of protecting groups, such as Trt at positions 5 and 14, followed by oxidation with hydrogen peroxide, and later removal of Acm groups at positions 7 and 12 for a second oxidation step. These multi-step liquid-phase reactions are notoriously difficult to control, often leading to scrambled disulfide isomers and low yields. Furthermore, the requirement to dissolve the peptide in large volumes of iodine solution for the second oxidation creates substantial downstream processing burdens, including difficult waste treatment and the high risk of iodine residue exceeding safety standards in the final API. Such inefficiencies make conventional methods economically unviable for large-scale industrial production.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN112500453A introduces a paradigm shift by executing both disulfide bond formations directly on the solid support. The process begins with the synthesis of the 7-12 fragment on a carboxyl resin, where the first disulfide bond (Cys7-Cys12) is formed via iodine oxidation while the peptide is still anchored. This protected fragment, Fmoc-(7-12)-OH, is then cleaved and used as a building block in the synthesis of the full heptadecapeptide on an amino resin. The second disulfide bond (Cys5-Cys14) is subsequently formed on this full-length resin-bound peptide. By keeping the oxidation steps on the solid phase, the method drastically reduces the formation of intermolecular polymers and ensures the correct spatial arrangement of cysteine residues. This pure solid-phase strategy eliminates the need for complex liquid-phase cyclization steps, thereby streamlining the workflow and significantly enhancing the overall purity profile of the crude peptide prior to final purification.
Mechanistic Insights into Fragment Condensation and On-Resin Oxidation
The core of this synthetic strategy lies in the meticulous application of Fmoc/tBu chemistry combined with strategic fragment condensation. The process initiates with the loading of Fmoc-Cys(Trt)-OH onto a 2-chlorotrityl chloride (2-CTC) resin or similar carboxyl resin. Sequential coupling of Fmoc-Phe-OH, Fmoc-Arg(pbf)-OH, Fmoc-Gly-OH, Fmoc-Arg(pbf)-OH, and Fmoc-Cys(Trt)-OH builds the 7-12 hexapeptide chain. Crucially, before cleaving this fragment, the resin is treated with an iodine solution in DMF at controlled temperatures (0-50°C) to oxidize the free thiol groups of the cysteine residues at positions 7 and 12. This on-resin oxidation is kinetically favorable due to the pseudo-dilution effect inherent in solid-phase synthesis, which prevents intermolecular cross-linking. Once the first disulfide bridge is secured, the fragment is cleaved under mild acidic conditions (e.g., 20-50% TFE/DCM) to yield the cyclic Fmoc-(7-12)-OH intermediate, ready for the next stage of elongation.

Following the preparation of the cyclic fragment, the synthesis proceeds on an amino resin, such as Rink Amide-AM resin, to construct the full 17-amino acid sequence. The pre-formed Fmoc-(7-12)-OH fragment is coupled into the chain alongside other Fmoc-protected amino acids like Arg, Gly, Val, Leu, and Tyr. The use of composite coupling agents containing HOBt or HOAt with DIC ensures high coupling efficiency and minimizes racemization during the assembly of this complex sequence. Once the full linear chain is assembled on the resin, a second iodine oxidation is performed to form the outer disulfide bond between Cys5 and Cys14. This sequential, dual-oxidation protocol on the solid support guarantees that the two disulfide bridges are formed in the correct register, effectively eliminating the scrambling issues common in solution-phase methods. The final cleavage using a cocktail of TFA, thioanisole, and scavengers releases the fully folded Iseganan peptide with high structural fidelity.
How to Synthesize Iseganan Efficiently
The synthesis of Iseganan via this fragment condensation route represents a sophisticated balance of chemical precision and process engineering. To achieve the high purity levels required for pharmaceutical applications, operators must strictly adhere to the stoichiometry of coupling reagents and the timing of oxidation steps as defined in the patent literature. The utilization of Fmoc-(7-12)-OH as a distinct intermediate allows for quality control checkpoints before the final assembly, reducing the risk of batch failure. Detailed standard operating procedures regarding resin swelling, coupling times, and washing protocols are critical to minimizing deletion sequences. For a comprehensive breakdown of the specific reaction conditions, reagent ratios, and step-by-step operational guidelines, please refer to the standardized synthesis protocol below.
- Synthesize the 7-12 hexapeptide fragment on a carboxyl resin using Fmoc-amino acids and oxidize the first disulfide bond (Cys7-Cys12) on-resin.
- Cleave the protected fragment to obtain Fmoc-(7-12)-OH and couple it to an amino resin along with remaining N-terminal amino acids.
- Perform the second on-resin iodine oxidation to form the Cys5-Cys14 disulfide bond, followed by global deprotection and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the solid-phase fragment synthesis method described in Patent CN112500453A offers compelling economic and logistical benefits. Traditional liquid-phase cyclization methods often suffer from low yields and extensive purification requirements, which drive up the cost of goods sold (COGS) and extend lead times. By adopting this novel on-resin oxidation strategy, manufacturers can achieve a more streamlined production process that inherently reduces waste generation and solvent consumption. The elimination of difficult post-oxidation workups, such as removing excess iodine from solution, translates directly into reduced processing time and lower operational expenditures. Furthermore, the improved regioselectivity means that a higher percentage of the crude material meets purity specifications, thereby increasing the overall throughput of the manufacturing facility without requiring additional capital investment in purification equipment.
- Cost Reduction in Manufacturing: The implementation of pure solid-phase synthesis eliminates the need for expensive and hazardous liquid-phase cyclization reagents and the associated containment infrastructure. By forming disulfide bonds on the resin, the process avoids the significant product loss often encountered during solution-phase precipitation and redissolution steps. This efficiency gain leads to substantial cost savings in raw material utilization and waste disposal, making the production of high-purity Iseganan more economically sustainable. Additionally, the simplified workflow reduces labor hours per batch, further contributing to a lower overall manufacturing cost structure.
- Enhanced Supply Chain Reliability: The robustness of the fragment condensation approach ensures greater batch-to-batch consistency, which is critical for maintaining a stable supply of API intermediates. Since the critical 7-12 fragment can be synthesized and validated independently, any potential issues can be identified and resolved early in the process, preventing the loss of valuable full-length peptide material. This modularity enhances supply chain resilience by allowing for the stocking of key intermediates, thereby reducing the risk of production delays caused by raw material shortages or synthesis failures. Consequently, partners can rely on more predictable delivery schedules and consistent product availability.
- Scalability and Environmental Compliance: Scaling peptide synthesis from laboratory to commercial production often presents significant challenges, particularly regarding exothermic reactions and solvent handling. The solid-phase method described herein operates under mild conditions (0-50°C) and utilizes standard peptide synthesis equipment, facilitating a smoother scale-up from kilograms to metric tons. Moreover, the reduction in solvent usage and the avoidance of heavy iodine contamination in waste streams align with increasingly stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risks but also supports corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Iseganan using the advanced solid-phase methodology. These insights are derived directly from the technical specifications and beneficial effects reported in the underlying patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this peptide into their development pipelines. The answers provided reflect the current state-of-the-art in peptide manufacturing as defined by the referenced intellectual property.
Q: Why is on-resin disulfide bond formation preferred for Iseganan?
A: On-resin oxidation prevents intermolecular polymerization and ensures correct pairing of cysteine residues (Cys5-Cys14 and Cys7-Cys12), significantly improving regioselectivity and final product purity compared to solution-phase cyclization.
Q: What are the advantages of the fragment condensation strategy?
A: Synthesizing the 7-12 fragment separately allows for early verification of the internal disulfide bond structure. This modular approach reduces the risk of deletion sequences in the final 17-mer peptide and simplifies purification.
Q: How does this method address iodine residue issues?
A: By performing oxidation on the solid support followed by thorough washing before cleavage, the method minimizes iodine carryover into the final crude peptide, avoiding the extensive post-treatment required in traditional liquid-phase oxidation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iseganan Supplier
As the demand for novel antimicrobial therapies grows, securing a manufacturing partner with deep expertise in complex peptide synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced technologies like the solid-phase fragment condensation method to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global pharmaceutical clients. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art HPLC and mass spectrometry to verify the correct formation of disulfide bonds and the absence of impurities in every batch of Iseganan we produce.
We invite you to collaborate with us to optimize your supply chain for this critical antibiotic candidate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient synthesis routes can reduce your overall project costs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-quality peptide intermediates and APIs.
