Advanced Metal-Free Synthesis of 8-Cyclohexyl-2-Fluoro-Vidarabine for Commercial Scale-Up
The pharmaceutical landscape for anti-leukemia therapeutics is constantly evolving, driven by the urgent need for agents that overcome drug resistance while maintaining stringent safety profiles. Patent CN103467552A introduces a groundbreaking synthesis method for 8-cyclohexyl-2-fluoro-vidarabine, a potent nucleoside analog demonstrating superior inhibitory activity against HL-60 leukemia cells. This technical disclosure addresses critical bottlenecks in nucleoside modification, specifically eliminating the reliance on toxic heavy metal catalysts that have historically plagued the production of complex arabinoside derivatives. For R&D directors and procurement specialists alike, this metal-free methodology represents a paradigm shift towards greener, more cost-effective, and scalable manufacturing of high-value pharmaceutical intermediates. The process not only simplifies the operational workflow but also ensures that the final active pharmaceutical ingredient (API) meets rigorous purity specifications required for clinical progression.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for modifying the C8 position of purine nucleosides often depend heavily on transition metal catalysis, such as palladium or copper-mediated cross-coupling reactions. These conventional approaches introduce significant downstream processing challenges, primarily due to the difficulty in completely removing trace metal residues to parts-per-million levels mandated by regulatory bodies like the FDA and EMA. Furthermore, these metal-catalyzed reactions frequently require expensive ligands, inert atmospheres, and strictly anhydrous conditions, which drastically inflate the cost of goods sold (COGS) and complicate supply chain logistics. The presence of heavy metals also poses environmental hazards, necessitating costly waste treatment protocols that hinder the sustainability goals of modern chemical manufacturing. Additionally, prior art methods for synthesizing similar vidarabine derivatives often suffer from low overall yields and poor regioselectivity, leading to complex impurity profiles that are difficult to characterize and control during scale-up.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a streamlined, metal-free strategy that leverages free radical chemistry to achieve direct C-H functionalization. This innovative route bypasses the need for pre-halogenated substrates and expensive catalysts, relying instead on readily available reagents like cyclohexane and tert-butyl peroxide. The process begins with a robust protection strategy followed by a high-temperature radical substitution that efficiently installs the cyclohexyl group at the C8 position of the purine ring. 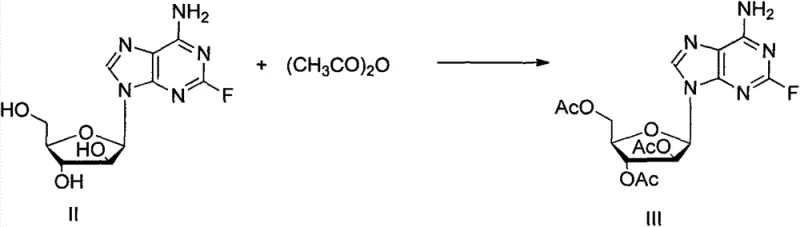 This approach not only simplifies the reaction setup by removing the need for specialized catalytic equipment but also significantly reduces the environmental footprint of the synthesis. By avoiding heavy metals entirely, the process inherently produces a cleaner crude product, minimizing the burden on purification units and ensuring a safer final product for patients. The operational simplicity allows for easier technology transfer from laboratory to pilot plant, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their production capabilities.
This approach not only simplifies the reaction setup by removing the need for specialized catalytic equipment but also significantly reduces the environmental footprint of the synthesis. By avoiding heavy metals entirely, the process inherently produces a cleaner crude product, minimizing the burden on purification units and ensuring a safer final product for patients. The operational simplicity allows for easier technology transfer from laboratory to pilot plant, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their production capabilities.
Mechanistic Insights into Free Radical C8-Cyclohexylation
The core chemical transformation in this synthesis is a thermal free radical substitution reaction, a mechanism that offers distinct advantages over ionic pathways for modifying electron-rich heterocycles. In the second step of the synthesis, the triacetyl-protected intermediate is subjected to elevated temperatures (140°C) in the presence of cyclohexane and tert-butyl peroxide. The peroxide acts as a radical initiator, undergoing homolytic cleavage to generate tert-butoxy radicals which subsequently abstract a hydrogen atom from the cyclohexane solvent, creating a cyclohexyl radical. This highly reactive carbon-centered radical then attacks the electron-deficient C8 position of the purine ring, displacing a hydrogen atom and forming the new carbon-carbon bond. This direct C-H activation strategy is particularly elegant because it avoids the multi-step sequences typically required to install alkyl groups on nucleobases, thereby reducing the number of unit operations and potential points of failure in the manufacturing process.
Impurity control is intrinsically managed through the strategic use of protecting groups and the specificity of the radical mechanism. The initial acetylation of the sugar hydroxyl groups serves a dual purpose: it prevents unwanted side reactions at the ribose moiety during the harsh radical conditions and enhances the solubility of the nucleoside in the non-polar cyclohexane medium. 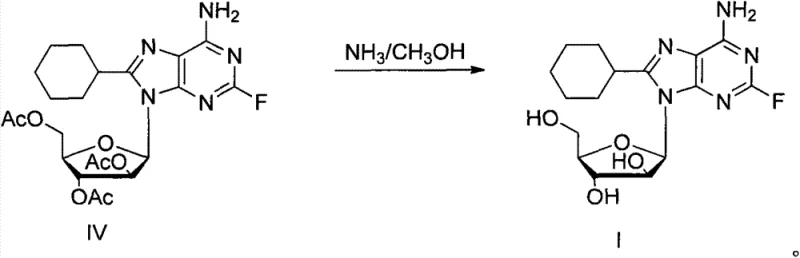 The subsequent deprotection step using ammonia in methanol is highly selective for ester hydrolysis, leaving the glycosidic bond and the newly formed C8-C bond intact. This orthogonality ensures that the final product, 8-cyclohexyl-2-fluoro-vidarabine, is obtained with high structural fidelity. The absence of metal catalysts means there are no metal-coordinated byproducts to separate, resulting in a much simpler impurity profile that facilitates easier crystallization and purification. For quality control teams, this translates to more consistent batch-to-batch reproducibility and reduced analytical testing burdens.
The subsequent deprotection step using ammonia in methanol is highly selective for ester hydrolysis, leaving the glycosidic bond and the newly formed C8-C bond intact. This orthogonality ensures that the final product, 8-cyclohexyl-2-fluoro-vidarabine, is obtained with high structural fidelity. The absence of metal catalysts means there are no metal-coordinated byproducts to separate, resulting in a much simpler impurity profile that facilitates easier crystallization and purification. For quality control teams, this translates to more consistent batch-to-batch reproducibility and reduced analytical testing burdens.
How to Synthesize 8-Cyclohexyl-2-Fluoro-Vidarabine Efficiently
The synthesis protocol described in the patent provides a clear, three-step roadmap for producing this high-value anti-leukemia intermediate with excellent overall efficiency. The process is designed to be operationally simple, utilizing standard laboratory glassware and common industrial solvents, which lowers the barrier for implementation in diverse manufacturing settings. The detailed standardized synthesis steps below outline the precise conditions, stoichiometry, and workup procedures required to achieve the reported high yields.
- Protect the hydroxyl groups of 2-fluoro-vidarabine (II) using acetic anhydride and triethylamine with DMAP catalysis at 60°C to obtain the triacetyl protected intermediate (III) with high yield.
- Perform a free radical substitution reaction by heating the protected intermediate (III) with cyclohexane and tert-butyl peroxide at 140°C for 24 hours to introduce the cyclohexyl group at the C8 position, yielding compound IV.
- Execute the final deprotection step by reacting compound IV with ammonia in methanol at room temperature for 24 hours to remove acetyl groups, crystallizing the final product 8-cyclohexyl-2-fluoro-vidarabine (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers substantial strategic benefits that extend beyond mere chemical elegance. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements. Furthermore, the simplified purification requirements reduce the consumption of chromatography media and solvents, leading to a drastic reduction in waste generation and disposal costs. This aligns perfectly with global initiatives for green chemistry and sustainable manufacturing, potentially qualifying the production facility for environmental incentives and improving the corporate social responsibility (CSR) profile of the supply chain.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts and expensive ligands directly lowers the raw material costs per kilogram of the final product. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor hours and equipment usage time, contributing to significant operational expenditure savings. The high yields reported in each step (92%, 85%, and 87%) indicate a highly efficient atom economy, minimizing the loss of valuable starting materials and maximizing output from existing reactor volumes.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as acetic anhydride, cyclohexane, and ammonia, are commodity chemicals with robust, global supply chains, ensuring consistent availability and price stability. Unlike specialized catalysts that may have long lead times or single-source dependencies, these bulk chemicals can be sourced from multiple vendors, reducing the risk of production stoppages due to material shortages. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process conditions, particularly the use of thermal radical initiation rather than sensitive catalytic cycles, are inherently more robust and easier to scale from gram to ton quantities. The absence of toxic heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive validation of metal clearance methods or monitoring of effluent for metal content. This facilitates faster regulatory approval for new drug applications (NDAs) and reduces the environmental permitting burden for manufacturing sites, accelerating the time-to-market for the final therapeutic agent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 8-cyclohexyl-2-fluoro-vidarabine, based on the specific details provided in the patent literature. These insights are curated to assist technical decision-makers in evaluating the feasibility and value proposition of this synthesis route for their specific project needs.
Q: Why is the metal-free synthesis route preferred for 8-cyclohexyl-2-fluoro-vidarabine?
A: Conventional synthesis methods often rely on heavy metal catalysts which leave toxic residues difficult to remove to pharmaceutical standards. The novel route disclosed in patent CN103467552A eliminates heavy metals entirely, ensuring higher safety profiles and simplified purification processes suitable for clinical applications.
Q: What is the critical chemical transformation in this synthesis pathway?
A: The core innovation lies in the direct C8-H functionalization of the purine ring via a free radical mechanism. By utilizing tert-butyl peroxide as a radical initiator in cyclohexane at elevated temperatures, the process efficiently installs the cyclohexyl moiety without requiring pre-functionalized halogenated precursors.
Q: What represents the primary therapeutic potential of this compound?
A: Biological testing indicates potent inhibitory activity against HL-60 leukemia cells with an IC50 value of 0.36 μM, outperforming standard treatments like doxorubicin in specific assays. This suggests significant potential for developing next-generation anti-leukemia therapeutics with improved efficacy profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Cyclohexyl-2-Fluoro-Vidarabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, metal-free synthesis routes to ensure the highest quality and safety for oncology drug candidates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 8-cyclohexyl-2-fluoro-vidarabine that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation for comprehensive impurity profiling.
We invite you to collaborate with us to leverage this innovative technology for your anti-leukemia drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your project timelines and enhance your competitive advantage in the pharmaceutical market.
