Advanced Aqueous Synthesis of Gabapentin and Pregabalin Intermediates for Commercial Scale-Up
Advanced Aqueous Synthesis of Gabapentin and Pregabalin Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, safe, and cost-effective pathways for the production of high-volume therapeutic agents, particularly for neurological disorders where demand is escalating. Patent CN1642899A introduces a transformative methodology for the synthesis of substituted gamma-amino acids, specifically targeting the blockbuster drugs gabapentin and pregabalin. This intellectual property delineates a novel process for producing substituted acrylates via Wadsworth-Emmons olefination, distinctively utilizing alkali metal carbonates as bases in aqueous solvents rather than traditional alkali metal hydrides in anhydrous conditions. This shift represents a paradigm change in process chemistry, moving away from hazardous, moisture-sensitive reagents toward benign, water-based systems that facilitate easier handling and waste management. The strategic implementation of this technology allows manufacturers to bypass the stringent requirements for absolute organic solvents and protective atmospheres during the critical carbon-carbon bond-forming step.
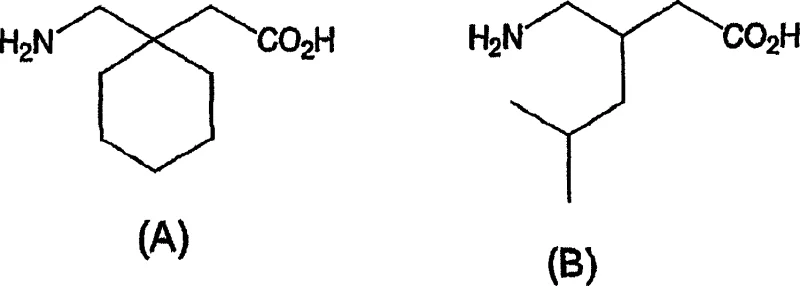
By leveraging this aqueous protocol, producers can achieve high yields of high-purity substituted acrylates, which serve as the foundational building blocks for the subsequent synthesis of complex amino acid derivatives. The ability to execute these reactions in water not only enhances operational safety but also aligns with green chemistry principles by reducing the reliance on volatile organic compounds (VOCs). For R&D directors and process engineers, this patent offers a validated route that simplifies the synthetic sequence while maintaining rigorous quality standards required for active pharmaceutical ingredients (APIs). The following analysis dissects the technical merits of this approach, contrasting it with legacy methods to highlight its potential for optimizing supply chains and reducing manufacturing overheads in the competitive landscape of generic pharmaceutical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates for gabapentin and pregabalin has relied heavily on the use of strong, moisture-sensitive bases such as sodium hydride (NaH) in absolute organic solvents like tetrahydrofuran (THF). This conventional approach imposes severe logistical and safety constraints on manufacturing facilities, necessitating the use of specialized equipment capable of maintaining strictly anhydrous conditions and inert gas atmospheres throughout the reaction. The handling of sodium hydride carries inherent risks due to its pyrophoric nature, requiring meticulous safety protocols and increasing the complexity of standard operating procedures. Furthermore, the requirement for absolute solvents demands energy-intensive drying processes and rigorous quality control to prevent moisture ingress, which can quench the reaction or lead to hazardous hydrogen gas evolution. These factors collectively contribute to elevated operational expenditures and extended cycle times, creating bottlenecks in large-scale production environments where throughput and safety are paramount concerns for supply chain managers.
The Novel Approach
In stark contrast, the methodology described in CN1642899A replaces the hazardous alkali metal hydrides with readily available alkali metal carbonates, such as potassium carbonate, and utilizes water as the primary solvent medium for the initial olefination step. This innovation fundamentally alters the risk profile of the synthesis, eliminating the need for protective gases and absolute solvents during the formation of the critical acrylate intermediate. The reaction proceeds efficiently at temperatures ranging from 0°C to 25°C, often facilitated by simple ice cooling, which is far more energy-efficient than the cryogenic conditions sometimes required for sensitive organometallic reactions. As illustrated in the specific embodiment for gabapentin precursors, the reaction between cyclohexanone and triethyl phosphonoacetate in the presence of potassium carbonate and water yields ethyl cyclohexylidene acetate with exceptional efficiency.
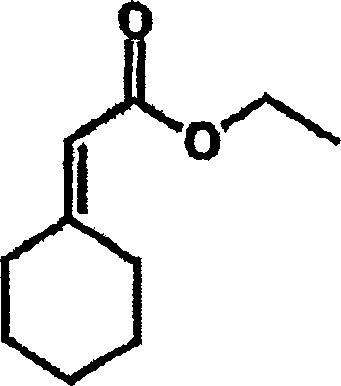
This aqueous system not only simplifies the workup procedure, typically involving straightforward extraction with ethers, but also ensures that the resulting acrylates possess high purity suitable for immediate downstream processing. By removing the dependency on expensive drying agents and inert gas manifolds, this novel approach drastically reduces the capital and operational intensity of the manufacturing process. For procurement teams, this translates to a more resilient supply chain less vulnerable to disruptions in the availability of specialized anhydrous solvents or hazardous reagents, thereby ensuring consistent production continuity for high-demand pharmaceutical intermediates.
Mechanistic Insights into Aqueous Wadsworth-Emmons Olefination
The core chemical transformation driving this process is the Wadsworth-Emmons olefination, a variant of the Wittig reaction that utilizes phosphonate carbanions to convert ketones or aldehydes into alpha,beta-unsaturated esters. In the context of this patent, the mechanism involves the deprotonation of trialkyl phosphonoacetates by the carbonate base to generate the reactive phosphonate anion. While carbonates are generally considered weaker bases than hydrides, the specific reaction conditions and the acidity of the phosphonate alpha-protons allow for sufficient anion generation to drive the nucleophilic attack on the carbonyl carbon of the ketone or aldehyde substrate. The use of water as a solvent is particularly ingenious; it stabilizes the ionic intermediates and facilitates the dissolution of the inorganic carbonate base, creating a homogeneous or semi-homogeneous reaction environment that promotes efficient mass transfer. This mechanistic pathway avoids the formation of phosphine oxide byproducts that are often difficult to remove in traditional Wittig reactions, instead yielding water-soluble phosphate salts that are easily separated during the aqueous workup phase.
Impurity control is a critical aspect of this mechanism, as the high selectivity of the carbonate-mediated olefination minimizes side reactions such as self-condensation of the aldehyde or over-alkylation. The patent data indicates that the crude acrylates produced contain only trace amounts of starting materials, allowing them to be carried forward to the Michael addition step without rigorous purification. This "telescoping" capability is vital for maintaining high overall yields and reducing solvent consumption. Following the olefination, the subsequent Michael addition of nitromethane, catalyzed by tetrabutylammonium fluoride, proceeds with high regioselectivity to install the nitromethyl group. The final stages involve catalytic hydrogenation using Raney nickel to reduce the nitro group and cyclize the molecule into a gamma-lactam, followed by acidic hydrolysis to open the ring and form the final amino acid hydrochloride salt. Each step is optimized to preserve the structural integrity of the molecule while maximizing atom economy.
How to Synthesize Gabapentin Intermediates Efficiently
The synthesis of gabapentin intermediates via this patented route offers a streamlined protocol that balances chemical efficiency with operational simplicity. The process begins with the preparation of the acrylate backbone through the aqueous olefination described previously, setting the stage for the introduction of the nitrogen functionality. Detailed standard operating procedures for this synthesis emphasize the importance of temperature control during the initial mixing of reagents to manage the exotherm, followed by a prolonged stirring period to ensure complete conversion. The subsequent steps involving nitromethane addition and hydrogenation require standard laboratory safety precautions but benefit from the high purity of the incoming intermediate. For a comprehensive understanding of the specific stoichiometric ratios, reaction times, and isolation techniques required to replicate these results in a pilot or production plant, please refer to the standardized synthesis guide below.
- Perform Wadsworth-Emmons olefination by mixing cyclohexanone and triethyl phosphonoacetate with potassium carbonate in water at 0-25°C to form ethyl cyclohexylidene acetate.
- Conduct Michael addition by reacting the acrylate intermediate with nitromethane using tetrabutylammonium fluoride catalyst in tetrahydrofuran.
- Reduce the nitro compound to a lactam using Raney nickel and hydrogen, followed by acidic hydrolysis to yield the final gamma-amino acid hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of the aqueous Wadsworth-Emmons protocol described in CN1642899A presents compelling economic and logistical benefits for organizations managing the procurement and supply of pharmaceutical intermediates. By fundamentally altering the reagent profile of the synthesis, this method addresses several chronic pain points associated with the traditional manufacturing of gabapentin and pregabalin. The elimination of pyrophoric reagents and the shift to water-based chemistry significantly lower the barrier to entry for contract manufacturing organizations (CMOs) that may lack specialized infrastructure for handling hazardous materials. This accessibility broadens the potential supplier base, fostering competition and enhancing supply security. Furthermore, the simplified workup and purification requirements reduce the consumption of organic solvents and energy, directly impacting the variable costs associated with production. These factors combine to create a more agile and cost-efficient manufacturing model that is better suited to the fluctuating demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The replacement of sodium hydride and absolute tetrahydrofuran with potassium carbonate and water results in substantial raw material cost savings, as inorganic carbonates are significantly cheaper and more abundant than specialized organometallic bases. Additionally, the removal of solvent drying steps and the reduced need for inert gas purging lower utility costs and equipment maintenance expenses. The high yields reported in the patent examples suggest that material throughput is maximized, minimizing waste disposal costs associated with failed batches or low-conversion reactions. This economic efficiency allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like potassium carbonate and water mitigates the risk of supply chain disruptions often associated with specialized, hazardous reagents that have limited suppliers and strict transportation regulations. The stability of the reagents allows for longer shelf life and easier storage, reducing inventory write-offs and ensuring that production lines can remain operational even during periods of logistical strain. The robustness of the aqueous reaction conditions also means that the process is less sensitive to minor variations in environmental conditions, leading to more consistent batch-to-batch quality and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The use of water as a solvent inherently simplifies the scale-up process, as heat dissipation and mixing are more manageable in aqueous systems compared to viscous or flammable organic media. This facilitates the transition from laboratory scale to multi-ton commercial production with minimal process re-engineering. From an environmental perspective, the reduction in volatile organic compound (VOC) emissions and the generation of less hazardous waste streams align with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and enhances the sustainability profile of the final product, which is an increasingly important factor for pharmaceutical buyers evaluating their supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing workflows or for procurement specialists assessing the long-term viability of suppliers utilizing this technology.
Q: What are the safety advantages of using potassium carbonate in water versus sodium hydride?
A: Using potassium carbonate in water eliminates the need for pyrophoric sodium hydride and absolute organic solvents, significantly reducing fire hazards and the requirement for inert gas protection during the initial olefination step.
Q: Can this aqueous process be scaled for industrial production?
A: Yes, the patent explicitly demonstrates that the olefination process performs effectively on both 0.5 molar and multi-molar scales without compromising purity or yield, making it highly suitable for commercial manufacturing.
Q: How does this method impact the purity of the final gamma-amino acid?
A: The method produces substituted acrylates in high purity which can be used immediately in subsequent steps without extensive purification, leading to high-quality final products like gabapentin hydrochloride with minimized impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gabapentin and Pregabalin Supplier
The technological advancements detailed in CN1642899A underscore the potential for more efficient and sustainable production of critical neurological therapeutics. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative process chemistry in delivering high-quality intermediates to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the identity and quality of every batch, adhering to the highest international standards for pharmaceutical manufacturing. We are committed to leveraging advanced synthetic routes to optimize cost structures and enhance supply security for our partners.
We invite you to collaborate with us to explore how this aqueous synthesis technology can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this greener, more efficient process. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can serve as your strategic partner in the successful commercialization of gabapentin and pregabalin.
